Oxaliplatin-based versus cisplatin-based chemotherapy as first-line treatment in advanced gastric cancer: a meta-analysis.
-
摘要:
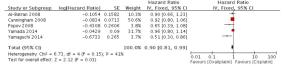
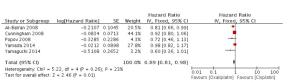
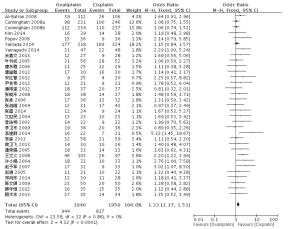
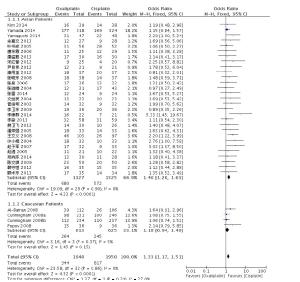
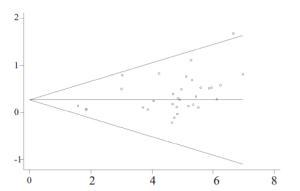
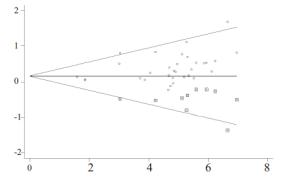
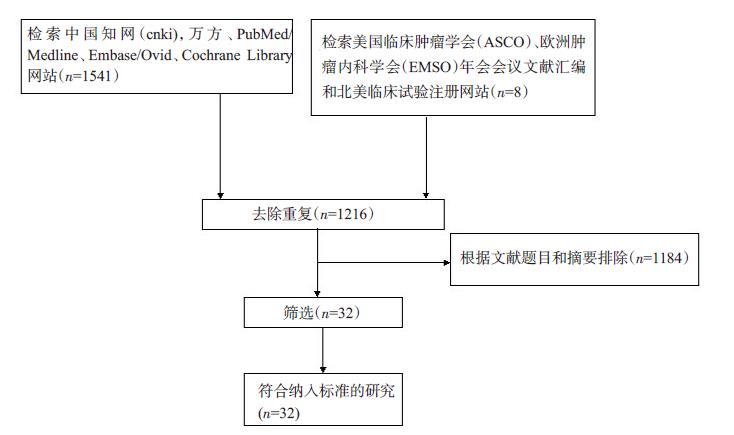
目的探讨奥沙利铂与顺铂治疗进展期胃癌的疗效、预后及不良反应的优劣。 方法检索中国知网(CNKI)、万方、PubMed/ Medline、Embase/Ovid、Cochrane Library、历年美国临床肿瘤学会(ASCO)和欧洲肿瘤内科学会(EMSO)年会文献汇编及北美临床试验注册网。评价指标为总体生存期(OS)、无进展生存期(FPS)、客观缓解率(ORR)及严重不良反应。 结果纳入32篇文献,共3994位患者参与(奥沙利铂组1992例,顺铂组2002例)。采用含奥沙利铂方案的总生存期(HR=0.90, 95%CI 0.81~0.99)、无进展生存期(HR=0.89, 95%CI 0.81~0.98)、有效率(OR=1.33, 95%CI 1.17~1.51)明显优于顺铂组。白人亚组和亚洲人种亚组均受益于含奥沙利铂的化疗方案(OR=1.55, 95%CI 1.05~2.01)。严重不良反应方面奥沙利铂方案较少发生的贫血(OR=0.49, 95%CI 0.38~0.62)、白细胞减少(OR=0.37, 95%CI 0.27~0.51)、中性粒细胞减少(OR=0.49, 95%CI 0.40~0.59)等骨髓抑制,易发生严重的恶心/呕吐(OR=0.37, 95%CI 0.21~0.61)和周围神经病变(OR=7.16, 95%CI 4.06~12.64),差异有统计学意义。 结论与顺铂方案相比,奥沙利铂方案具有较好的疗效与预后,不良反应相对较少,可被推荐和替代顺铂用于治疗进展期胃癌。未来需要大样本高质量的研究提供更多信息进一步探讨不同因素对两种方案的影响。 Abstract:Objective To evaluate the efficacy, prognosis and safety of oxaliplatin-based chemotherapy regimens versus cisplatin-based chemotherapy regimens as first-line treatment in patients with advanced gastric cancer. Methods A comprehensive literature search (up to December 31, 2014) was performed in CNKI, WangFang, Medline/PubMed, Embase/ Ovid and Cochrane Library. We also searched abstracts present at the conferences of ASCO and EMSO annual meeting and relevant clinical trials from clinical trial registry of North America (www.clinicaltrials.gov). Primary outcomes were overall survival (OS), overall response rate (ORR) and grade 3-4 toxicities. Results Thirty-two randomized controlled trial (RCTs) were included and 3994 patients were analyzed (1992 in oxaliplatin-based regimen and 2002 in cisplatin-based regimen). The meta-analysis showed that oxaliplatin-based regimen significantly prolong the OS (HR=0.90, 95%CI 0.81-0.99) and FPS (HR=0.89, 95% CI 0.81-0.98) compare to cisplatin-based regimen. Pooled estimate demonstrated that ORR of oxaliplatin-based regimen was superior (OR=1.33, 95%CI 1.17-1.51), similar trend was drawn in Asian patients subgroup and Caucasian patients subgroup. In grade III or IV toxicities, oxalipaltin-based regimen had a lower incidence of anemia (OR=0.49, 95%CI 0.38-0.62), leucopenia (OR=0.37, 95% CI 0.27-0.51), neutropenia (OR=0.49, 95% CI 0.40-0.59), but nausea/vomiting (OR=0.37, 95% CI 0.21-0.61) and peripheral neuropathy (OR=7.16, 95%CI 4.06-12.64) occurred commonly. All above had statistical significant. Conclusion Oxaliplatin-based regimen have better efficacy prognosis and fewer toxicities compared with cisplatin-based regimen, can be recommended and replace cisplatin for advanced gastric cancer. More large size and high quality RCTs need to confirm the results and provide more information in comparing the two regimens. -
Key words:
- oxaliplatin /
- cisplatin /
- advanced gastric cancer /
- chemotherapy /
- meta-analysis
-
表 1 严重不良反应效应量合并结果
不良反应 实验组 对照组 异质性 OR(95%CI) P P I2 恶心/呕吐 137/1582 237/1620 <0.00001 0.65 0.37(0.21, 0.61) <0.0001 口角炎 32/1177 28/1196 0.21 24% 1.03(0.51, 2.08) 0.92 腹泻 111/1192 75/1223 0.02 0.5 1.49(0.86, 2.59) 0.15 贫血 124/1159 225/1164 0.58 0 0.49(0.38, 0.62) <0.00001 白细胞减少 60/938 145/944 0.13 0.31 0.37(0.27, 0.51) <0.00001 中性粒细胞减少 270/1112 440/1130 0.11 0.39 0.49(0.40, 0.59) <0.00001 血小板减少 73/1131 80/1140 0.95 0 0.92(0.66, 1.28) 0.64 周围神经炎 89/1202 11/1223 0.84 0 7.16(4.06, 12.64) <0.00001 乏力 120/965 116/983 0.02 0.63 1.01(0.58, 1.74) 0.99 脱发 140/690 238/697 0.96 0 0.47(0.36, 0.60) <0.00001 -
[1] NCCN. The NCCN gastric cancer clinical practice guidelines inoncology(version 1.2015) EB/OL][2015-11-23]. http://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. [2] Kelland LR. Preclinical perspectives on Platinum resistance[J].Drugs, 2000, 59(Suppl 4): 1-8; discussion 37-8. [3] 余邦良, 马亚平, 刘全忠, 等. 铂类抗癌药物研究进展[J]. 海南大学学报:自然科学版, 2005, 23(1): 72-80. http://www.cnki.com.cn/Article/CJFDTOTAL-HNDK200501017.htm [4] Jadad AR, Moore RA, Carroll D, et al. Assessing the quality ofreports of randomized clinical trials: is blinding necessary? J].Control Clin Trials, 1996, 17(1): 1-12. doi: 10.1016/0197-2456(95)00134-4 [5] Begg CB, Mazumdar M. Operating characteristics of a rankcorrelation test for publication bias[J]. Biometrics, 1994, 50(4):1088-101. doi: 10.2307/2533446 [6] Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysisdetected by a simple, graphical test[J]. BMJ, 1997, 315(719):629-34. [7] Egger M, Mazumdar M. Operating characteristics of a rankcorrelation test for publication bias[J]. Biometrics, 1994, 50(4):1088-101. doi: 10.2307/2533446 [8] Taylor SJ, Tweedie RI. Practical estimates of the efect of publicationbias in meta-analysis[J]. Australas Epidemiol. 1998. 5: 14-7 https://www.researchgate.net/publication/2426834_Practical_Estimates_of_the_Effect_of_Publication_Bias_in_Meta-Analysis [9] Cunningham D, Starling N, Rao S, et al. Capecitabine andoxaliplatin for advanced esophagogastric cancer[J]. N Engl J Med,2008, 358(1): 36-46. doi: 10.1056/NEJMoa073149 [10] Al-Batran SE, Hartmann JT, Probst S, et al. Phase Ⅲ trial inmetastatic gastroesophageal adenocarcinoma with fluorouracil,leucovorin plus either oxaliplatin or cisplatin: a study of theArbeitsgemeinschaft Internistische Onkologie[J]. J Clin Oncol,2008, 26(9): 1435-42. doi: 10.1200/JCO.2007.13.9378 [11] Popov I, Radosevic-Jelic L, Jezdic S, et al. Biweekly oxaliplatin,fluorouracil and leucovorin versus cisplatin, fluorouracil andleucovorin in patients with advanced gastric cancer[J]. J BUON,2009, 13(4): 505-11. [12] Kim YS, Sym SJ, Park SH, et al. A randomized phase Ⅱ study ofweekly docetaxel/cisplatin versus weekly docetaxel/oxaliplatin asfirst-line therapy for patients with advanced gastric cancer[J].Cancer Chemother Pharmacol, 2014, 73(1): 163-9. doi: 10.1007/s00280-013-2334-3 [13] Yamada Y, Higuchi K, Nishikawa K, et al. Phase Ⅲ studycomparing oxaliplatin plus S-1 with cisplatin plus S-1 inchemotherapy-naive patients with advanced gastric cancer[J].Annals of Oncology, 2015, 26(1): 141-8. doi: 10.1093/annonc/mdu472 [14] Yamaguchi K, Hironaka S, Sugimoto N, et al. Randomized phase Ⅱstudy of S-1 plus oral leucovorin (SL) versus SL plus oxaliplatin(SOL) versus S-1 plus cisplatin (SP) in patients with advancedgastric cancer (AGC): Updated overall survival data.[J]. AnnOncol. 2014. 25(suppl 4): iv213-iv214 [15] 宋红蕾, 毕延智, 陈慧暖, 等. CS方案和SOX方案治疗晚期胃癌的临床观察[J]. 癌症进展, 2012, 10(5): 514-8. http://www.cnki.com.cn/Article/CJFDTOTAL-AZJZ201205024.htm [16] 韩守恒, 戴文香, 伍尤华. DOF和DCF方案治疗进展期胃癌的疗效分析[J]. 中南医学科学杂志, 2012, 40(6): 594-6, 599. http://www.cnki.com.cn/Article/CJFDTOTAL-HYYY201206017.htm [17] 张晓东, 陆俊国, 姚卫东, 等. DOF与DCF方案治疗进展期胃癌的临床对照观察[J]. 临床医药实践杂志, 2008(19): 957-60. http://www.cnki.com.cn/Article/CJFDTOTAL-SXLC200819004.htm [18] 陈文琪, 李小琴, 谢明慧. FOLFOX4方案及PLF方案治疗晚期胃癌的毒副作用评价[J]. 中外医疗, 2009(28): 76-7. http://www.cnki.com.cn/Article/CJFDTOTAL-HZZZ200928058.htm [19] 张雷. FOLFOX4与FLP方案化疗晚期胃癌的疗效对比研究[J]. 临床合理用药杂志, 2014, 7(11): 59-60. http://www.cnki.com.cn/Article/CJFDTOTAL-PLHY201411043.htm [20] 李淼, 赵成茂. FOLFOX 4与FLP方案在晚期胃癌化疗中的疗效分析[J]. 青海医药杂志, 2013, 43(3): 4-7. http://www.cnki.com.cn/Article/CJFDTOTAL-QHYZ201303003.htm [21] 余嘉文. FOLFOX4与FLP方案治疗晚期胃癌的疗效比较[J]. 中国现代药物应用, 2013, 7(8): 13-4. http://www.cnki.com.cn/Article/CJFDTOTAL-ZWYY201308008.htm [22] 唐东霞, 李春艳, 刘璇, 等. OLF方案与LFP方案治疗晚期胃癌的临床观察[J]. 肿瘤基础与临床, 2006, 19(2): 126-7. http://www.cnki.com.cn/Article/CJFDTOTAL-HLZL200602022.htm [23] 康用堂. PF方案与OLF方案用于治疗晚期胃癌的疗效比较[J]. 白求恩军医学院学报, 2012, 10(5): 394-5. http://www.cnki.com.cn/Article/CJFDTOTAL-BQEN201205031.htm [24] 郑向东, 鲍官明, 王俊, 等. 奥沙利铂或顺铂联合替吉奥一线治疗进展期胃癌近期疗效观察[J]. 医学信息, 2014, 27(7): 415-6. http://www.cnki.com.cn/Article/CJFDTOTAL-LNBJ201604025.htm [25] 李建群, 朱云根. 奥沙利铂联合5-氟尿嘧啶、亚叶酸钙化疗晚期胃癌的临床研究[J]. 临床医学工程, 2014, 21(11): 1447-8. http://www.cnki.com.cn/Article/CJFDTOTAL-YBQJ201411036.htm [26] 张泉, 高鹏, 方晓华, 等. 奥沙利铂联合5-氟尿嘧啶、亚叶酸钙治疗晚期胃癌的临床疗效观察[J]. 实用癌症杂志, 2006, 21(2): 180-2. http://www.cnki.com.cn/Article/CJFDTOTAL-SYAZ200602024.htm [27] 文世民, 潘荣强. 奥沙利铂联合5-氟尿嘧啶/亚叶酸钙治疗晚期胃癌临床疗效观察[J]. 川北医学院学报, 2004, 19(3): 11-2. http://www.cnki.com.cn/Article/CJFDTOTAL-NOTH200403005.htm [28] 潘荣强, 李光明, 别俊, 等. 奥沙利铂联合5-氟脲嘧啶/亚叶酸钙治疗晚期胃癌临床研究[J]. 四川肿瘤防治, 2005, 18(4): 227-8, 234. http://www.cnki.com.cn/Article/CJFDTOTAL-SCZF200504008.htm [29] 赵予军, 简国庆, 吕素兰. 奥沙利铂联合亚叶酸钙、5-氟尿嘧啶治疗晚期胃癌的临床研究[J]. 中国医药导报, 2007, 4(30): 22-3. http://www.cnki.com.cn/Article/CJFDTOTAL-NXYX200701021.htm [30] 李卫东, 吴迪, 邹青峰, 等. 奥沙利铂为主的EOF方案治疗晚期胃癌的临床研究[J]. 广西医学, 2009, 31(5): 657-9. http://www.cnki.com.cn/Article/CJFDTOTAL-GYYX200905028.htm [31] 曾皓明. 奥沙利铂治疗晚期胃癌的临床观察[J]. 中国厂矿医学, 2003,16(6): 14-5. [32] 张迦维, 缪建华, 赵帆. 多西他赛联合奥沙利铂治疗晚期转移性胃癌近期疗效观察[J]. 山东医药, 2008, 48(46): 76-7. http://www.cnki.com.cn/Article/CJFDTOTAL-SDYY200846056.htm [33] 唐溢聪, 卢秋霞, 林秀强, 等. 多西紫杉醇及氟尿嘧啶联合奥沙利铂与顺铂方案治疗晚期胃癌的随机对照研究[J]. 中外医学研究, 2013, 11(21): 7-9. http://www.cnki.com.cn/Article/CJFDTOTAL-YJZY201321007.htm [34] 顾术东, 刘艳, 刘凡, 等. 卡培他滨联合奥沙利铂或顺铂一线治疗晚期胃癌临床观察[J]. 中国医药导报, 2013, 10(26): 96-8, 101. http://www.cnki.com.cn/Article/CJFDTOTAL-YYCY201326034.htm [35] 王文义, 徐扬. 顺铂、奥沙利铂、紫杉醇三种联合化疗方案治疗晚期胃癌的临床观察[J]. 中国煤炭工业医学杂志, 2008, 11(11): 1726-7. http://www.cnki.com.cn/Article/CJFDTOTAL-ZMGY200811060.htm [36] 尹良伟, 马海英, 张春妹, 等. 替吉奥分别联合奥沙利铂、多西他赛与顺铂治疗进展期胃癌的比较研究[J]. 现代中西医结合杂志, 2012, 21(24): 2628-30. http://www.cnki.com.cn/Article/CJFDTOTAL-XDJH201224005.htm [37] 樊卫飞, 王峻, 孟丽娟, 等. 替吉奥联合奥沙利铂对比替吉奥联合顺铂方案一线治疗老年晚期胃癌的临床研究[J]. 临床肿瘤学杂志, 2013,18(1): 50-3. http://www.cnki.com.cn/Article/CJFDTOTAL-LCZL201301012.htm [38] 叶书成, 袁国珍, 郝万红, 等. 以奥沙利铂为主联合化疗晚期胃癌的研究[J]. 临床荟萃, 2005, 20(20): 1150-2. http://www.cnki.com.cn/Article/CJFDTOTAL-LCFC200520005.htm [39] 赵遴. 紫杉醇联合顺铂或奥沙利铂治疗晚期胃癌疗效比较[J]. 山东医药, 2005, 45(16): 60. http://www.cnki.com.cn/Article/CJFDTOTAL-SDYY200516054.htm [40] 许小娟, 钱晓萍, 刘宝瑞, 等. 奥沙利铂联合FL方案与FLP方案治疗进展期胃癌近期疗效观察[J]. 实用临床医药杂志, 2004, 8(5): 52-4. http://www.cnki.com.cn/Article/CJFDTOTAL-XYZL200405016.htm [41] Montagnani F, Turrisi G, Marinozzi C, et al. Effectiveness andsafety of oxaliplatin compared to cisplatin for advanced,unresectable gastric cancer: a systematic review and meta-analysis[J]. Gastric Cancer, 2011, 14(1): 50-5. doi: 10.1007/s10120-011-0007-7 [42] Kang YK, Kang WK, Shin DB, et al. Capecitabine/cisplatin versus5-fluorouracil/cisplatin as first-line therapy in patients withadvanced gastric cancer: a randomised phase Ⅲ noninferiority trial[J]. Ann Oncol, 2009, 20(4): 666-73. doi: 10.1093/annonc/mdn717 [43] Okines AF, Norman AR, Mccloud P, et al. Meta-analysis of theREAL-2 and ML17032 trials: evaluating capecitabine-basedcombination chemotherapy and infused 5-fluorouracil-basedcombination chemotherapy for the treatment of advancedoesophago-gastric cancer[J]. Ann Oncol, 2009, 20(9): 1529-34. doi: 10.1093/annonc/mdp047 -






 下载:
下载: