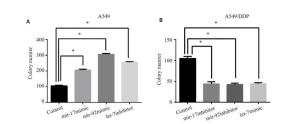
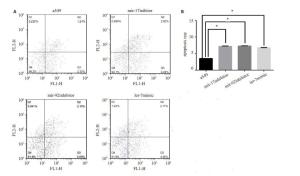
The relationship between the expression level of mir-17-5p、mir-92a、let-7b and the cisplatin-resistant
-
摘要:
目的探讨mir-17-5p、mir-92a、let-7b表达水平与非小细胞肺癌顺铂耐药关系。 方法以人非小细胞肺癌细胞系A549及其耐药株A549/DDP为研究对象,采用RT-PCR法检测mir-17-5p、mir-92a及let-7b在细胞中的表达水平,采用cck8检测其细胞存活情况,采用细胞克隆平台方法,检测转染前后细胞的增殖情况,采用流式细胞仪检测转染前后细胞的凋亡情况。 结果(1) A549/DDP细胞mir-17-5p的表达水平是A549细胞的2.11±0.25倍(P<0.05);A549/DDP细胞mir-92a的表达水平是A549细胞的 7.40 ± 1.05 倍(P<0.05);而A549/DDP 细胞let-7b 的表达水平是A549 细胞的(26.54 ± 2.90)%(P<0.05);(2)A549 转染mir-17-5pmimic,mir-92a mimic 以及let-7b inhibitor 后对顺铂的敏感性下降(P<0.05);A549/ddp 转染mir-17-5p inhibitor, mir-92a inhibitor以及let-7b mimic后对顺铂的敏感性增加(P<0.05);(3)A549转染mir-17-5p mimic,mir-92a mimic以及let-7b inhibitor 后,细胞形成克隆集落数量数量多于对照组(P<0.05);而A549/ddp 转染mir-17-5p inhibitor,mir-92a inhibitor 以及 let-7b mimic 后,细胞形成克隆集落数量数量少于对照组(P<0.05);(4)A549 转染mir-17-5p mimic,mir-92a mimic 以及let-7b inhibitor后,细胞凋亡率明显低于对照组(P<0.05);而a549/ddp转染mir-17-5p inhibitor,mir-92a inhibitor以及let-7b mimic后, 细胞凋亡率明显高于对照组(P<0.05)。结论Mir-17-5p、mir-92a表达水平升高,let-7b表达水平下降,可以促进肺癌细胞增殖, 抑制其凋亡以及使肺癌细胞对顺铂敏感性下降。 Abstract:Objective To discuss miR-17, miR-92 and let-7 expression in non small cell lung cancer and cisplatin resistance and clinical application. To provide experimental data for prediction of lung cancer patients as a sign of cisplatin chemotherapy sensitivity. Methods Non small cell lung cancer cell line A549 and its resistant strain A549/DDP were used as the cell research object. The expression level of miR-17, miR-92 and let-7 in the cell was detected by RT-PCR method. CCK8 was used to detect the survival of the transfected cells after Cisplatin maintenance culture compare to untransfected cells. After transfection, cell proliferation was detected by Cell plate cloning experiments and apoptosis rate was measured by Flow cytometry. Result (1) The expression level of miR-17 in A549/DDP cells was (2.11 ± 0.25) times (P<0.05) of A549 cells, and the expression level of miR-92 in A549/DDP cells was (7.40±1.05) times of A549 cells (P<0.05). However, the expression of let-7 in A549/DDP cells was (26.54 ± 2.90)% (P<0.05) of A549 cells; (2) A549 cells transfected with mir-17b, 92a mimic and let-7 microRNA inhibitor, compared to control group, showed a lower sensitivity to cisplatin (P<0.05); while the sensitivity to cisplatin of A549/DDP cells transfected with mir-17b, 92a inhibitor and let-7 miRNA increased(P<0.05); (3) A549 transfected with mir-17b, 92a mimic and let-7 microRNA inhibitor, the cells formed colony number is greater than that of the control group, the difference is statistically significant (P<0.05); and A549/DDP transfectd with mir-17b, 92a inhibitor and let-7 miRNA, the cells formed clones colony quantity less than that of the control group, the difference is statistically significant (P<0.05); (4) The apoptosis rate of A549 cells transfected with mir-17b, 92a mimic and let-7 microRNA inhibitor, apoptosis rate was significantly lower than the control group, the difference has statistical significance (P<0.05); For A549/DDP cells transfected with mir-17b, 92a inhibitor and let-7 miRNA, the apoptosis rate was significantly higher than that of the control group, the difference was statistically significant (P<0.05). Conclusion Mir-17, miR-92 expression level increased, let-7 expression level decreased, can promote the proliferation of lung cancer cells, inhibit apoptosis, as well as the decrease of lung cancer cell sensitivity to cisplatin. -
Key words:
- lung cancer /
- miR-17 /
- miR-92 /
- let-7 /
- cisplatin /
- sensitivity
-
[1] Jemal A, Thun MJ, Ries LA, et al. Annual report to the nation on the status of cancer, 1975-2005, featuring trends in lung cancer, tobacco use, and tobacco control[J]. J Natl Cancer Inst, 2008, 100 (23): 1672-94. [2] Parkin DM, Pisani P, Ferlay J. Global cancer statistics[J]. CA Cancer J Clin, 1999, 49(1): 33-64, 1. [3] Xing L, Todd NW, Yu L, et al. Early detection of squamous cell lung cancer in sputum by a panel of microRNA markers[J]. Mod Pathol, 2010, 23(8): 1157-64. [4] Xie Y, Todd NW, Liu Z, et al. Altered miRNA expression in sputum for diagnosis of non-small cell lung cancer[J]. Lung Cancer, 2010, 67(2): 170-6. [5] Shen J, Todd NW, Zhang H, et al. Plasma microRNAs as potential biomarkers for non-small-cell lung cancer [J]. Laboratory Investigation, 2011, 91(4): 579-87. [6] Yanaihara N, Caplen N, Bowman E, et al. Unique microRNA molecular profiles in lung cancer diagnosis and prognosis[J]. Cancer Cell, 2006, 9(3): 189-98. [7] Patnaik SK, Kannisto E, Knudsen S, et al. Evaluation of MicroRNA expression profiles that May predict recurrence of localized stage I Non-Small cell lung cancer after surgical resection[J]. Cancer Res, 2010, 70(1): 36-45. [8] Lebanony D, Benjamin H, Gilad S, et al. Diagnostic assay based on hsa-miR-205 expression distinguishes squamous from nonsquamous non-small-cell lung carcinoma[J]. J Clin Oncol, 2009, 27 (4): 2030-7. [9] Zhao J, Fu WF, Liao HY, et al. The regulatory and predictive functions of miR-17 and miR-92 families on cisplatin resistance of non-small cell lung cancer[J]. BMC Cancer, 2015, 15(7): 731. [10] Jiang ZY, Yin J, Fu WF, et al. miRNA 17 family regulates Cisplatin- Resistant and metastasis by targeting TGFbetaR2 in NSCLC[J]. PLoS One, 2014, 9(4): e94639. [11] Sarkar FH, Li Y, Wang Z, et al. Implication of microRNAs in drug resistance for designing novel cancer therapy[J]. Drug Resist Updat, 2010, 13(3): 57-66. [12] Fennell DA, Summers Y, Cadranel J, et al. Cisplatin in the modern era: The backbone of first-line chemotherapy for non-small cell lung cancer[J]. Cancer Treat Rev, 2016, 44(5): 42-50. [13] 郗照勇, 刘扬中. 顺铂耐药的分子机制[J]. 中国科学: 化学, 2014(04): 410-22. [14] Dasari S, Tchounwou PB. Cisplatin in cancer therapy: Molecular mechanisms of action[J]. Eur J Pharmacol, 2014, 740(5): 364-78. [15] Galasso M, Sana ME, Volinia S. Non-coding RNAs: a key to future personalized molecular therapy[J]. Genome Med, 2010, 42(2): 12. -







 下载:
下载: