Value of 18F-FDG combined with 11C-CFT PET imaging in analyzing Parkinson's disease
-
摘要:
目的 分析18氟-氟代脱氧葡萄糖(18F-FDG)和11C-甲基-N-2β-甲基酯-3β-(4-氟-苯基)托烷(11C-CFT)PET显像联合诊断、病情评价、鉴别诊断帕金森病(PD)的应用价值。 方法 选取我院2018年9月~2022年3月收治的107例根据临床症状和口服美多芭有效性初步诊断为PD的患者作为PD组,另选取43例健康体检者作为对照组,两组均进行18F-FDG和11C-CFT PET显像。11C-CFT分析:将PD组分为H&Y 1~2(早期)和H&Y 3~5(晚期)2个级别,测得PD组和正常对照组尾状核、前壳核、后壳核的11C-CFT标准摄取值,采用独立样本t检验比较PD组和对照组的差异,再将尾状核、前壳核、后壳核分为3组,采用单因素方差分析进行组间对比。18F-FDG分析:结合患者的运动症状进行壳核、丘脑、尾状核及额顶颞枕叶的代谢改变情况分析。 结果 11C-CFT PET显像:H & 1~2(早期)时起病肢体对侧后壳核为低摄取(P < 0.05),与正常组比较部分患者起病肢体对侧前壳核、起病肢体同侧后壳核呈低摄取,差异有统计学意义(P < 0.05),而起病肢体同侧前壳核、尾状核基本正常,H & 3~5(晚期)时起病肢体对侧后壳核摄取更低(P < 0.05),起病肢体对侧和同侧后壳核、前壳核均呈不均匀性低摄取,差异有统计学意义(P < 0.05);尾状核、前壳核、后壳核三者组间比较,见早、晚期时起病肢体对侧和同侧组间差异均有统计学意义(P < 0.05)。18F-FDG PET显像:107例PD患者表现为81.31%壳核代谢增高、54.20%丘脑代谢增高、26.17%尾状核代谢增高,大脑皮层的代谢减低区见89.72%顶叶代谢减低、57.94%额叶代谢减低、68.22颞叶代谢减低、14.02%枕叶代谢减低。 结论 18F-FDG联合11C-CFT PET双核素影像能够为临床提供客观的数据,辅助诊断、鉴别诊断和评价PD。 -
关键词:
- 帕金森病 /
- 18F-FDG /
- 11C-CFT /
- 正电子发射计算机断层 /
- 诊断
Abstract:Objective To study the application of 18F-FDG and 11C-CFT PET imaging in the combined diagnosis, evaluation, and differential diagnosis of Parkinson's disease (PD). Methods A total of 107 patients admitted to our hospital from September 2018 to March 2022 who were initially diagnosed as PD according to clinical symptoms and the effectiveness of oral metoba were selected, and 43 healthy subjects were selected as the control group. Both groups underwent 18F-FDG and 11C-CFT PET imaging. 11C- CFT analysis: The PD group was divided into two levels: H&Y 1- 2 (early) and H&Y 3- 5 (late). The 11C- CFT standard uptake values of the caudate nucleus, anterior putamen nucleus, and posterior putamen nucleus were measured in the PD group and the control group. Independent sample t-test was used to compare the PD group and the control group. The caudate nucleus, anterior putamen nucleus, and posterior putamen nucleus were then divided into three groups, Using oneway ANOVA for inter group comparison; 18F-FDG analysis: Analyze the metabolic changes in the putamen, thalamus, caudate nucleus, and frontal parietal temporal occipital lobe based on the patient's motor symptoms. Results 11C-CFT PET imaging: At the early stage of H&Y 1-2, the contralateral posterior putamen of the affected limb showed low metabolism (P < 0.05). Compared with the control group, some patients showed low metabolism in the contralateral anterior putamen and ipsilateral posterior putamen of the affected limb, and the results were statistically significant (P < 0.05), while the ipsilateral anterior putamen and caudate nuclei of the affected limb were basically normal. In H&Y 3- 5 (late stage), the metabolism of the posterior putamen on the opposite side of the affected limb was lower (P < 0.05). The opposite and ipsilateral posterior putamen and anterior putamen of the affected limb exhibited uneven hypometabolism, and the results were statistically significant (P < 0.05); Comparing the caudate nucleus, anterior putamen nucleus, and posterior putamen nucleus, there were differences between the contralateral and ipsilateral groups of the affected limb in the early and late stages (P < 0.05), providing additional evidence that the lesion originated in the posterior putamen nucleus; 18F-FDG PET imaging: 107 Parkinson's patients showed 81.31% increase in putamen metabolism, 54.20% increase in thalamus metabolism, and 26.17% increase in caudate nucleus metabolism. The metabolic reduced area of the cerebral cortex showed 89.72% decrease in parietal lobe metabolism, 57.94% decrease in frontal lobe metabolism, 68.22% decrease in temporal lobe metabolism, and 14.02% decrease in occipital lobe metabolism. Conclusion 18F-FDG combined with 11C-CFT PET binuclear imaging provide objective data for clinical diagnosis, differential diagnosis, and evaluation of PD. -
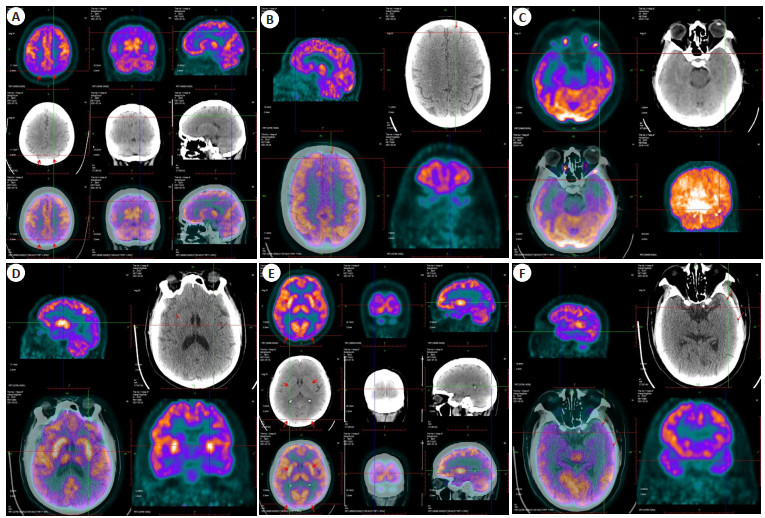
图 1 18F-FDG PET显像
Figure 1. 18F-FDG PET imaging. A: Bilateral parietal lobule was hypometabolicm; B: Left superior frontal gyrus was hypometabolic; C: Low metabolism in left superior temporal gyrus; D: Bilateral putamen and thalamus were hypermetabolic, especially the putamen; E: Bilateral putamen and thalamus had high metabolism, and bilateral occipital lobe had low metabolism; F: The left superior and middle temporal gyrus were hypometabolic.
图 2 正常对照组(A)及PD组(B)尾状核、前壳核、后壳核SUV值波动范围
Figure 2. Fluctuation range of SUV values in caudate nucleus, anterior putamen and posterior putamen in control group (A) and PD group (B). A: The metabolic degree of anterior putamen was higher than that of caudate nucleus and posterior putamen, SUV values fluctuate from 1.86 to 2.12; B: The metabolism of caudate nucleus, anterior putamen and posterior putamen all decreased, with the most obvious decrease in the posterior putamen, followed by the anterior putamen and finally the caudate nucleus. The degree of reduction of SUV value fluctuated from 1.31 to 2.00.
图 3 11C-CFT PET显像
Figure 3. 11C-CFT PET imaging. A: Normal 11C-CFT PET images; B: The right putamen had a mild to moderate reduction; C: The left putamen showed moderate to severe reduction; D: The putamen was severely reduced on both sides, especially on the right; E: The left putamen showed moderate to severe reduction; F: After deep brain electrode implantation, the right putamen showed moderate reduction, and the left showed moderate to severe reduction.
表 1 PD组18F-FDG代谢异常者例数和主要运动症状
Table 1. Number of abnormal 18F-FDG metabolism cases and main motor symptoms in PD group (n=107)
Abnormal metabolic site Clinical symptoms Bradykinesia (n=66) Rigidity (n=53) Tremor (n=80) Abnormal gait (n=58) Hypometabolic area of cerebral cortex Parietal lobe 61 48 73 53 Frontal lobe 37 32 47 33 Temporal lobe 43 33 51 44 Occipital lobe 9 9 10 9 Hypermetabolic area Putamen nucleus 52 44 65 48 Thalamus 37 32 45 35 Caudate nucleus 21 15 22 17 表 2 PD组、正常对照组尾状核、前壳核、后壳核11C-CFT摄取值对比
Table 2. Comparison of 11C-CFT uptake values in caudate nucleus, anterior putamen and posterior putamen nucleus of PD group and control group (Mean±SD)
Group Caudate nucleus P Anterior putamen P Posterior putamen P Control group (n=43) 2.02±0.44 2.12±0.44 1.86±0.49 PD group H&Y 1-2 (early, n=47) The opposite side of the starting limb 1.97±0.39 0.614 1.87±0.39 0.006 1.26±0.30 <0.001 The same side of the starting limb 2.05±0.44 0.755 2.05±0.45 0.491 1.46±0.35 <0.001 PD group H&Y 3-5 (late, n=60) The opposite side of the starting limb 1.93±0.38 0.299 1.78±0.36 <0.001 1.19±0.24 <0.001 The same side of the starting limb 2.00±0.44 0.832 1.91±0.42 0.015 1.34±0.33 <0.001 PD: Parkinson's disease. 表 3 PD组、正常对照组双侧尾状核、前壳核及后壳核组间对比
Table 3. Comparison of bilateral caudate nucleus, anterior putamen and posterior putamen nucleus between PD group and control group (Mean±SD)
Group Caudate nucleus Anterior putamen Posterior putamen P Control group (n=43) 2.02±0.44 2.12±0.44 1.86±0.49 0.101 PD group (n=107) 2.00±0.41 1.90±0.39 1.31±0.30 <0.001 H&Y 1-2 (early, n=47) The opposite side of the starting limb 1.97±0.39 1.87±0.39 1.25±0.30 <0.001 The same side of the starting limb 2.05±0.44 2.05±0.45 1.46±0.35 <0.001 H&Y 3-5 (late, n=60) The opposite side of the starting limb 1.93±0.38 1.78±0.36 1.19±0.24 <0.001 The same side of the starting limb 2.00±0.43 1.91±0.42 1.34±0.33 <0.001 表 4 正常对照组和PD组尾状核、前壳核及后壳核组间差异比较的P值
Table 4. Comparison of P value of caudate nucleus, anterior putamen and posterior putamen nucleus in PD and control group
Group C/A C/P A/C A/P P/C P/A Control group 0.310 0.101 0.310 0.008 0.101 0.008 PD group 0.127 <0.001 0.127 <0.001 <0.001 <0.001 H&Y 1-2(early) The opposite side of the starting limb 0.538 <0.001 0.538 <0.001 <0.001 <0.001 The same side of the starting limb >0.999 <0.001 >0.999 <0.001 <0.001 <0.001 H&Y 1-2(early) The opposite side of the starting limb 0.039 <0.001 0.039 <0.001 <0.001 <0.001 The same side of the starting limb 0.615 <0.001 0.615 <0.001 <0.001 <0.001 C: caudate nucleus; A: anterior putamen nucleus; P: posterior putamen nucleus. -
[1] 龚涛, 程卫玲, 陶娜, 等. 未经治疗的帕金森病患者全脑静息态功能磁共振成像的分数低频振幅研究[J]. 中国临床新医学, 2023, 16(4): 330-5. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYLN202304005.htm [2] 肖宁, 热米拉·玉山, 陆鹏, 等. 磁共振NODDI技术在帕金森认知障碍患者海马微观结构的应用[J]. 分子影像学杂志, 2022, 45(1): 35-9. doi: 10.12122/j.issn.1674-4500.2022.01.07 [3] 王慧, 刘子钰, 白侠, 等. 11C-β-CFT: 多巴胺转运蛋白显像剂的合成及其在帕金森病诊断中的应用[J]. 分子影像学杂志, 2023, 46(4): 605-8. doi: 10.12122/j.issn.1674-4500.2023.04.05 [4] 陈瑜, 李如雯, 谢树英, 等. 新型18F标记的多巴胺正电子发射显影剂前体的合成[J]. 化学试剂, 2022, 44(8): 1241-7. https://www.cnki.com.cn/Article/CJFDTOTAL-HXSJ202208024.htm [5] 邓伟胜, 楼云龙, 苏中振. 正电子发射断层扫描PET/CT在帕金森病患者早期诊断中的应用[J]. 医学信息, 2020, 33(18): 170-2. https://www.cnki.com.cn/Article/CJFDTOTAL-YXXX202018057.htm [6] 夏天. 18F-FDG PET/CT显像在意识障碍患者脑功能评估中的应用价值[J]. 影像研究与医学应用, 2021, 5(15): 39-40. https://www.cnki.com.cn/Article/CJFDTOTAL-YXYY202115016.htm [7] Caminiti SP, Sala A, Iaccarino L, et al. Brain glucose metabolism in Lewy body dementia: implications for diagnostic criteria[J]. Alzheimers Res Ther, 2019, 11(1): 20. doi: 10.1186/s13195-019-0473-4 [8] Ikeda K, Ebina J, Kawabe K, et al. Dopamine transporter imaging in parkinson disease: progressive changes and therapeutic modification after anti-parkinsonian medications[J]. Intern Med, 2019, 58(12): 1665-72. doi: 10.2169/internalmedicine.2489-18 [9] Sun X, Liu F, Liu QY, et al. Quantitative research of 11C-CFT and 18F-FDG PET in Parkinson's disease: a pilot study with NeuroQ software[J]. Front Neurosci, 2019, 13: 299. doi: 10.3389/fnins.2019.00299 [10] Heim B, Krismer F, De Marzi R, et al. Magnetic resonance imaging for the diagnosis of Parkinson's disease[J]. J Neural Transm, 2017, 124(8): 915-64. doi: 10.1007/s00702-017-1717-8 [11] 张杨, 蒋森, 刘娜, 等. 早期帕金森病步态障碍的临床特点及研究进展[J]. 医学信息, 2022, 35(6): 34-6, 40. https://www.cnki.com.cn/Article/CJFDTOTAL-YXXX202206008.htm [12] 齐彩云, 刘疏影, 许二赫. 不同运动亚型帕金森病影像学研究进展[J]. 中风与神经疾病杂志, 2020, 37(9): 851-6. https://www.cnki.com.cn/Article/CJFDTOTAL-ZFSJ202009022.htm [13] 李星江, 薛培源, 崔耀月, 等. 影像组学在帕金森病中的应用及研究进展[J]. 中国医药科学, 2022, 12(22): 34-7. https://www.cnki.com.cn/Article/CJFDTOTAL-GYKX202222009.htm [14] 杨云竣, 黄飚. 阿尔茨海默病与帕金森病的影像组学研究进展[J]. 国际医学放射学杂志, 2020, 43(1): 41-4. https://www.cnki.com.cn/Article/CJFDTOTAL-GWLC202001012.htm [15] 代玉银, 于丹, 赵红光, 等. 正电子放射性分子显像剂在老年帕金森病的应用进展[J]. 中国老年学杂志, 2020, 40(13): 2908-11. https://www.cnki.com.cn/Article/CJFDTOTAL-ZLXZ202013073.htm [16] 史倩文, 杨玲艳. 帕金森病患者认知功能障碍的研究进展[J]. 现代电生理学杂志, 2020, 27(1): 45-53. https://www.cnki.com.cn/Article/CJFDTOTAL-SDDS202001014.htm [17] 宋天彬, 卢洁. 18F-FDG PET脑显像和多巴胺能神经元突触功能PET脑显像研究帕金森病进展[J]. 中国医学影像技术, 2019, 35 (12): 1884-8. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYXX202212002.htm [18] Walker Z, Gandolfo F, Orini S, et al. Clinical utility of FDG PET in Parkinson's disease and atypical Parkinsonism associated with dementia[J]. Eur J Nucl Med Mol Imaging, 2018, 45(9): 1534-4 doi: 10.1007/s00259-018-4031-2 [19] 黄开合, 吴亮, 田有勇. 正电子发射计算机断层显像在帕金森病诊断中的应用[J]. 医学综述, 2020, 26(3): 559-64. https://www.cnki.com.cn/Article/CJFDTOTAL-YXZS202003029.htm [20] 左睿, 许璐, 庞华. 帕金森病神经影像学研究进展[J]. 中风与神经疾病杂志, 2021, 38(6): 574-6. https://www.cnki.com.cn/Article/CJFDTOTAL-ZFSJ202106028.htm [21] 马璐, 杨斌. 一体化PET/MR在神经系统中的应用进展[J]. 医学研究生学报, 2020, 33(11): 1209-13. https://www.cnki.com.cn/Article/CJFDTOTAL-JLYB202011018.htm [22] Albrecht F, Ballarini T, Neumann J, et al. FDG-PET hypometabolism is more sensitive than MRI atrophy in Parkinson's disease: a whole-brain multimodal imaging meta-analysis[J]. NeuroImage, 2019, 21: 101594. doi: 10.1016/j.nicl.2018.11.004 [23] 刘璇, 马建华. 帕金森病合并认知障碍18F-FDG PET/CT及血清学指标相关研究[J]. 新疆医科大学学报, 2020, 43(2): 158-9. [24] 许保磊, 章晓君, 孙炎, 等. 帕金森病合并抑郁的影响因素及脑部葡萄糖代谢的改变[J]. 临床神经病学杂志, 2022, 35(5): 324-8. https://www.cnki.com.cn/Article/CJFDTOTAL-LCSJ202205002.htm [25] 韩贵娟, 谢晓菲, 宋普姣, 等. 帕金森患者脑多巴胺转运体11C-CFT PET/CT显像特点的分析[J]. 国际放射医学核医学杂志, 2022(5): 270-6. https://www.cnki.com.cn/Article/CJFDTOTAL-HJBI202303010.htm -







 下载:
下载: