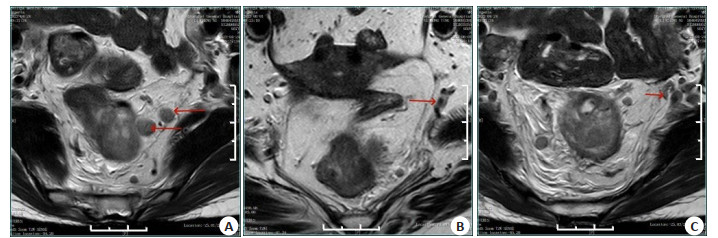
Clinical value of dynamic enhanced MR Combined with high resolution MR for lymph node metastasis of rectal adenocarcinoma
-
摘要:
目的 探讨动态增强MR联合高分辨率MR对直肠腺癌淋巴结转移的临床价值。 方法 纳入上海交通大学附属第一人民医院2018年1月~2022年12月收治的196例经病理证实为直肠癌患者的临床资料,所有患者均在该院完成高分辨率MRI和动态增强扫描MRI检查。根据术后病理将患者分为无淋巴结转移组(n=124)和淋巴结转移组(n=72),分析两组间临床特征、动态增强MR定量参数和高分辨率MR影像表现的差异,采用Logistic回归分析筛选预测直肠癌淋巴结转移的因素,采用ROC曲线计算不同参数预测淋巴结转移的效能。 结果 单因素Logistic回归分析显示,直肠腺癌淋巴结转移的危险因素是年龄 < 59.5岁(OR:0.329)、N分期MR影像学表现(mrN stage)为N1-N2期(OR:6.857)、癌胚抗原异常(OR:2.742)、T分期MR影像学表现(mrT stage)为T3期(OR:2.959)、mrMRF受累阳性(OR:5.577)、动态增强扫描MRI定量参数Ktrans < 0.298 min-1(OR:0.210)及mr EMVI阳性(OR:2.261)。多因素Logistic回归分析显示mrN stage阳性和Ktrans是直肠腺癌淋巴结转移的独立预测因素;mrN stage+Ktrans预测直肠腺癌淋巴结转移的AUC(0.788)高于单独的mrN stage(0.713)及Ktrans(0.650)。 结论 动态增强MR定量参数Ktrans联合mrN stage能够提高直肠腺癌淋巴结转移患者的预测能力。 -
关键词:
- 动态对比增强磁共振成像 /
- 高分辨率磁共振成像 /
- 直肠腺癌 /
- 淋巴结转移
Abstract:Objective To investigate the clinical value of dynamic contrast-enhanced MR combined with high resolution MR in lymph node metastasis of rectal adenocarcinoma. Methods Clinical data of 262 patients with pathologically confirmed rectal cancer admitted to the First People's Hospital Affiliated to Shanghai Jiao Tong University from January 2018 to December 2022 were collected. All patients underwent high resolution MR and contrast-enhanced MR in the hospital. According to postoperative pathology, the patients were divided into two groups: no lymph node metastasis group (n=124) and lymph node metastasis group (n=72). The differences in clinical features, quantitative parameters of dynamic contrast-enhanced MR and high resolution MR between the two groups were analyzed, and the factors predicting lymph node metastasis of rectal cancer were screened by Logistic regression analysis. ROC curve was used to calculate the efficacy of different parameters in predicting lymph node metastasis. Results Univariate Logistic regression analysis showed that the risk factors for lymph node metastasis of rectal adenocarcinoma were age < 59.5 years old (OR: 0.329), mrN stage N1-N2 (OR: 6.857)、abnormal carcinoembryonic antigen (OR: 2.742), mrT stage T3 (OR: 2.959), mrMRF positive (OR: 5.577), Quantitative parameter Ktrans of dynamic contrast-enhanced MR < 0.298 min-1 (OR: 0.210) and mr EMVI positive (OR: 2.261). Multivariate Logistic regression analysis showed that mrN stage positive and Ktrans were independent predictors of lymph node metastasis in colorectal adenocarcinoma. The AUC of mrN stage + Ktrans (0.788) in predicting lymph node metastasis of rectal adenocarcinoma was higher than single mrN stage (0.713) and Ktrans (0.650). Conclusion Quantitative parameter Ktrans of dynamic contrast-enhanced MR combined with mrN stage can improve the prediction ability of patients with rectal adenocarcinoma lymph node metastasis. -
表 1 两组一般资料
Table 1. General data between the two groups (n)
Index With LNM(n=72) Without LNM(n=124) t/χ2 P Gender(Male/Female, n) 49/23 72/52 1.925 0.165 Age (years, Mean±SD) 62.4±8.9 66.4±7.6 2.451 0.037 CEA (n) 5.903 0.015 <5 ng/mL 42 93 ≥5 ng/mL 30 31 CA19-9 (n) 1.926 0.165 <39 U/mL 64 117 ≥39 U/mL 8 7 Tumor length(cm, Mean±SD) 4.3±1.3 3.8±1.3 -1.920 0.057 Tumor location (n) 2.450 0.294 Upper 20 40 Middle 36 48 Lower 16 36 mrT stage (n) 12.645 <0.001 T1-T2 24 74 T3 48 50 mrN stage (n) 35.153 <0.001 N0 25 96 N1-N2 47 28 mrMRF (n) 7.867 0.005 Negative 51 108 Positive 21 16 mrEMVI (n) 4.934 0.026 Negative 50 103 Positive 22 21 CEA: Carcinoembryonic antigen; CA19-9: Glycoprotein antigen 19-9; mrT stage: HR-MRI reported tumor stage; mrN stage: HRMRI reported lymp node stage; mrMRF: HR-MRI reported mesorectal fasciae status; mrEMVI: HR-MRI reported extramural vascular invasion status; LNM: Lymph node metastasis. 表 2 两组DCE-MRI定量参数的比较
Table 2. Comparison of DCE-MRI quantitative parameters between the two groups
Variable With LNM(n=72) Without LNM(n=124) t P Kep [min-1, M(P25, P75)] 1.139(0.798, 2.372) 1.415(0.943, 2.449) 0.896 0.462 Ktrans [min-1, M(P25, P75)] 0.247(0.190, 0.330) 0.362(0.245, 0.502) 2.461 0.015 Ve (Mean±SD) 0.289±0.109 0.321±0.111 1.366 0.172 iAUC (Mean±SD) 13.966±8.526 16.565±9.529 0.885 0.377 Kep: Rate constant from extravascular-extracellular space to blood plasma (min-1); Ktrans: Volume transfer constant between extravascular-extracellular space and blood plasma (min-1); Ve: Extravascular-extracellular space volume per unit tissue volume; iAUC: Initial area under the curve. 表 3 不同参数预测直肠癌淋巴结转移的能力
Table 3. Ability of different parameters to predict lymph node metastasis of rectal cancer
Variable AUC Cutoff value Specificity(%) Sensitivity(%) Negative predictive value(%) Positive predictive value(%) P Age 0.626 < 59.5 years old 88.7 27.8 67.9 58.5 0.039 CEA 0.623 abnormal 77.4 47.2 71.6 54.8 0.043 mrN stage 0.713 N1-N2 77.4 66.7 80.0 63.2 < 0.001 mrT stage 0.632 T3 61.4 66.7 75.5 49.0 0.03 Ktrans 0.650 < 0.298 min-1 67.7 69.4 79.2 55.6 0.015 mr EMVI 0.592 Positive 82.3 36.1 68.9 54.2 0.131 mrMRF 0.607 Positive 93.5 27.8 69.0 71.4 0.079 CEA: Carcinoembryonic antigen. 表 4 预测直肠癌淋巴结转移的Logistic回归分析
Table 4. Logistic regression analysis for predicting lymph node metastasis of rectal cancer
Variable Univariate Logistic regression analysis Multivariate Logistic regression analysis OR(95% CI) P OR(95% CI) P Age 0.329(0.112,0.967) 0.043 CEA 2.742(1.129,6.658) 0.025 mrN stage 6.857(2.749,17.096) < 0.001 5.573(2.150,14.462) < 0.001 mrT stage 2.959(1.254,6.987) 0.013 Ktrans(min-1) 0.210(0.086,0.509) 0.001 0.273(0.105,0.709) 0.008 mr EMVI 2.261(1.022,6.721) 0.045 mrMRF 5.577(1.600,19.434) 0.007 表 5 Ktrans、mrN stage和联合预测直肠癌淋巴结转移效能的比较
Table 5. Comparison of Ktrans, mrN stage and combined prediction of lymph node metastasis in rectal cancer
Variable AUC 95% CI Specificity(%) Sensitivity(%) mrN stage 0.713 0.613-0.829 77.4 66.7 Ktrans 0.650 0.529-0.767 67.7 69.4 Ktrans+mrN stage 0.788 0.694-0.882 56.5 88.9 -
[1] Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-49. doi: 10.3322/caac.21660 [2] Baidoun F, Elshiwy K, Elkeraie Y, et al. Colorectal cancer epidemiology: recent trends and impact on outcomes[J]. Curr Drug Targets, 2021, 22(9): 998-1009. doi: 10.2174/18735592MTEx9NTk2y [3] Parascandola M, Pearlman PC, Eldridge L, et al. The development of global cancer research at the United States national cancer institute[J]. J Natl Cancer Inst, 2022, 114(9): 1228-37. doi: 10.1093/jnci/djac104 [4] Dekker E, Tanis PJ, Vleugels JLA, et al. Colorectal cancer[J]. Lancet, 2019, 394(10207): 1467-80. doi: 10.1016/S0140-6736(19)32319-0 [5] Patel RK, Rahman S, Schwantes IR, et al. Updated management of colorectal cancer liver metastases: scientific advances driving modern therapeutic innovations[J]. Cell Mol Gastroenterol Hepatol, 2023, 16(6): 881-94. doi: 10.1016/j.jcmgh.2023.08.012 [6] 周杨, 陈首名. 结直肠癌MSCT诊断及与病理分期对照研究[J]. 中国CT和MRI杂志, 2022, 20(4): 153-5. https://www.cnki.com.cn/Article/CJFDTOTAL-CTMR202204061.htm [7] 赵龙, 杨长江, 林易霖, 等. 人工智能在结直肠癌病理研究中的应用进展[J]. 中华实验外科杂志, 2022, 39(3): 597-601. https://www.cnki.com.cn/Article/CJFDTOTAL-ZWWK202211011.htm [8] Ruusuvuori P, Valkonen M, Latonen L. Deep learning transforms colorectal cancer biomarker prediction from histopathology images [J]. Cancer Cell, 2023, 41(9): 1543-5. doi: 10.1016/j.ccell.2023.08.006 [9] Wagner SJ, Reisenbüchler D, West NP, et al. Transformer-based biomarker prediction from colorectal cancer histology: a large-scale multicentric study[J]. Cancer Cell, 2023, 41(9): 1650-61. e4. doi: 10.1016/j.ccell.2023.08.002 [10] Zhang S, Cai GX, Xie PY, et al. Improving prognosis and assessing adjuvant chemotherapy benefit in locally advanced rectal cancer with deep learning for MRI: a retrospective, multi-cohort study[J]. Radiother Oncol, 2023, 188: 109899. doi: 10.1016/j.radonc.2023.109899 [11] Kim HR, Kim SH, Nam KH. Association between dynamic contrast-enhanced MRI parameters and prognostic factors in patients with primary rectal cancer[J]. Curr Oncol, 2023, 30(2): 2543-54. doi: 10.3390/curroncol30020194 [12] 李博云, 曾旭. DCE-MRI联合DWI在直肠癌术前TN分期中的应用价值[J]. 中国CT和MRI杂志, 2023, 21(2): 142-4. https://www.cnki.com.cn/Article/CJFDTOTAL-CTMR202302030.htm [13] 中国结直肠癌诊疗规范专家组. 国家卫生健康委员会中国结直肠癌诊疗规范(2020年版)[J]. 中华胃肠外科杂志, 2020, 23(6): 521-40. [14] Golia Pernicka JS, Sheedy SP, Ernst RD, et al. MR staging of anal cancer: what the radiologist needs to know[J]. Abdom Radiol (NY), 2019, 44(11): 3726-39. doi: 10.1007/s00261-019-02020-4 [15] 秦小雪, 张文, 高红, 等. 高分辨率磁共振成像与正电子发射计算机体层显像仪在直肠癌周围转移性小淋巴结检出及术前T分期诊断中的应用价值[J]. 中国医药导报, 2023, 20(16): 145-8, 153. https://www.cnki.com.cn/Article/CJFDTOTAL-YYCY202316033.htm [16] Peacock O, Manisundaram N, DiBrito S, et al. Magnetic resonance imaging directed surgical decision making for lateral pelvic lymph node dissection in rectal cancer after total neoadjuvant therapy (TNT)[J]. Ann Surg, 2022, 276: 654-64. doi: 10.1097/SLA.0000000000005589 [17] Scheenen TWJ, Zamecnik P. The role of magnetic resonance imaging in (future) cancer staging[J]. Invest Radiol, 2021, 56(1): 42-9. doi: 10.1097/RLI.0000000000000741 [18] Inchingolo R, Maino C, Cannella R, et al. Radiomics in colorectal cancer patients[J]. World J Gastroenterol, 2023, 29(19): 2888-904. doi: 10.3748/wjg.v29.i19.2888 [19] Fillon M. Study identifies signs and symptoms of colorectal cancer risk at younger ages[J]. CA A Cancer J Clinicians, 2023, 73(5): 448-50. doi: 10.3322/caac.21810 [20] Zeng DX, Yang Z, Tan LH, et al. Risk factors for lateral pelvic lymph node metastasis in patients with lower rectal cancer: a systematic review and meta-analysis[J]. Front Oncol. 2023, 13: 1219608. doi: 10.3389/fonc.2023.1219608 [21] Aldilaijan AF, Kim YI, Kim CW, et al. Clinical implication of tissue carcinoembryonic antigen expression in association with serum carcinoembryonic antigen in colorectal cancer[J]. Sci Rep, 2023, 13: 7616. doi: 10.1038/s41598-023-34855-9 [22] Muñoz-Montaño WR, López-Basave HN, Castillo-Morales A, et al. Persistent high levels of carcinoembryonic antigen after tumor resection are associated with poorer survival outcomes in patients with resected colon cancer[J]. BMC Cancer, 2023, 23(1): 678. doi: 10.1186/s12885-023-11126-4 [23] Xu HS, Zhao WY, Guo WB, et al. Prediction model combining clinical and MR data for diagnosis of lymph node metastasis in patients with rectal cancer[J]. J Magn Reson Imaging, 2021, 53(3): 874-83. doi: 10.1002/jmri.27369 [24] Tong PF, Sun DQ, Chen GQ, et al. Biparametric magnetic resonance imaging-based radiomics features for prediction of lymphovascular invasion in rectal cancer[J]. BMC Cancer, 2023, 23(1): 61. doi: 10.1186/s12885-023-10534-w [25] 陈鹰, 王贵良, 马荣, 等. 高分辨率MRI联合动态增强扫描对老年直肠癌术前T、N分期的诊断价值[J]. 中国中西医结合影像学杂志, 2022, 20(6): 523-7. https://www.cnki.com.cn/Article/CJFDTOTAL-JHYX202206004.htm [26] Yuan J, Liu K, Zhang Y, et al. Quantitative dynamic contrast-enhance MRI parameters for rectal carcinoma characterization: correlation with tumor tissue composition[J]. World J Surg Oncol, 2023, 21(1): 306. doi: 10.1186/s12957-023-03193-5 [27] Bakke KM, Meltzer S, Grøvik E, et al. Sex differences and tumor blood flow from dynamic susceptibility contrast MRI are associated with treatment response after chemoradiation and longterm survival in rectal cancer[J]. Radiology, 2020, 297(2): 352-60. doi: 10.1148/radiol.2020200287 [28] Yu J, Xu Q, Huang DY, et al. Prognostic aspects of dynamic contrast-enhanced magnetic resonance imaging in synchronous distant metastatic rectal cancer[J]. Eur Radiol, 2017, 27(5): 1840-7. doi: 10.1007/s00330-016-4532-y [29] El Khababi N, Beets-Tan RGH, Tissier R, et al. Comparison of MRI response evaluation methods in rectal cancer: a multicentre and multireader validation study[J]. Eur Radiol, 2023, 33(6): 4367-77. -







 下载:
下载: