Correlation between gray matter volume changes and clinical indicators in patients with primary dysmenorrhea based on voxel-based
-
摘要:
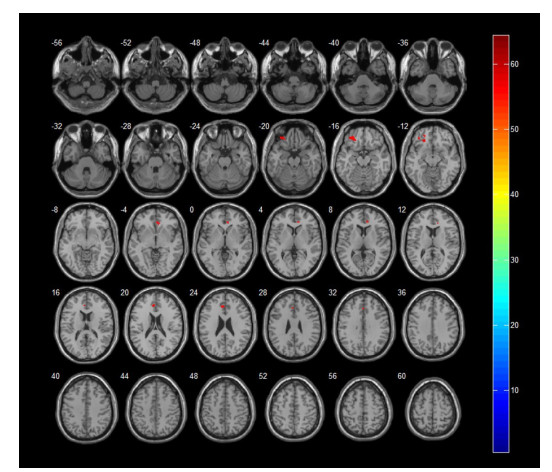
目的 基于体素形态学测量技术探究原发性痛经患者脑灰质体积改变及其与临床量表评分间的相关性。 方法 收集符合纳入标准的41例原发性痛经患者和41例健康对照者在月经来潮的第1~3天内进行高分辨率T1结构像扫描,并且填写痛经程度量表、COX痛经症状量表、焦虑自评量表、抑郁自评量表。采用SPM8软件在全脑水平比较灰质体积,观察两组灰质体积显著变化的脑区。 结果 原发性痛经患者较健康对照脑灰质体积缩小,差异脑区主要包括:左侧眶部额中回、左侧前扣带回、右侧前扣带回(P < 0.001),未发现灰质体积明显增加的脑区。痛经组和健康组在痛经程度量表、COX痛经症状量表、焦虑自评量表、抑郁自评量表评分的差异有统计学意义(P < 0.001)。左侧前扣带回与痛经程度量表、COX痛经症状量表、焦虑自评量表、抑郁自评量表评分存在负相关关系。 结论 基于体素形态学测量技术可以准确评估原发性痛经患者脑灰质微结构改变,这些灰质体积缩小的脑区主要涉及疼痛传递、处理及情绪调节相关的脑区,其灰质体积的变化在原发性痛经疼痛的发生及持续中可能有着重要作用。 Abstract:Objective To investigate voxel- based morphometry in primary dysmenorrhea patients with altered brain gray matter volume and the correlation between it and clinical scale scores. Methods Forty-one patients with primary dysmenorrhea and 41 healthy controls who met the inclusion criteria were collected to undergo high-resolution T1 structural imaging scans during days 1-3 of menstruation They completed the dysmenorrhea severity scale, the COX dysmenorrhea symptom scale, the anxiety self-assessment scale and the depression self-assessment scale. The SPM8 software was used to compare gray matter volumes at the whole brain level. We observed the brain areas with significant changes in gray matter volumes in both groups. Results The gray matter volume of the patients with primary dysmenorrhea was reduced compared with that of the healthy control group. The differences mainly included the left orbital middle frontal gyrus, left anterior cingulate gyrus and right anterior cingulate gyrus (P < 0.001), but no brain regions with a significant increase in gray matter volume were found. There were significant differences in dysmenorrhea severity scale, the COX dysmenorrhea symptom scale, the anxiety self-assessment scale and the depression self-assessment scale scores between the dysmenorrhea group and the healthy group (P < 0.001). Moreover, the left anterior cingulate gyrus was negatively correlated with the scores of dysmenorrhea severity scale, the COX dysmenorrhea symptom scale, the anxiety self-assessment scale and the depression selfassessment scale. Conclusion Based on voxel- based morphometry analysis, we can accurately assess the gray matter microstructural changes in the brain of patients with primary dysmenorrhea, and these brain regions with reduced gray matter volume are mainly involved in brain regions related to pain transmission, processing and emotion regulation. The changes in gray matter volume may play an important role in the occurrence and persistence of primary dysmenorrhea. -
Key words:
- primary dysmenorrhea /
- voxel-based morphometry /
- volume of gray matter
-
表 1 PDM组和健康对照组临床资料比较
Table 1. Comparison of clinical data between PDM group and healthy control group (n=41)
临床资料 健康组 PDM组 t/Z P 年龄(岁) 25(24, 26) 24(21, 26) -1.83 > 0.05 身高(cm) 160(158, 163.5) 162(160, 166.5) -0.49 > 0.05 体质量(kg) 52(49.5, 57) 52(46.5, 57.5) 0.37 > 0.05 病程(年) 0(0, 0) 06 2.5, 9) -8.34 < 0.001 VAS 1(0, 2) 5(4, 6) -7.85 < 0.001 CMSS总发作时间 5(2.5, 8.5) 21(1525) -6.90 < 0.001 CMSS平均严重程度 4(2, 9) 16(13, 26.5) -6.74 < 0.001 SAS 27(23, 30) 37(24, 44) -4.83 < 0.001 SDS 28(25, 34) 38(29, 45) -3.78 < 0.001 数据均以中位数(上下四分位数)表示. PDM: 原发性痛经; VAS: 痛视 觉模拟量表; CMSS: COX痛经症状量表; SAS: 焦虑自评量表; SDS: 抑郁自评量表 表 2 两组受试者脑灰质、白质、脑脊液以及全脑体积比较
Table 2. Comparison of gray matter, white matter, cerebrospinal fluid and whole brain volume between the two groups (cm3, n= 41, Mean±SD)
项目 健康组 PDM组 P 脑灰质总体积 637.07±42.32 649.81±41.38 0.172 脑白质总体积 502.40±40.25 498.84±82.36 0.804 脑脊液总体积 198.85±21.11 199.46±20.05 0.893 全脑体积 1338.31±82.51 1348.11±116.51 0.662 表 3 两组受试者灰质体积差异脑区分布
Table 3. Brain region distribution of gray matter volume difference between the two groups
脑区 体素数 最大激活点t值 MNI坐标 X Y Z 左侧眶部额中回 172 3.38 -33 39 -16.5 右侧前扣带回 137 3.07 10.5 34.5 -3 左侧前扣带回 142 3.31 -3 36 21 注: 表中为两组间灰质体积缩小的脑区(P < 0.001, 未校正, 体素>100). -
[1] ACOG Committee. ACOG committee opinion No. 760 summary: dysmenorrhea and endometriosis in the adolescent[J]. Obstet Gynecol, 2018, 132(6): 1517-8. doi: 10.1097/AOG.0000000000002981 [2] Kho KA, Shields JK. Diagnosis and management of primary dysmenorrhea[J]. JAMA, 2020, 323(3): 268-9. doi: 10.1001/jama.2019.16921 [3] Yang MX, Chen XZ, Bo LN, et al. Moxibustion for pain relief in patients with primary dysmenorrhea: a randomized controlled trial [J]. PLoS One, 2017, 12(2): e0170952. doi: 10.1371/journal.pone.0170952 [4] Zhang YN, Huo JW, Huang YR, et al. Altered amplitude of lowfrequency fluctuation and regional cerebral blood flow in females with primary dysmenorrhea: a resting-state fMRI and arterial spin labeling study[J]. J Pain Res, 2019, 12: 1243-50. doi: 10.2147/JPR.S177502 [5] Jin LM, Yang XJ, Liu P, et al. Dynamic abnormalities of spontaneous brain activity in women with primary dysmenorrhea [J]. J Pain Res, 2017, 10: 699-707. doi: 10.2147/JPR.S121286 [6] Liu P, Yang J, Wang G, et al. Altered regional cortical thickness and subcortical volume in women with primary dysmenorrhoea[J]. Eur J Pain, 2016, 20(4): 512-20. doi: 10.1002/ejp.753 [7] Seifert F, Maihöfner C. Functional and structural imaging of paininduced neuroplasticity[J]. Curr Opin Anaesthesiol, 2011, 24(5): 515-23. doi: 10.1097/ACO.0b013e32834a1079 [8] 王金辉, 刘波, 喻大华, 等. 前庭性偏头痛患者基于体素的脑灰质体积研究[J]. 磁共振成像, 2021, 12(3): 67-70, 88. https://www.cnki.com.cn/Article/CJFDTOTAL-CGZC202103016.htm [9] Cauda F, et al. Gray matter alterations in chronic pain: a networkoriented meta-analytic approach[J]. Neuroimage Clin, 2014, 4: 676-86. [10] Gündel H. Altered cerebral response to noxious heat stimulation in patients with somatoform pain disorder[J]. PAIN®, 2008, 137(2): 413-21. [11] 王亚琦, 张淑娴, 徐芹艳, 等. 紧张型头痛患者灰质体积改变及与疼痛程度评分的相关性研究[J]. 临床放射学杂志, 2020, 39(11): 2161-5. https://www.cnki.com.cn/Article/CJFDTOTAL-LCFS202011007.htm [12] Ong WY, Stohler CS, Herr DR. Role of the prefrontal cortex in pain processing[J]. Mol Neurobiol, 2019, 56(2): 1137-66. [13] May A. Structural brain imaging: a window into chronic pain[J]. Neuroscientist, 2011, 17(2): 209-20. [14] Xiao X, Ding M, Zhang YQ. Role of the anterior cingulate cortex in translational pain research[J]. Neurosci Bull, 2021, 37(3): 405-22. [15] Xiao X, et al. A new perspective on the anterior cingulate cortex and affective pain[J]. Neurosci Biobehav Rev, 2018, 90: 200-11. [16] Liu P, Liu YF, Wang GL, et al. Changes of functional connectivity of the anterior cingulate cortex in women with primary dysmenorrhea[J]. Brain Imaging Behav, 2018, 12(3): 710-7. [17] Liu P, Liu YF, Wang GL, et al. Aberrant default mode network in patients with primary dysmenorrhea: a fMRI study[J]. Brain Imaging Behav, 2017, 11(5): 1479-85. [18] Wei SY, Chao HT, Tu CH, et al. Changes in functional connectivity of pain modulatory systems in women with primary dysmenorrhea [J]. Pain, 2016, 157(1): 92-102. [19] Zhao RH, Zhou H, Huang LY, et al. Neuropathic pain causes pyramidal neuronal hyperactivity in the anterior cingulate cortex [J]. Front Cell Neurosci, 2018, 12: 107. [20] Bushnell MC, Čeko M, Low LA. Cognitive and emotional control of pain and its disruption in chronic pain[J]. Nat Rev Neurosci, 2013, 14(7): 502-11. [21] Bliss TVP, Collingridge GL, Kaang BK, et al. Synaptic plasticity in the anterior cingulate cortex in acute and chronic pain[J]. Nat Rev Neurosci, 2016, 17(8): 485-96. [22] Barthas F, et al. The anterior cingulate cortex is a critical hub for pain-induced depression[J]. Biol Psychiatry, 2015, 77(3): 236-45. [23] Sellmeijer J, Mathis V, Hugel S, et al. Hyperactivity of anterior cingulate cortex areas 24a/24b drives chronic pain- induced anxiodepressive-like consequences[J]. J Neurosci, 2018, 38(12): 3102-15. [24] Michaelides A, Zis P. Depression, anxiety and acute pain: links and management challenges[J]. Postgrad Med, 2019, 131(7): 438-44. -







 下载:
下载: