Study on synthesis of ACPP peptide and its effect on covalent modification of HPMApolymer
-
摘要:
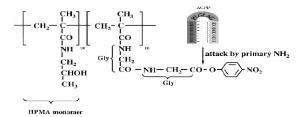
目的 合成一种可活化细胞穿膜肽ACPP,并初步探索其穿膜活性及其对高分子聚合物pHPMA的共价修饰作用。 方 法通过化学合成的方法合成可活化细胞穿膜肽ACPP,通过免疫荧光法检测细胞内mmp2蛋白的表达;通过荧光显微镜观察鉴定ACPP的穿膜活性。通过化学修饰法合成接合物pc-Ad.egfp 及ACPP-pc-Ad.egfp。通过荧光显微镜观察及动态光散射法初步鉴定接合物的合成,通过荧光显微镜观察鉴定接合物ACPP-pc-Ad.egfp 亚细胞分布。 结果成功合成了可活化细胞穿膜肽ACPP;ACPP具有肿瘤靶向性穿膜活性;通过ACPP修饰高分子聚合物pHPMA合成了ACPP-pc-Ad.egfp;经荧光显微镜观察证实ACPP-pc-Ad.egfp 具有较强的感染细胞的能力,并由ACPP介导大分子进行非内吞性跨膜运输。 结论成功合成了ACPP,ACPP 具有肿瘤靶向性穿膜活性,并成功修饰高分子聚合物pHPMA。 Abstract:ObjectiveTo achieve a novel macromolecules to traverse cell membranes by synthesis and characterization of a tumor- targeted ACPP and an ACPP modified- HPMA polymer conjugates. Methods Activable cell- penetrating peptide (ACPP) were prepared by free radical precipitation copolymerization method, the expression of mmp2 was measured by Immunofluorescence; the tumor-targeted cell-penetrating ability of ACPP was verified by inverted fluorescence microscopy; ACPP- pc- Ad.egfp conjugates was checked by inverted fluorescence microscopy and dynamic light scattering (DLS); Intracellular distribution of ACPP-pc-Ad. egfp was examined by inverted fluorescence microscopy. ResultsPeptides ACPP was synthesized as devised and ACPP targeted and penetrated the tumor cells. inverted fluorescence microscopy and DLS assays demonstrated the formation of ACPP-pc- Ad.egfp conjugates; ACPP-pc- Ad.egfp showed strong ability to translocate into tumor cells exhibited nonendocytotic cytoplasmic delivery. Conclusiontumo- targeted ACPP was synthesized and modified the HPMApolymer successfully. -
Key words:
- ACPP /
- tumor-targeted /
- pHPMA /
- covalent modification
-
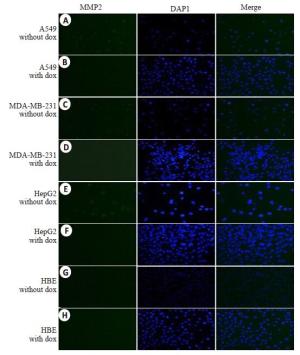
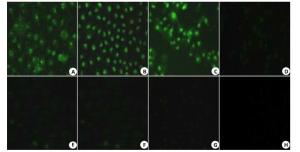
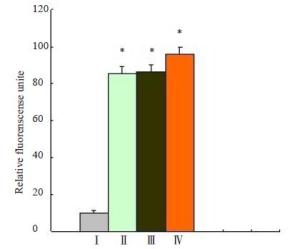
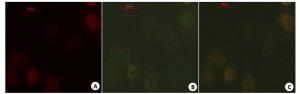
图 3 荧光检测可活化细胞穿膜肽穿透细胞膜能力
Figure 3. The internalization of ACPP was determined by fluorescenct staining labled by FITC was incubated with cells, A549 cells(A,E),MDA-MB-231cells(B,F),HepG2 cells(C,G) and HBE cells(D,G) for 4 h, respectively.(E-H) FITC fluorescence of ACPP in A549、MDA-MB-231、HepG2 and HBE treated with Doxycycline (Original magnification: ×200).
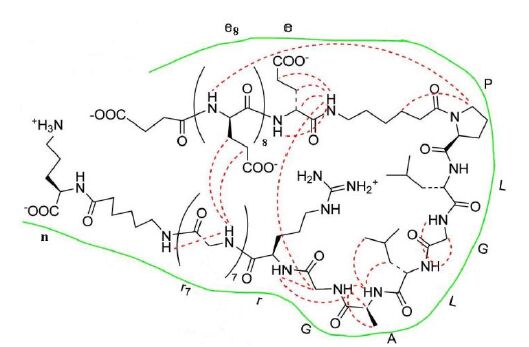
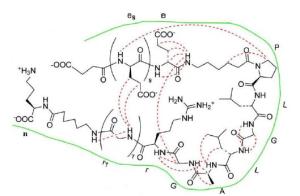
图 8 ACPPs与pc-Ad.egfp的细胞内共定位检测
Figure 8. Intracellular distribution of of [(FITC) ACPP-pc-Ad.egfp(PI)] by inverted fluorescence microscopy.after 4 h incubation with A549 cells at 37 ℃ showed cytoplasmic uptake of ACPP marker FITC(green); (B) showed cytoplasmic uptake of Ad.egfp marker PI FITC(red); superimposition of the two dyes (green-red staining) confirms ACPP mediated transportation of pc-Ad.egfp to the cytoplasm.
-
[1] Kopecek J. Reactive copolymers of N-(2-hydroxypropyl)methacrylamide with N-methacryloylated derivatives of L-leucine and L phenylalanine [J]. Macromol Chem, 1977, 178(8): 2169-83. doi: 10.1002/macp.1977.021780804 [2] Ringsdorf H. Structure and properties of pharmacologically active polymers[J]. Polym Sci Polym Symp, 1975, 51(1): 135-53. http://cn.bing.com/academic/profile?id=88f6490f88e1d3dbf14b175d0b53fd03&encoded=0&v=paper_preview&mkt=zh-cn [3] Fisher KD, Stallwood Y, Green NK, et al. Polymer- coated adenovirus permits efficient retargeting and evades neutralising antibodies[J]. Gene Ther, 2001, 8(5): 341-8. doi: 10.1038/sj.gt.3301389 [4] Rihova B. Biocompatibility and immunocompatibility of water-soluble polymersbased on HPMA[J]. Composites Part B- Engineering, 2007, 38: 386-97. doi: 10.1016/j.compositesb.2006.07.007 [5] Nori A, Jensen KD, Tijerina M, et al. Tat- conjugated synthetic macromolecules facilitate cytoplasmic drug delivery to human ovarian carcinoma cells[J]. Bioconjug Chem, 2003, 14(1): 44-50. doi: 10.1021/bc0255900 [6] Blankenberg FG, Mandl S, Cao YA, et al. Tumor imaging using a standardized radiolabeled adapter protein docked to vascular endothelial growth factor[J]. J Nucl Med, 2004, 45(8): 1373-80. http://cn.bing.com/academic/profile?id=cfcbe8ffe07445ecb27a57cc89565131&encoded=0&v=paper_preview&mkt=zh-cn [7] Shi NQ, Gao W, Xiang B, et al. Enhancing cellular uptake of activable cell- penetrating peptide- doxorubicin conjugate by enzymatic cleavage[J]. Int J Nanomedicine, 2012, 7: 1613-21. http://cn.bing.com/academic/profile?id=b8e8262558ea6280042f4838414dbcf1&encoded=0&v=paper_preview&mkt=zh-cn [8] Olson ES, Jiang T, Aguilera TA, et al. Activatable cell penetrating peptides linked to nanoparticles as dual probes for in vivo fluorescence and Mr imaging of proteases[J]. Proc Natl Acad Sci USA, 2010, 107(9): 4311-6. doi: 10.1073/pnas.0910283107 [9] Sawant RR, Patel NR, Torchilin VP. Therapeutic delivery using cellpenetrating peptides[J]. European Journal of Nanomedicine, 2013, 5 (3): 111-67. http://cn.bing.com/academic/profile?id=9c1a8af5bd8aefc11808c16f2fe56b93&encoded=0&v=paper_preview&mkt=zh-cn [10] Jiang T, Olson ES, Nguyen QT, et al. Tumor imaging by means of proteolytic activation of cell- penetrating peptides[J]. Proc Natl Acad Sci U S A, 2004, 101(51): 17867-72. doi: 10.1073/pnas.0408191101 [11] Mccawley LJ, Matrisian LM. Matrix metalloproteinases: multifunctional contributors to tumor progression[J]. Mol Med Today, 2000, 6 (4): 149-56. doi: 10.1016/S1357-4310(00)01686-5 [12] Takeuchi H, Bilchik A, Saha S, et al. c-MET expression level in primary colon Cancer: a predictor of tumor invasion and lymph node metastases[J]. Clin Cancer Res, 2003, 9(4): 1480-8. http://cn.bing.com/academic/profile?id=69e1d9371c5c8327ce498d583ee0dff1&encoded=0&v=paper_preview&mkt=zh-cn [13] Christensen JG, Burrows J, Salgia R. c-Met as a target for human Cancer and characterization of inhibitors for therapeutic intervention [J]. Cancer Lett, 2005, 225(1): 1-26. doi: 10.1016/j.canlet.2004.09.044 [14] Ringsdorf H. Structure and properties of pharmacologically active polymers[J]. Polym Sci Polym Symp, 1975, 51(1): 135-53. http://cn.bing.com/academic/profile?id=88f6490f88e1d3dbf14b175d0b53fd03&encoded=0&v=paper_preview&mkt=zh-cn [15] Nori A, Jensen KD, Tijerina M, et al. Subcellular trafficking of HPMA copolymer-Tat conjugates in human ovarian carcinoma cells [J]. J Control Release, 2003, 91(1/2): 53-9. http://cn.bing.com/academic/profile?id=831a80dcb8b2a234d53562beab8a5d44&encoded=0&v=paper_preview&mkt=zh-cn -






 下载:
下载: