Diagnostic value of whole-body bone scan and SPECT/CT in nontuberculous mycobacterial-infected bone destruction in HIV-negative patients
-
摘要:
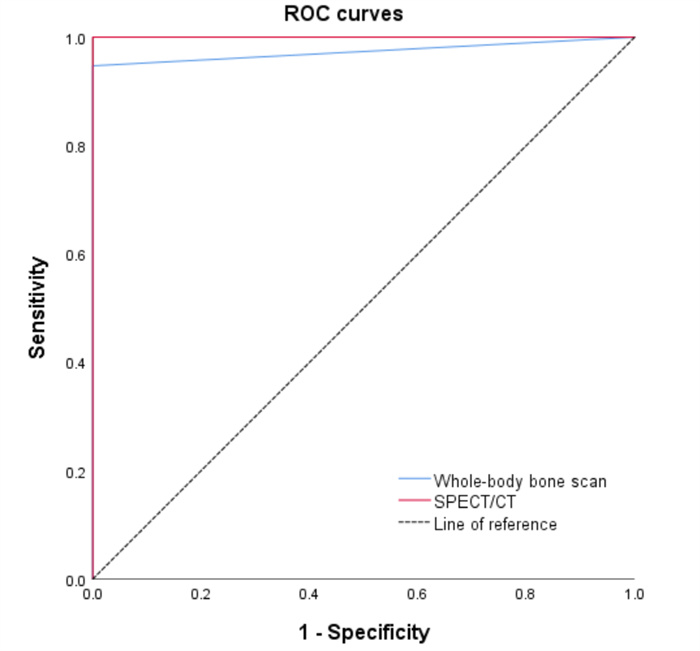
目的 探讨全身骨显像与SPECT/CT在人类免疫缺陷病毒阴性非结核分枝杆菌感染骨破坏的影像学特点及诊断价值。 方法 回顾性分析2020年11月~2022年7月在我院行全身骨显像及SPECT/CT显像并且确诊为非结核分枝杆菌(NTM)感染的25例患者,总结NTM感染骨破坏的临床表现、影像学表现等,并比较两种显像方法的诊断效能。 结果 全身骨显像诊断NTM感染骨破坏的敏感度为94.7%,特异性为100%,诊断符合率为96%;SPECT/CT显像诊断NTM感染骨破坏的敏感度、特异性、诊断符合率均为100%;二者对NTM感染骨破坏的诊断符合率的差异有统计学意义(P < 0.05)。病变主要分布于肋骨、四肢长骨、脊柱等部位,在全身骨显像上表现为放射性浓聚影,SPECT/CT可见以溶骨性改变为主。 结论 全身骨显像联合SPECT/CT断层融合显像可以监测到全身多个部位的病变,较好地显示NTM感染骨破坏的代谢情况及影像学特征,为临床诊疗提供帮助。 Abstract:Objective To investigate the imaging characteristics and diagnostic value of whole-body bone scan and SPECT/CT in nontuberculous mycobacterial-infected bone destruction in HIV-negative patients. Methods We retrospectively analyzed 25 patients with nontuberculous mycobacterial (NTM) who underwent whole-body bone scan and SPECT/CT in our hospital from November 2020 to July 2022. The clinical and imaging characteristics of NTM-infected bone destruction were summarized, and the diagnostic efficacy of the two imaging methods was compared. Results The sensitivity of the whole-body bone scan for the diagnosis of NTM-infected bone destruction was 94.7%, the specificity was 100%, and the diagnostic coincidence rate was 96%. The sensitivity, specificity, and diagnostic compliance rate of SPECT/CT were 100%. There was a significant difference in the diagnostic compliance rate of the two methods in diagnosing NTM-infected bone destruction (P < 0.05). The lesions were mainly distributed in the ribs, long bone of extremities, and spinal column, manifested as intense radioactive uptake on whole-body bone scan, and mostly showed osteolytic changes on SPECT/CT. Conclusion Whole- body bone scan combined with SPECT/CT imaging can monitor lesions in multiple parts of the body and better show the metabolic situation and imaging characteristics of NTM-infected bone destruction, which can help clinical diagnosis and treatment. -
Key words:
- nontuberculous mycobacterial /
- bone destruction /
- whole-body bone scan /
- SPECT/CT
-
图 2 NTM感染骨破坏的全身骨显像及SPECT/CT断层显像图像
Figure 2. Whole-body bone scan and SPECT/CT images of NTM-infected bone destruction. A: Whole-body bone scan showed increased radioactive concentrations in the left side of the 10th posterior rib, L1 vertebral body, L5 vertebral body and S1 vertebral body in a 42-year-old man. B: SPECT/CT scan revealed destruction of the bone in the L1 vertebral body, L5 vertebral body and S1 vertebral body with a clear and sclerotic edge (arrows). C: Whole-body bone scan showed increased radioactive concentrations in the left clavicle, right sternoclavicular joint, sternum, multiple ribs, multiple vertebral bodies, bilateral shoulder joints, right humerus, left femur, right knee, right tibia and left fibula in a 75-year-old woman. D: SPECT/CT scan revealed irregular osteolytic bone destruction and patchy high-density shadow in the sternum and multiple vertebral bodies.
表 1 全身骨显像与SPECT/CT显像对NTM感染骨破坏的诊断结果对比
Table 1. Comparison of whole-body bone scan and SPECT/CT imaging for the diagnosis of NTM-infected bone destruction [n(%)]
SPECT/CT Whole-body bone scan Total Bone destruction No bone destruction Bone destruction 18(72) 1(4) 19(76) No bone destruction 0(0) 6(24) 6(24) Total 18(72) 7(28) 25(100) χ2 =20.301, P < 0.001. 表 2 NTM感染骨破坏的影像学表现
Table 2. Distribution of lesions of bone destruction in NTM infection (n)
Distribution Numbers of patients Numbers of lesions Skull 11 47 Sternum 12 24 Clavicle 9 14 Rib 16 112 Spine 12 60 Pelvis 9 38 Long bones of the upper limbs 10 26 Long bones of the lower limbs 11 45 Sternoclavicular joint 4 5 Interphalangeal joint 2 6 Radiocarpal joint 3 6 Elbow joint 4 5 Shoulder joint 10 19 Knee joint 5 26 Ankle joint 6 13 Total 446 表 3 NTM感染骨破坏患者的临床特征
Table 3. Clinical features of patients with bone destruction in NTM infection
Symptom Numbers of patients (n) Percentage (%) Fever 13 68.42 Ostealgia 14 73.68 Arthralgia 5 26.32 Weight loss 12 63.16 Erythema 12 63.16 Subcutaneous abscess 13 68.42 Lymphadenopathy 13 68.42 Anorexia 5 26.32 -
[1] 叶素素, 刘晓清, 周宝桐, 等. 播散性非结核分枝杆菌病的临床和实验室检查特征[J]. 中国医学科学院学报, 2019, 41(2): 242-7. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYKX201902016.htm [2] Falkinham JO 3rd. Environmental sources of nontuberculous mycobacteria [J]. Clin Chest Med, 2015, 36(1): 35-41. doi: 10.1016/j.ccm.2014.10.003 [3] Wu UI, Holland SM. Host susceptibility to non-tuberculous mycobacterial infections [J]. Lancet Infect Dis, 2015, 15(8): 968-80. doi: 10.1016/S1473-3099(15)00089-4 [4] Wei MC, Banaei N, Yakrus MA, et al. Nontuberculous mycobacteria infections in immunocompromised patients: single institution experience [J]. J Pediatr Hematol Oncol, 2009, 31(8): 556-60. doi: 10.1097/MPH.0b013e31819ed274 [5] Griffith DE, Aksamit TR. Managing Mycobacterium avium complex lung disease with a little help from my friend [J]. Chest, 2021, 159(4): 1372-81. doi: 10.1016/j.chest.2020.10.031 [6] Ali J. A multidisciplinary approach to the management of nontuberculous mycobacterial lung disease: a clinical perspective [J]. Expert Rev Respir Med, 2021, 15(5): 663-73. doi: 10.1080/17476348.2021.1887734 [7] Thomson RM, working group at Queensland TB Control Centre and Queensland Mycobacterial Reference Laboratory NM. Changing epidemiology of pulmonary nontuberculous mycobacteria infections [J]. Emerg Infect Dis, 2010, 16(10): 1576-83. doi: 10.3201/eid1610.091201 [8] Winthrop KL, Marras TK, Adjemian J, et al. Incidence and prevalence of nontuberculous mycobacterial lung disease in a large U.S. managed care health plan, 2008-2015 [J]. Annals ATS, 2020, 17 (2): 178-85. doi: 10.1513/AnnalsATS.201804-236OC [9] Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated health care delivery systems [J]. Am J Respir Crit Care Med, 2010, 182(7): 970- 6. doi: 10.1164/rccm.201002-0310OC [10] 董双霞. 非结核分支杆菌肺病10例的影像学特点及分析[J]. 中国医药科学, 2013, 3(22): 205-7. https://www.cnki.com.cn/Article/CJFDTOTAL-GYKX201322096.htm [11] 宋文艳, 赵大伟, 张彤. 艾滋病并发非结核分枝杆菌感染的影像表现(附5例报告[) J]. 实用放射学杂志, 2011, 27(4): 501-4. [12] Tang MX, Huang J, Zeng W, et al. Retrospective analysis of 10 cases of disseminated nontuberculous mycobacterial disease with osteolytic lesions [J]. Infect Drug Resist, 2021, 14: 4667-79. doi: 10.2147/IDR.S337956 [13] 孙童, 韦智晓, 王秀萍, 等. 全身骨显像与SPECT/CT在马尔尼菲篮状菌感染骨破坏中的诊断价值[J]. 分子影像学杂志, 2022, 45(1): 44-8. doi: 10.12122/j.issn.1674-4500.2022.01.09 [14] 张峰, 焦举, 谢良骏. 99mTc-MDP SPECT/CT全身骨显像诊断前列腺癌骨转移的临床价值[J]. 实用医学杂志, 2017, 33(11): 1774-7. https://www.cnki.com.cn/Article/CJFDTOTAL-SYYZ201711015.htm [15] Chan JF, Lau SK, Yuen KY, et al. Talaromyces (Penicillium) marneffei infection in non-HIV-infected patients[J]. Emerg Microbes Infect, 2016, 5(1): 1-9. [16] Qiu Y, Feng X, Zeng W, et al. Immunodeficiency disease spectrum in HIV-negative individuals with talaromycosis[J]. J Clin Immunol, 2021, 41(1): 221-3. doi: 10.1007/s10875-020-00869-5 [17] Cowman S, van Ingen J, Griffith DE, et al. Non-tuberculous mycobacterial pulmonary disease[J]. Eur Respir J, 2019, 54(1): 1900250. doi: 10.1183/13993003.00250-2019 [18] Qiu Y, Zhang JQ, Li BX, et al. Bacillus cereus isolated from a positive bone tissue culture in a patient with osteolysis and hightiter anti-interferon- γ autoantibodies: a case report[J]. Medicine, 2019, 98(43): e17609. doi: 10.1097/MD.0000000000017609 [19] 张永学, 高再荣. 核医学[M]. 3版. 北京: 科学出版社, 2016. [20] 张伟, 罗晓燕. SPECT/CT显像诊断转移性骨肿瘤的价值[J]. 中国卫生标准管理, 2021, 12(5): 80-2. https://www.cnki.com.cn/Article/CJFDTOTAL-WSBZ202105030.htm [21] 李占银, 阿尖措, 苏桂芳. SPECT/CT结合三相骨显像诊断假体周围感染的价值分析[J]. 中国CT和MRI杂志, 2019, 17(12): 139-42. https://www.cnki.com.cn/Article/CJFDTOTAL-CTMR201912044.htm [22] Hirsch R, Miller SM, Kazi S, et al. Human immunodeficiency virus-associated atypical mycobacterial skeletal infections[J]. Semin Arthritis Rheum, 1996, 25(5): 347-56. doi: 10.1016/S0049-0172(96)80020-5 [23] Nightingale SD, Byrd LT, Southern PM, et al. Incidence of Mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus- positive patients[J]. J Infect Dis, 1992, 165(6): 1082-5. doi: 10.1093/infdis/165.6.1082 [24] Patel SY, Ding L, Brown MR, et al. Anti-IFN-γ autoantibodies in disseminated nontuberculous mycobacterial infections [J]. J Immunol, 2005, 175(7): 4769-76. doi: 10.4049/jimmunol.175.7.4769 [25] Xu XL, Lao XJ, Zhang CL, et al. Chronic Mycobacterium avium skin and soft tissue infection complicated with scalp osteomyelitis possibly secondary to anti-interferon-γ autoantibody formation[J]. BMC Infect Dis, 2019, 19(1): 203. doi: 10.1186/s12879-019-3771-3 [26] Chi CY, Lin CH, Ho MW, et al. Clinical manifestations, course, and outcome of patients with neutralizing anti-interferon-γ autoantibodies and disseminated nontuberculous mycobacterial infections [J]. Medicine, 2016, 95(25): e3927. doi: 10.1097/MD.0000000000003927 [27] Takayanagi H, Ogasawara K, Hida S, et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross- talk between RANKL and IFN-gamma [J]. Nature, 2000, 408(6812): 600-5. doi: 10.1038/35046102 [28] Kelchtermans H, Billiau A, Matthys P. How interferon-gamma keeps autoimmune diseases in check [J]. Trends Immunol, 2008, 29 (10): 479-86. doi: 10.1016/j.it.2008.07.002 [29] Gao YH, Grassi F, Ryan MR, et al. IFN-gamma stimulates osteoclast formation and bone loss in vivo via antigen-driven T cell activation [J]. J Clin Invest, 2007, 117(1): 122-32. doi: 10.1172/JCI30074 [30] Krisnawati DI, Liu YC, Lee YJ, et al. Blockade effects of anti-interferon-(IFN-) γ autoantibodies on IFN-γ-regulated antimicrobial immunity [J]. J Immunol Res, 2019, 2019: 1629258. -







 下载:
下载: