Observation of the clinical efficacy and safety of autologous bone preserved using bone tissue bank technology in cranioplasty
-
摘要:
目的 观察骨组织库技术保存自体颅骨在颅骨缺损修补术中的临床疗效及安全性。 方法 收集南方医科大学顺德医院神经外科自2019年9月~2022年7月行幕上颅骨缺损修补手术的病例为研究对象,依据修补材料不同分为自体颅骨组(经骨组织库保存的自体颅骨修补手术,n=30)和三维钛网组(经3D数字塑形钛网的颅骨修补手术,n=30),两组患者一般资料具有可比性(P > 0.05)。对比两组术后1月内感染率、硬膜外积液/积血发生率、手术切口愈合不良发生率、癫痫发生率、脑内出血发生率等直接手术并发症指标,并计算“自体颅骨组”基于头颅CT影像的骨吸收率。 结果 两组患者均取得术后至少6月的随访,自体颅骨组与三维钛网组术后1月内手术相关感染率(0例vs 0例)、硬膜外积液/积血发生率(3例vs7例)、手术切口愈合不良发生率(0例vs 0例)、术后癫痫发生率(0例vs 0例)、脑出血发生率(0例vs1例)的差异均无统计学意义(P > 0.05)。术后6月自体颅骨组发生骨吸收4例(13.3%),其中1例严重骨吸收患者需要人工材料再次修补。两组间总并发症发生率的差异无统计学意义[7例(23%)vs 8例(35%),P > 0.05]。 结论 经骨组织库技术保存自体颅骨是颅骨修补术安全有效的方式之一,但仍存在骨吸收风险,临床上可根据患者具体情况作出选择。 Abstract:Objective To observe the clinical efficacy and safety of autogenous skull preserved by bone tissue bank technology in cranioplasty. Methods Hospitalized patients with supratentorial skull defect repair surgery in the Department of Neurosurgery, Shunde Hospital of Southern Medical University from September 2019 to July 2022 were enrolled. According to different repair materials, the patients were divided into the autologous skull repair group (n=30, autologous skull repair surgery preserved by bone tissue bank) and the 3D titanium mesh group (n=30, skull repair surgery with 3D digital shaped titanium mesh). The general data of the two groups was comparable (P > 0.05). Indicators of complications directly related to surgery including infection rate within 1 month after surgery, incidence of epidural effusion/hematoma, incidence of poor healing of surgical incision, epilepsy incidence and incidence of intracerebral hemorrhage were compared between the two groups and bone absorptivity based on skull CT images was measured in the autologous skull repair group. Results Both groups were followed up for at least 6 months after surgery. There was no significant difference in the infection rate within 1 month after surgery (0 case vs 0 case), incidence of epidural effusion/hematoma (3 cases vs 7 cases), incidence of poor healing of surgical incision (0 case vs 0 case), epilepsy incidence (0 case vs 0 case) and incidence of intracerebral hemorrhage (0 case vs 1 case) between the autologous skull group and 3D titanium mesh group (P > 0.05). Bone resorption occurred in 4 patients (13.3%) in the autogenous skull group 6 months after operation, and one of the patients with severe bone resorption required re-repair with artificial materials. There was no significant difference in the total complication rate between the autologous skull group and 3D titanium mesh group [7 cases (23%) vs 8 cases (35%), P > 0.05]. Conclusion The technique of preserving autogenous skull through bone tissue bank is one of the safe and effective methods in cranioplasty while remaining a risk of bone resorption, it can be an alternative depending on the specific situation of patients clinically. -
Key words:
- skull defect /
- cranioplasty /
- autologous bone
-
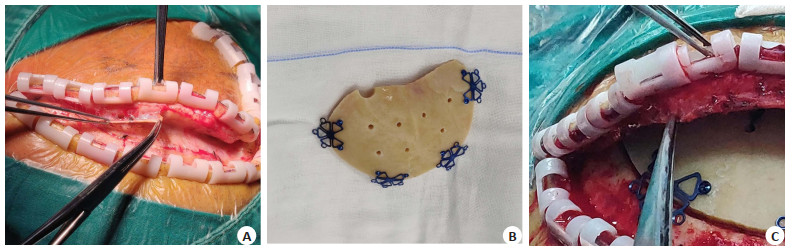
图 1 右侧额颞顶部颅骨缺损行自体颅骨瓣修补手术病例
Figure 1. A case of autologous skull flap repair for right frontotemporal top skull defect. A: Separation along the potential space between dura and periosteal; B: Autogenous skull flap with connecting plate (several holes drilled into the skull plate); C: The autologous cranial flap was fixed and the flap was retained with periosteal suture.
图 2 双侧额颞顶部行自体颅骨瓣修补手术后发生骨瓣吸收病例
Figure 2. A case of bone resorption occurred after bilateral frontotemporal autogenous cranial flap repair. A: Head CT scan 9 months after surgery showed severe absorption of bilateral autogenous skull flap, with the right side being obvious; B: Three-dimensional CT reconstruction showed obvious absorption of the right autogenous skull flap and scattered small skull fragments on the bone window; C: Three-dimensional titanium mesh was used to repair the right skull defect.
图 3 自体颅骨修补术后随诊病例
Figure 3. A follow-up case after autologous cranioplasty. A: CT scan of the head 1 week after surgery showed good alignment of the autogenous skull, and the autogenous bone flap and skull suture were obvious (△); B: Three-dimensional CT reconstruction showed obvious suture between autogenous bone flap and skull 1 week after surgery (*); C: Head CT showed that the gap between autogenous bone flap and skull was blurred, and partial bone healing 7 months after surgery(☆); D: Three-dimensional CT reconstruction showed local narrowing of the space between autogenous bone flap and skull, and partial bone healing 7 months after surgery (☆).
表 1 自体颅骨组与三维钛网组患者一般资料比较
Table 1. Comparison of general data between the two groups (n=30)
Index Autologous skull group 3D titanium mesh group t/χ2 P Gender (n, Male/Female) 24/6 24/6 0 > 0.999 Age[years, M(P25, P75)] 49.5(29.8, 56.0) 47(38.0, 56.0) -0.074 0.941 Single/Bilateral skull defect (n) 29/1 29/1 0 > 0.999 Skull decompression area [cm2, M(P25, P75)] 173.0(146.3, 183.5) 176.5(162.5, 185.8) -0.925 0.935 表 2 自体颅骨组与三维钛网组手术相关并发症指标比较
Table 2. Comparison of postoperative complications between two groups of patients [n(%), n=30]
Index Autologous skull group 3D titanium mesh group t/χ2 P Infection within 1 month after surgery 0(0) 0(0) - - Poor healing of surgical incision 0(0) 0(0) - - Epilepsy 0(0) 0(0) - - Epidural effusion/hematoma 3(10) 7(23) 1.920 0.166 Intracerebral hemorrhage 0(0) 1(3.33) - - Bone resorption 4(13.3) 0(0) - - Mild bone resorption 3(10) 0(0) - - Severe bone resorption 1(3.33) 0(0) - - Dead 0(0) 0(0) - - Total complication 7(23) 8(35) 0.089 0.766 -
[1] 中华神经外科学会神经创伤专业组, 中华创伤学会神经损伤专业组, 中国神经外科医师协会神经创伤专家委员会. 创伤性颅骨缺损成形术中国专家共识[J]. 中华神经外科杂志, 2016, 32(8): 767-70. doi: 10.3760/cma.j.issn.1001-2346.2016.08.002 [2] da Costa Benalia VH, Pedrozo CAG, Kormanski MK, et al. Spontaneous bone flap resorption following cranioplasty using autologous bone[J]. J Craniofacial Surg, 2020, 32(1): 293-6. [3] Yu QS, Chen L, Qiu ZY, et al. Skull repair materials applied in cranioplasty: history and progress[J]. Transl Neurosci Clin, 2017, 3(1): 48-57. doi: 10.18679/CN11-6030_R.2017.007 [4] Piitulainen JM, Kauko T, Aitasalo KMJ, et al. Outcomes of cranioplasty with synthetic materials and autologous bone grafts[J]. World Neurosurg, 2015, 83(5): 708-14. doi: 10.1016/j.wneu.2015.01.014 [5] Klinger DR, Madden C, Beshay J, et al. Autologous and acrylic cranioplasty: a review of 10 years and 258 cases[J]. World Neurosurg, 2014, 82(3/4): e525-30. [6] Li A, Azad TD, Veeravagu A, et al. Cranioplasty complications and costs: a national population-level analysis using the MarketScan longitudinal database[J]. World Neurosurg, 2017, 102: 209-20. doi: 10.1016/j.wneu.2017.03.022 [7] Capitelli-McMahon H, Kahlar N, Rahman S. 402A systematic review and meta-analysis of autologous vs titanium cranioplasty implants[J]. Br J Surg, 2021, 108(Supplement2): 133. [8] 范英俊, 邱治春, 苗树船, 等. 三维塑形钛网与超低温保存自体颅骨在早期颅骨修补中的临床效果观察[J]. 实用医院临床杂志, 2019, 16(5): 170-2. https://www.cnki.com.cn/Article/CJFDTOTAL-YYLC201905051.htm [9] Hassan H, Ali A, Abdalla A. Autogenous Bone Graft versus Artificial Substitutes in Cranioplasty[J]. Open J Modern Neurosurg, 2019(3): 338-55. [10] Yeap MC, Tu PH, Liu ZH, et al. Long-term complications of cranioplasty using stored autologous bone graft, three-dimensional polymethyl methacrylate, or titanium mesh after decompressive craniectomy: a single-center experience after 596 procedures[J]. World Neurosurg, 2019, 128: e841-50. doi: 10.1016/j.wneu.2019.05.005 [11] Malcolm JG, Zayan M, Rindler RS, et al. Autologous cranioplasty is associated with increased reoperation rate: a systematic review and meta-analysis[J]. World Neurosurg, 2018, 116: 60-8. doi: 10.1016/j.wneu.2018.05.009 [12] Ernst G, Qeadan F, Carlson AP. Subcutaneous bone flap storage after emergency craniectomy: cost-effectiveness and rate of resorption[J]. J Neurosurg, 2018, 129(6): 1604-10. doi: 10.3171/2017.6.JNS17943 [13] Bose S, Roy M, Bandyopadhyay A. Recent advances in bone tissue engineering scaffolds[J]. Trends Biotechnol, 2012, 30(10): 546-54. doi: 10.1016/j.tibtech.2012.07.005 [14] Kim JH, Kim JH, Kwon TH, et al. Aseptic bone flap resorption after cranioplasty with autologous bone: incidence, risk factors, and clinical implications[J]. World Neurosurg, 2018, 115: e111-8. doi: 10.1016/j.wneu.2018.03.197 [15] Yang J, Sun T, Yuan Y, et al. Risk factors for bone flap resorption after autologous bone cranioplasty: Protocol for a systematic review and meta-analysis[J]. Medicine (Baltimore), 2020, 99(28): e21035. doi: 10.1097/MD.0000000000021035 [16] 李宝庆, 李宝东, 张凯, 等. 自体颅骨聚维酮碘浸泡冷冻保存定期颅骨修补[J]. 中国煤炭工业医学杂志, 2011, 14(2): 245-6. https://www.cnki.com.cn/Article/CJFDTOTAL-ZMGY201102053.htm [17] 王文福, 刘栋, 白易欣, 等. 自体颅骨不同保存方法的超微结构改变与临床研究[J]. 潍坊医学院学报, 2010, 32(6): 479-80. [18] 孙鹏, 孟庆海, 杨新生, 等. 自体颅骨深低温保存再植的应用研究[J]. 中华神经外科杂志, 2009, 25(5): 458-60. doi: 10.3760/cma.j.issn.1001-2346.2009.05.032 [19] Frassanito P, De Bonis P, Mattogno PP, et al. The fate of a macro-porous hydroxyapatite cranioplasty four years after implantation: Macroscopical and microscopical findings in a case of recurrent atypical meningioma[J]. Clin Neurol Neurosurg, 2013, 115(8): 1496-8. doi: 10.1016/j.clineuro.2012.11.032 [20] Pasick CM, Margetis K, Santiago GF, et al. Adult cranioplasty[J]. J Craniofacial Surg, 2019, 30(7): 2138-43. doi: 10.1097/SCS.0000000000005659 [21] 于明琨, 王永谦. 自体骨移植修补颅骨缺损的材料与方法[J]. 中国临床康复, 2006, 10(41): 126-9. https://www.cnki.com.cn/Article/CJFDTOTAL-XDKF200641041.htm [22] Kumar NG, Sudeep S, Balwan R. Cranioplasty of hemispherical defects using calcium phosphate cements along with titanium mesh: our experience[J]. J Maxillofac Oral Surg, 2015, 14(4): 920-4. doi: 10.1007/s12663-015-0776-3 [23] Mirabet V, García D, Yagüe N, et al. The storage of skull bone flaps for autologous cranioplasty: literature review[J]. Cell Tissue Bank, 2021, 22(3): 355-67. doi: 10.1007/s10561-020-09897-2 [24] Corliss B, Gooldy T, Vaziri S, et al. Complications after in vivo and ex vivo autologous bone flap storage for cranioplasty: a com-parative analysis of the literature[J]. World Neurosurg, 2016, 96: 510-5. doi: 10.1016/j.wneu.2016.09.025 [25] Honeybul S, Morrison DA, Ho KM, et al. A randomised controlled trial comparing autologous cranioplasty with custom-made titanium cranioplasty: long-term follow-up[J]. Acta Neurochir, 2018, 160(5): 885-91. doi: 10.1007/s00701-018-3514-z [26] Falko S, Pedro D, Jan W, et al. Cranioplasty after decompressive craniectomy: is there a rationale for an initial artificial bone-substitute implant? A single-center experience after 631 procedures[J]. J Neurosurg, 2016, 124(3): 710-5. doi: 10.3171/2015.4.JNS159 [27] De Cola MD, Corallo F, Pria D, et al. Timing for cranioplasty to improve neurological outcome: a systematic review[J]. Brain Behav, 2018, 8(11): e01106. doi: 10.1002/brb3.1106 [28] Pasick CM, Margetis K, Santiago GF, et al. Adult cranioplasty[J]. J Craniofac Surg, 2019, 30(7): 2138-43. doi: 10.1097/SCS.0000000000005659 [29] 朱锡德, 高振娟, 郑国栋, 等. 自体颅骨修补创伤性颅骨缺损术后并发症的分析[J]. 中华神经外科杂志, 2020, 36(12): 1247-51. doi: 10.3760/cma.j.cn112050-20200126-00033 [30] Honeybul S, Morrison DA, Ho KM, et al. A randomised controlled trial comparing autologous cranioplasty with custom-made titanium cranioplasty: long-term follow-up[J]. Acta Neurochir, 2018, 160(5): 885-91. doi: 10.1007/s00701-018-3514-z [31] Korhonen TK, Salokorpi N, Ohtonen P, et al. Classification of bone flap resorption after cranioplasty: a proposal for a computed tomography-based scoring system[J]. Acta Neurochir, 2019, 161(3): 473-81. [32] Korhonen TK, Posti JP, Niinimäki J, et al. Two-center validation of the Oulu resorption score for bone flap resorption after autologous cranioplasty[J]. Clin Neurol Neurosurg, 2022, 212: 107083. -







 下载:
下载: