Correlation analysis of microcirculation disorders with infarct size, myocardial strain, and clinical prognosis in patients with acute myocardial infarction
-
摘要:
目的 分析心脏磁共振评估急性心肌梗死患者微循环障碍(MVO)与梗死面积、心肌应变及临床预后的相关性。 方法 选择2022年6~12月蚌埠医学院第一附属医院收治的24例因ST段抬高型心肌梗死(STEMI)行经皮冠状动脉介入治疗术治疗的患者为对象,其中男性22例,女性2例,年龄55.3±11.3岁。所有患者于术后5~7 d进行心脏磁共振检查,根据是否出现MVO,将24例患者分为MVO组(n=16)和非MVO组(n=8),比较两组基线资料、心功能、心肌梗死面积(LGE%)、心肌应变。对出院的STEMI患者进行平均6月的门诊或电话随访,记录心血管不良事件(MACEs)的发生情况,比较两组发生MACEs事件的差异。本研究定义的MACEs事件包括:再发胸痛、心力衰竭、脑卒中、再发心梗、出血、再次血运重建、支架内血栓、支架内再狭窄、死亡。 结果 MVO组梗死节段径向应变、梗死节段周向及整体周向应变功能均低于无MVO组(P < 0.05);MVO组心肌梗死面积大于无MVO组(25.18%±10.51% vs 9.93%±5.96%)。MVO组左心室射血分数与径向应变及周向应变呈极强相关关系[r=0.815 (0.536~0.934),P < 0.001;r=-0.938(-0.978~-0.852),P < 0.001],与纵向应变呈强相关关系[r=-0.767(-0.915~-0.437),P < 0.001]。二元回归分析中LGE%及梗死节段周向应变是STEMI患者发生MVO的独立危险因素。单因素分析ROC曲线显示,LGE%可以辅助于MVO的诊断,曲线下面积(AUC)为0.922(0.796~1.000),其最佳截断点为14.92%,敏感度为87.5%,特异性为87.5%(P < 0.05)。梗死节段周向应变对MVO也具有诊断价值,AUC为0.781(0.591~0.971),其最佳截断点为10.58%,敏感度为62.5%,特异性为87.5%(P < 0.05)。LGE%联合梗死节段周向应变后,其对MVO诊断的AUC及敏感度均上升,AUC为0.938(0.827~1.000),敏感度为93.8%,特异性为87.5%(P < 0.05)。随访所有STEMI患者,10例发生MACEs事件(41.7%),两组患者发生MACEs事件的差异无统计学意义(P=0.558)。 结论 LGE%、心肌梗死节段周向应变是STEMI患者经皮冠状动脉介入治疗术后出现MVO的独立危险因素,也分别对MVO具有较高的诊断价值,两者联合诊断MVO时诊断价值更高。径向应变、周向应变、纵向应变与MVO组左心室射血分数具有较强的相关性。 Abstract:Objective To explore the correlation analysis between myocardial MRI assessment of microcirculation disorders (MVO), infarct size, myocardial strain and clinical prognosis in patients with acute myocardial infarction. Methods A total of 24 patients treated with percutaneous coronary intervention for ST segment elevation myocardial infarction (STEMI) at the First Affiliated Hospital of Bengbu Medical College from June 2022 to December 2022 were selected as the study subjects, including 22 male and 2 female patients, with an average age of 55.3±11.3 years old. All patients underwent cardiac magnetic resonance imaging 5-7 d after surgery. Based on the presence and absence of microcirculation disorders, the 24 patients were divided into MVO group (n=16) and non MVO group (n=8), then compared the baseline data, cardiac function, myocardial infarction area (LGE%) and myocardial strain between the two groups. The discharged STEMI patients were followed up for an average of 6 months in the outpatient clinic or by telephone to record the occurrence of adverse cardiovascular events (MACE events), and compare the differences in the MACE events between the two groups. The MACE events defined in this study include recurrent chest pain, heart failure, stroke, recurrent myocardial infarction, bleeding, revascularization, in-stent thrombosis, in-stent restenosis and death. Results The radial strain, circumferential strain, and overall circumferential strain of the infarcted segment in the MVO group were lower than those in the non MVO group (P < 0.05); The myocardial infarction area in the MVO group was larger than that in the non MVO group (25.18%±10.51% vs 9.93%±5.96%). The left ventricular ejection fraction in the MVO group was highly correlated with radial and circumferential strain [r=0.815(0.536-0.934), P < 0.001; r=-0.938(-0.978—-0.852), P < 0.001], and strongly correlated with longitudinal strain [r=-0.767(-0.915—-0.437), P < 0.001]. LGE% and circumferential strain of the infarcted segment in binary regression analysis are independent risk factors for MVO in STEMI patients. In univariate analysis, ROC curve showed that LGE% could assist in the diagnosis of MVO. The AUC was 0.922 (0.796-1.000), with an optimal cutoff point of 14.92%, sensitivity of 87.5% and specificity of 87.5% (P < 0.05). The circumferential strain of the infarcted segment also has diagnostic value for MVO, with an AUC of 0.781 (0.591- 0.971), an optimal cutoff point of 10.58%, sensitivity of 62.5%, and specificity of 87.5% (P < 0.05). After LGE% was combined with the circumferential strain of the infarcted segment, its AUC and sensitivity for MVO diagnosis increased, with an AUC of 0.938 (0.827-1.000), a sensitivity of 93.8%, and a specificity of 87.5%(P < 0.05). MACE events occurred in a total of 10 STEMI patients followed up in the study, accounting for 41.7% of the total number. There was no statistically significant difference in the occurrence of MACEs events between the two groups of patients (P=0.558). Conclusion LGE% and circumferential strain of myocardial infarction segment are independent risk factors for MVO after percutaneous coronary intervention in STEMI patients, and they also have high diagnostic value for MVO. The combination of the two has higher diagnostic value for MVO. There is a strong correlation between radial strain, circumferential strain, and longitudinal strain and left ventricular ejection fraction in the MVO group. -
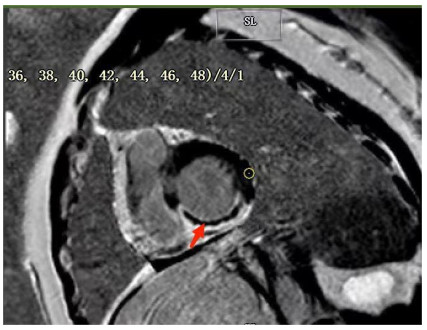
图 2 男,55岁,急性下壁心肌梗死介入术后7 d行CMR检查
Figure 2. A 55- year- old male underwent CMR examination 7 d after interventional surgery for acute inferior myocardial infarction. A: The left ventricular myocardial radial strain decreased (normal value 30%-40%); B: The left ventricular myocardial circumferential strain decreased (normal value - 15%- 20%); C: The left ventricular myocardial longitudinal strain peakdecreased (normal value-15%-20%).
表 1 MVO组与非MVO组一般资料分析
Table 1. Analysis of general data of MVO group and non-MVO group
Index MVO group (n=16) Non-MVO group (n=8) P Gender(Male/Female) 14/2 8/0 0.296 Age(year) 57.31±12.41 51.25±7.25 0.223 Diabetes 6(37.5) 2(25.0) 0.607 Smoking 12(75.0) 6(75.0) 0.698 G(mmol/L) 8.30(6.29, 12.10) 8.34(5.89, 9.78) 0.624 UA (μmol/L) 291.00(249.15, 386.75) 377.00(304.00, 396.75) 0.270 CR(μmol/L) 66.50(57.25, 90.25) 69.50(55.50, 82.00) 0.878 LDL(mmol/L) 2.70(2.15, 2.92) 2.68(1.49, 3.85) 0.976 HDL(mmol/L) 0.96(0.82, 1.12) 0.91(0.86, 1.01) 0.806 TN-I(μg/L) 17.19(11.84, 29.73) 10.28(5.05, 19.75) 0.221 CK-MB(U/L) 42.50(31.50, 114.50) 40.00(29.75, 125.50) 0.902 G: Glucose; UA: Uric acid; CR: Creatinine; LDL: Low-density lipoprotein; HDL: High-density lipoprotein; TN-I: Troponin-I; CK-MB: Creatine kinase isoenzyme. 表 2 两组PCI术后结果相关信息分析
Table 2. Analysis of information related to the results after PCI between the two groups [n(%)]
Index MVO group (n=16) Non-MVO group (n=8) P Nember of diseased branches Single vessel 2(12.50) 3(37.50) 0.053 Two blood vessels 3(18.75) 2(25.00) 0.722 Three blood vessels 11(68.75) 3(37.50) 0.074 Criminal blood vessel LAD 8(50.00) 5(62.50) 0.562 LCX 2(12.50) 0(00.00) 0.296 RCA 6(37.50) 3(37.5) >0.999 Gensini (Mean±SD) 128.19±58.46 101.25±60.38 0.304 LAD: Left anterior descending artery; LCX: Left circumflex branch; RCA: Right coronary artery. 表 3 心功能及心肌梗死面积相关资料分析
Table 3. Analysis of data related to cardiac function and myocardial infarction area (Mean±SD)
Index MVO group (n=16) Non-MVO group (n=8) P LVEDV/BSA(mL/m2) 75.05±21.18 68.01±10.32 0.387 LVESV/BSA(mL/m2) 41.45±20.19 30.61±7.73 0.160 LVEF(%) 45.81±16.21 55.50±5.93 0.119 LGE(%) 25.18±10.51 9.93±5.96 <0.001 LVEDV/BSA: Left ventricular end-diastolic volume/body surface area; LVESV/BSA: Left ventricular end-systolic volume/body surface area; LVEF: Left ventricular ejection fraction; LGE: Late gadolinium enhancement. 表 4 心肌强化节段心肌应变功能参数分析
Table 4. Analysis of myocardial strain function parameters in dead segment of myocardial infarction (%, Mean±SD)
Functiongal parameters MVO group (n=16) Non-MVO group (n=8) P Radial strain in infarction stage 15.48±8.18 22.25±9.21 0.032 Circumferential strain in infarction stage -10.86±3.48 -14.81±4.23 0.023 Longitudinal strain in infarction stage -10.57±4.79 -11.61±1.52 0.438 表 5 心肌整体各方向应变比较
Table 5. Comparison of strain in all directions of myocardium (Mean±SD)
Functiongal parameters MVO group (n=16) Non-MVO group (n=8) P Global radial strain 26.19±9.81 31.78±11.27 0.223 Global circumferential strain -14.61±3.73 -17.86±3.14 0.046 Overall longitudinal strain -11.49±4.87 -12.87±2.91 0.472 表 6 MVO联合诊断的曲线下面积
Table 6. Area under the curve for MVO joint diagnosis
Project AUC P 95% CI LGE% 0.922 0.027 0.796-1.000 Circumferential strain of infarct segments 0.781 0.001 0.591-0.971 Joint diagnosis 0.938 0.001 0.827-1.000 -
[1] 中华医学会心血管病学分会, 中华心血管病杂志编辑委员会. 急性ST段抬高型心肌梗死诊断和治疗指南(2019)[J]. 中华心血管病杂志, 2019, 47(10): 766-83. https://www.cnki.com.cn/Article/CJFDTOTAL-ZJXB201709002.htm [2] Hamm CW, Bassand JP, Agewall S, et al. ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: the Task Force for the management of acute coronary syndromes (ACS) in patients presenting without persistent ST-segment elevation of the European Society of Cardiology (ESC)[J]. Eur Heart J, 2011, 32 (23): 2999-3054. doi: 10.1093/eurheartj/ehr236 [3] van Kranenburg M, Magro M, Thiele H, et al. Prognostic value of microvascular obstruction and infarct size, as measured by CMR in STEMI patients[J]. JACC, 2014, 7(9): 930-9. [4] de Waha S, Patel MR, Granger CB, et al. Relationship between microvascular obstruction and adverse events following primary percutaneous coronary intervention for ST-segment elevation myocardial infarction: an individual patient data pooled analysis from seven randomized trials[J]. Eur Heart J, 2017, 38(47): 3502-10. doi: 10.1093/eurheartj/ehx414 [5] 李璐, 赵世华. 磁共振成像识别急性心肌梗死后微循环障碍的研究进展[J]. 中华心血管病杂志, 2019, 47(4): 335-8. doi: 10.3760/cma.j.issn.0253-3758.2019.04.014 [6] Galasko GI, Basu S, Lahiri A, et al. A prospective comparison of echocardiographic wall motion score index and radionuclide ejection fraction in predicting outcome following acute myocardial infarction[J]. Heart, 2001, 86(3): 271-6. doi: 10.1136/heart.86.3.271 [7] Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation[J]. Eur Heart J, 2021, 42 (14): 1289-367. doi: 10.1093/eurheartj/ehaa575 [8] Kramer CM, Barkhausen J, Bucciarelli-Ducci C, et al. Standardized cardiovascular magnetic resonance imaging (CMR) protocols: 2020 update[J]. J Cardiovasc Magn Reson, 2020, 22(1): 17. doi: 10.1186/s12968-020-00607-1 [9] Cerqueira MD, Weissman NJ, Dilsizian V, et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association[J]. Circulation, 2002, 105(4): 539-42. doi: 10.1161/hc0402.102975 [10] Doherty DJ, Sykes R, Mangion K, et al. Predictors of microvascular reperfusion after myocardial infarction[J]. Curr Cardiol Rep, 2021, 23(3): 21. doi: 10.1007/s11886-021-01442-1 [11] Christoph G, Stark Anselm W, Kady F, et al. Diagnostic performance of cardiac magnetic resonance segmental myocardial strain for detecting microvascular obstruction and late gadolinium enhancement in patients presenting after a ST-elevation myocardial infarction[J]. Front Cardiovasc Med, 2022, 9: 909204. doi: 10.3389/fcvm.2022.909204 [12] Iwakura K, Ito H, Ikushima M, et al. Association between hyperglycemia and the no-reflow phenomenon inpatients with acute myocardial infarction[J]. J Am Coll Cardiol, 2003, 41(1): 1-7. doi: 10.1016/S0735-1097(02)02626-8 [13] Resnic FS, Wainstein M, Lee MKY, et al. No-reflow is an independent predictor of death and myocardial infarction after percutaneous coronary intervention[J]. Am Heart J, 2003, 145(1): 42-6. doi: 10.1067/mhj.2003.36 [14] Gick M, Jander N, Bestehorn HP, et al. Randomized evaluation of the effects of filter-based distal protection on myocardial perfusion and infarct size after primary percutaneous catheter intervention in myocardial infarction with and without ST-segment elevation[J]. Circulation, 2005, 112(10): 1462-9. doi: 10.1161/CIRCULATIONAHA.105.545178 [15] Marc MC, Iancu AC, Bãlãnescu Ş, et al. Microvascular obstruction in acute myocardial infarction: an old and unsolved mystery[J]. Med Pharm Rep, 2019, 92(3): 216-9. [16] Song LS, Ma XH, Zhao XX, et al. Validation of black blood late gadolinium enhancement (LGE) for evaluation of myocardial infarction in patients with or without pathological Q-wave on electrocardiogram (ECG)[J]. Cardiovasc Diagn Ther, 2020, 10(2): 124-34. doi: 10.21037/cdt.2019.12.11 [17] 蔡宜婷, 邢浩然, 郝明辉, 等. STEMI再灌注微血管损伤的心脏磁共振成像新进展[J]. 中华心血管病杂志, 2022, 50(12): 1237-42. doi: 10.3760/cma.j.cn112148-20220403-00240 [18] Rigolli M, Anandabaskaran S, Christiansen JP, et al. Bias associated with left ventricular quantification by multimodality imaging: a systematic review and meta-analysis[J]. Open Heart, 2016, 3(1): e000388. doi: 10.1136/openhrt-2015-000388 [19] Reant P, Barbot L, Touche C, et al. Evaluation of global left ventricular systolic function using three-dimensional echocardiography speckle-tracking strain parameters[J]. J Am Soc Echocardiogr, 2012, 25(1): 68-79. doi: 10.1016/j.echo.2011.10.009 [20] Pierpaolo P, Rolf S, Manuel BP, et al. Left ventricular global myocardial strain assessment: are CMR feature-tracking algorithms useful in the clinical setting?[J]. La Radiol Med, 2020, 125(5): 444-50. doi: 10.1007/s11547-020-01159-1 [21] Dobrovie M, Barreiro-Pérez M, Curione D, et al. Inter-vendor reproducibility and accuracy of segmental left ventricular strain measurements using CMR feature tracking[J]. Eur Radiol, 2019, 29(12): 6846-57. doi: 10.1007/s00330-019-06315-4 [22] Li H, Qu YY, Metze P, et al. Quantification of biventricular myocardial strain using CMR feature tracking: reproducibility in small animals[J]. BioMed Res Int, 2021, 2021: 1-14. [23] Bergerot C, Mewton N, Lacote- Roiron C, et al. Influence of microvascular obstruction on regional myocardial deformation in the acute phase of myocardial infarction: a speckle-tracking echocardiography study[J]. J Am Soc Echocardiogr, 2014, 27(1): 93-100. doi: 10.1016/j.echo.2013.09.011 [24] Bekkers SCAM, Smulders MW, Passos VL, et al. Clinical implications of microvascular obstruction and intramyocardial haemorrhage in acute myocardial infarction using cardiovascular magnetic resonance imaging[J]. Eur Radiol, 2010, 20(11): 2572-8. doi: 10.1007/s00330-010-1849-9 [25] 侯洁, 肖俊睿, 孙玉, 等. 磁共振延迟强化对急性心肌梗死的诊断价值[J]. 中华心血管病杂志, 2018, 46(8): 635-9. doi: 10.3760/cma.j.issn.0253-3758.2018.08.011 [26] Szummer K, Wallentin L, Lindhagen L, et al. Improved outcomes in patients with ST-elevation myocardial infarction during the last 20 years are related to implementation of evidence- based treatments: experiences from the SWEDEHEART registry 1995-2014 [J]. Eur Heart J, 2017, 38(41): 3056-65. doi: 10.1093/eurheartj/ehx515 [27] Durante A, Camici PG. Response by durante and camici to letter regarding article, "identification of high-risk patients after STsegment elevation myocardial infarction: comparison between angiographic and magnetic resonance parameters"[J]. Circ, 2017, 10(9): e005841. -







 下载:
下载: