Application value of the volumetric method in assessing the effectiveness of nonoperative treatment for patients with non-small cell lung cancer
-
摘要:
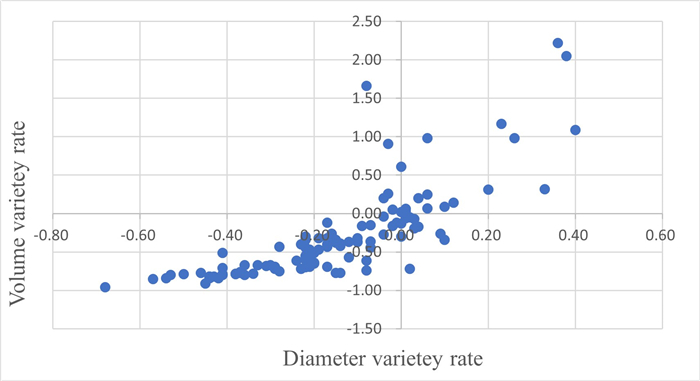
目的 分析非手术治疗后,在评估晚期非小细胞肺癌患者时,肿瘤体积与肿瘤最大直径[基于实体肿瘤疗效评价标准(RECIST)1.1版]是否相互关联,并评估晚期非小细胞肺癌肿瘤体积变化在疗效评估中的应用价值。 方法 回顾性分析108例经穿刺活检病理证实为非小细胞肺癌患者的CT影像学资料并进行随访,使用体积测量软件对治疗前后病灶体积进行测量,记录体积测量值与病灶最大直径,分别通过体积方法与RECIST 1.1版对疗效进行评估,并根据不同标准,将完全缓解和部分缓解归为缓解组,将疾病稳定与疾病进展归为未缓解组。采用Spearman相关系数评估RECIST最大直径与体积治疗前后变化率之间的相关性,采用Kaplan-Meier曲线评估不同评价标准中患者生存率之间的相关性。 结果 通过基线扫描和第1次随访扫描,计算了体积与直径相对于基线测量的变化率,评估结果显示两者具有中度相关性(r=0.881,P < 0.01)。在第1次随访中通过体积标准评估缓解组与未缓解组的K-M生存曲线的差异有统计学意义(P=0.002),根据RECIST标准进行评估时,两组差异无统计学意义(P=0.828)。 结论 体积测量对非小细胞肺癌治疗后疗效评估具有较好的应用价值。 -
关键词:
- 肺癌 /
- 非小细胞肺癌 /
- 体积测量 /
- RECIST疗效评估
Abstract:Objective To analyze whether tumor volume and tumor maximum diameter [based on the criteria for evaluating the efficacy of solid tumors (RECIST) version 1.1] are correlated with each other in evaluating patients with advanced non-small cell lung cancer (NSCLC) after non-surgical treatment and to assess the value of the application of the changes in tumor volume of advanced NSCLC in the evaluation of efficacy. Methods We retrospectively analyzed the CT imaging data of 108 patients with pathologically confirmed NSCLC by puncture biopsy and followed them up, measured the volume of the lesions before and after treatment using volumetric software, recorded the volume measurement values with the maximum diameter of the lesions, and then evaluated the therapeutic efficacy by the volumetric method and RECIST version 1.1, respectively. The complete response and partial response were categorized into the partial responders, and stable disease and progressive disease were categorized into the non-responders according to different criteria. Spearman correlation coefficients were used to assess the correlation between the maximum diameter of RECIST and the rate of change in volume before and after treatment, and Kaplan-Meier curves were used to evaluate the association between the survival of patients in different evaluation criteria. Results The rate of change of volume versus diameter relative to baseline measurements was calculated by baseline and first follow-up scans, and the assessment showed a moderate correlation (r=0.881, P < 0.01). The difference between the Kaplan-Meier curves of the responders and non-responders as assessed by the volume criterion at first follow up was statistically significant (P=0.002), and the difference between the two groups was not statistically significant when assessed according to the RECIST criterion (P=0.828). Conclusion Volumetric measurements have good application value for the assessment of the efficacy of NSCLC after treatment. -
图 2 非小细胞肺癌患者治疗前后病灶大小变化图
Figure 2. Changes in lesion size before and after treatment in non-small cell lung cancer patients. A-B: Female, 74 years old, with a maximum diameter of 80 mm and volume of 292 613.00 mm3 before treatment (A) and 61 mm and volume of 70 553.70 mm3 after treatment (B). Volume criteria were assessed as partial response and RECIST criteria as stable disease. C-D: Female, 49 years old with a maximum diameter of 29 mm and volume of 12 737.00 mm3 before treatment (C) and 26 mm and volume of 7991.80 mm3 after treatment (D). Volume criteria were assessed as partial response and RECIST criteria as stable disease.
表 1 治疗反应评估标准的比较
Table 1. Comparison of criteria for evaluation of treatment response
Criteria Complete response Partial response Progressive disease Stable disease RECIST1.1(2D) Disappearance of the lesions 30% decrease in diameter 20% increase in diameter Neither partial response nor progressive disease criteria met Volume(3D) Disappearance of the lesions 30% decrease in volume 20% increase in volume Neither partial response nor progressive disease criteria met RECIST: Response evaluation criteria in solid tumors. 表 2 患者基线特征
Table 2. Characteristics of the study patients
Variables n(%) Age(years) <60 29(26.85) ≥60 79(73.15) Gender Male 76(70.37) Female 32(29.63) Histology Squamous cell carcinoma 50(46.30) Adenocarcinoma 53(49.07) Other NSCLC 5(5.63) Smoking Yes 69(63.89) No 39(36.11) Drinking Yes 34(31.48) No 74(68.52) Chronic diseases Yes 73(67.59) No 35(32.41) RECIST1.1 Partial responder 27(25.00) Non-responder 81(75.00) Volume Partial responder 72(66.67) Non-responder 36(33.33) 表 3 RECIST1.1标准及体积标准疗效评估结果
Table 3. RECIST1.1 and Volume assessment results (n)
RECIST1.1 Volume Total PR SD PD PR 27 0 0 27 SD 45 21 8 74 PD 0 0 7 7 Total 72 21 15 108 表 4 患者基线特征的单因素及多因素COX回归分析
Table 4. Uni-and multivariable cox analysis of patient baseline characteristics
Characteristic Univariable survival analysis Multivariable survival analysis HR(95% CI) P HR(95% CI) P Gender 1.88(1.13-3.15) 0.016 1.27(0.56-2.86) 0.570 Age 1.82(1.06-3.10) 0.029 1.61(0.93-2.79) 0.086 Smoking 1.87(1.15-3.03) 0.012 1.25(0.57-2.76) 0.582 Histology 0.77(0.55-1.07) 0.120 Drinking 1.06(0.66-1.69) 0.819 Chronic diseases 0.99(0.62-1.56) 0.952 RECIST1.1 0.95(0.57-1.57) 0.828 Volume 0.48(0.30-0.76) 0.002 0.56(0.34-0.93) 0.024 -
[1] Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-49. doi: 10.3322/caac.21660 [2] 冯骁, 仓顺东. 中国肺癌筛查标准(T/CPMA 013-2020)解读[J]. 中华实用诊断与治疗杂志, 2021, 35(3): 217-9. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZD202103001.htm [3] Chen PX, Liu YH, Wen YK, et al. Non-small cell lung cancer in China[J]. Cancer Commun, 2022, 42(10): 937-70. doi: 10.1002/cac2.12359 [4] 李慧, 李伟, 王淑英. 吉非替尼联合化疗对晚期非小细胞肺癌的疗效及安全性分析[J]. 国际检验医学杂志, 2020, 41(24): 2957-61. doi: 10.3969/j.issn.1673-4130.2020.24.004 [5] 杨曦月, 杜小波. 晚期非小细胞肺癌治疗方案的研究进展及三线治疗探索[J]. 保健医学研究与实践, 2021, 18(4): 137-43. https://www.cnki.com.cn/Article/CJFDTOTAL-GXBJ202104030.htm [6] Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1)[J]. Eur J Cancer, 2009, 45(2): 228-47. doi: 10.1016/j.ejca.2008.10.026 [7] Litière S, Collette S, de Vries EGE, et al. RECIST-learning from the past to build the future[J]. Nat Rev Clin Oncol, 2017, 14(3): 187-92. doi: 10.1038/nrclinonc.2016.195 [8] Nishino M, Guo MY, Jackman DM, et al. CT tumor volume measurement in advanced non-small-cell lung cancer[J]. Acad Radiol, 2011, 18(1): 54-62. doi: 10.1016/j.acra.2010.08.021 [9] D.S. Gierada, C.E. Rydzak, M. Zei, 等. CT半自动体积测量对于实性肺结节观察者间的一致性评估的改进[J]. 国际医学放射学杂志, 2021, 44(1): 113-4. https://www.cnki.com.cn/Article/CJFDTOTAL-GWLC202101053.htm [10] Schiavon G, Ruggiero A, Schöffski P, et al. Tumor volume as an alternative response measurement for imatinib treated GIST patients[J]. PLoS One, 2012, 7(11): e48372. doi: 10.1371/journal.pone.0048372 [11] Siegel RL, Miller KD, Wagle NS, et al. Cancer statistics, 2023[J]. CA Cancer J Clin, 2023, 73(1): 17-48. doi: 10.3322/caac.21763 [12] 刘建伟, 郑爱民. 早期非小细胞肺癌局部治疗研究进展[J]. 介入放射学杂志, 2023, 32(5): 508-11. https://www.cnki.com.cn/Article/CJFDTOTAL-JRFS202305013.htm [13] 梁迪, 师金, 李道娟, 等. 人群肺癌筛查的研究进展[J]. 中国肿瘤, 2023, 32(1): 46-53. https://www.cnki.com.cn/Article/CJFDTOTAL-ZHLU202301007.htm [14] 温少迪, 沈波. 非小细胞肺癌免疫治疗作用机制及临床研究现状[J]. 南京医科大学学报: 自然科学版, 2020, 40(11): 1739-46. doi: 10.7655/NYDXBNS20201130 [15] 任胜祥, 邱天羽. 肺癌精准治疗的发展与展望[J]. 肿瘤防治研究, 2023, 50(5): 442-51. https://www.cnki.com.cn/Article/CJFDTOTAL-ZLFY202305002.htm [16] Fernandes AT, Mitra N, Xanthopoulos E, et al. The impact of extent and location of mediastinal lymph node involvement on survival in stage Ⅲ non-small cell lung cancer patients treated with definitive radiotherapy[J]. Int J Radiat Oncol, 2012, 83(1): 340-7. [17] Nishino M, Cryer SK, Okajima Y, et al. Tumoral cavitation in patients with non-small-cell lung cancer treated with antiangiogenic therapy using bevacizumab[J]. Cancer Imaging, 2012, 12(1): 225-35. [18] 黄玉宝, 苏晶, 彭晶玉, 等. 贝伐珠单抗治疗非鳞非小细胞肺癌后空洞的临床评价[J]. 中国合理用药探索, 2023, 20(4): 68-74. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYYS202304010.htm [19] Wolchok JD, Hoos A, O'Day S, et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria[J]. Clin Cancer Res, 2009, 15(23): 7412-20. [20] 许艺兰, 张晓鹏, 齐丽萍, 等. 比较单径测量法与体积测量法评价晚期非小细胞肺癌化疗的疗效[J]. 中国医学影像技术, 2015, 31(3): 401-4. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYXX201503033.htm [21] 张惠玲, 张宏伟, 何元华. 基于CT测量的肿瘤体积对非小细胞肺癌患者新辅助化疗疗效的评估价值[J]. 分子影像学杂志, 2023, 46(3): 527-31. doi: 10.12122/j.issn.1674-4500.2023.03.26 -







 下载:
下载: