Evaluation of colorectal cancer metastasis by molecular visualization imaging based on cytoskeleton
-
摘要:
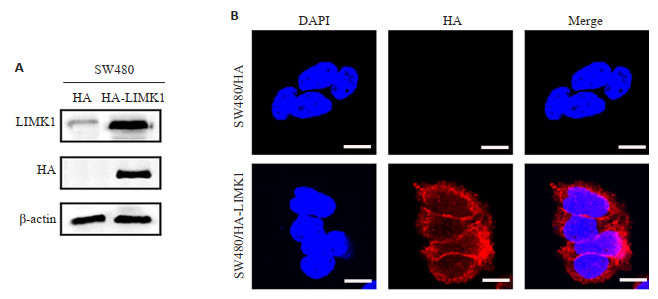
目的 基于细胞骨架的分子可视化成像技术阐明LIM蛋白1(LIMK1)诱导结直肠转移的具体机制。 方法 构建过表达LIMK1处理组细胞SW480/HA-LIMK1及空白对照组细胞SW480/HA,鬼笔环肽染色法分子可视化成像技术检测过表达LIMK1对SW480细胞骨架蛋白的影响;沉默LIMK1、ACTN4和MYH9,鬼笔环肽染色法分子可视化成像技术检测其对细胞骨架的影响,Transwell迁移实验检测沉默ACTN4和MYH9对LIMK1介导的细胞迁移的影响。 结果 在SW480细胞系中,过表达LIMK1后细胞骨架蛋白F-肌动蛋白染色明显增强,而使用siRNA沉默ACTIN4或MYH9的表达后,这种增加可以被恢复。此外,在SW480细胞系中沉默这两种蛋白,可以抑制LIMK1介导的恶性迁移运动表型(P<0.05)。 结论 LIMK1可能通过与ACTIN4或MYH9相互作用来调节细胞骨架,促进结直肠癌细胞的迁移和侵袭。 Abstract:Objective To elucidate the specific mechanism of colorectal metastasis induced by LIMK1 based on molecular visualization imaging technique of cytoskeleton. Methods The effects of overexpressed LIMK1 on the cytoskeletal of SW480 cells were detected by molecular visualization imaging technique, and the effects of silencing LIMK1, ACTN4 and MYH9 on the cytoskeleton were detected by Phalloidin molecular visualization imaging technique. Transwell migration assay was used to detect the effects of silenced ACTN4 and MYH9 on LIMK1-mediated cell migration. Results In SW480 cell line, the staining of cytoskeleton protein F-actin was significantly enhanced after overexpression of LIMK1, but this increase could be restored after silencing the expression of ACTIN4 or MYH9 with siRNA. In addition, silencing these two proteins in SW480 cell lines inhibited the phenotype of LIMK1-mediated malignant migration(P<0.05). Conclusion LIMK1 may regulate the cytoskeleton and promote the migration and invasion of colorectal cancer cells by interacting with ACTIN4 or MYH9. -
Key words:
- colorectal cancer /
- LIMK1 /
- ACTIN4 /
- MYH9 /
- cytoskeleton
-
表 1 用于转染的siRNA序列
Table 1. siRNA sequences for transfection
基因名称 Sense 5' to 3' Antisense 5' to 3' LIMK1 CGCACCAGUACUAUGAGAATT UUCUCAUAGUACUGGUGCGTT ACTN4 GACCAGAGCUGAUUGAGUATT UACUCAAUCAGCUCUGGUCTT MYH9 GGAGCGGAACACUGACCAGTT CUGGUCAGUGUUCCGCUCCTT 阴性对照 UUCUCCGAACGUGUCACGUTT ACGUGACACGUUCGGAGAATT -
[1] Siegel RL, Miller KD, Goding Sauer A, et al. Colorectal cancer statistics, 2020[J]. CAA Cancer J Clin, 2020, 70(3): 145-64. doi: 10.3322/caac.21601 [2] Fife CM, McCarroll JA, Kavallaris M. Movers and shakers: cell cytoskeleton in cancer metastasis[J]. Br J Pharmacol, 2014, 171(24): 5507-23. doi: 10.1111/bph.12704 [3] Mori T, Okano I, Mizuno K, et al. Comparison of tissue distribution of two novel serine/threonine kinase genes containing the LIM motif (LIMK-1 and LIMK-2) in the developing rat[J]. Mol Brain Res, 1997, 45(2): 247-54. doi: 10.1016/S0169-328X(96)00257-4 [4] Nagata K, Ohashi K, Yang N, et al. The N-terminal LIM domain negatively regulates the kinase activity of LIM-kinase 1[J]. Biochem J, 1999, 343(Pt 1): 99-105. [5] Ohashi K, Hosoya T, Takahashi K, et al. A Drosophila homolog of LIM-kinase phosphorylates cofilin and induces actin cytoskeletal reorganization[J]. Biochem Biophys Res Commun, 2000, 276(3): 1178-85. doi: 10.1006/bbrc.2000.3599 [6] Nishimura Y, Yoshioka K, Bernard O, et al. A role of LIM kinase 1/cofilin pathway in regulating endocytic trafficking of EGF receptor in human breast cancer cells[J]. Histochem Cell Biol, 2006, 126(5): 627-38. doi: 10.1007/s00418-006-0198-x [7] Ishaq M, Lin BR, Bosche M, et al. LIM kinase 1-dependent cofilin 1 pathway and actin dynamics mediate nuclear retinoid receptor function in T lymphocytes[J]. BMC Mol Biol, 2011, 12: 41. doi: 10.1186/1471-2199-12-41 [8] Bagheri-Yarmand R, Mazumdar A, Sahin AA, et al. LIM kinase 1 increases tumor metastasis of human breast cancer cells via regulation of the urokinase-type plasminogen activator system[J]. Int J Cancer, 2006, 118(11): 2703-10. doi: 10.1002/ijc.21650 [9] Davila M, Frost AR, Grizzle WE, et al. LIM kinase 1 is essential for the invasive growth of prostate epithelial cells[J]. J Biol Chem, 2003, 278(38): 36868-75. doi: 10.1074/jbc.M306196200 [10] Liao Q, Li R, Zhou R, et al. LIM kinase 1 interacts with myosin-9 and alpha-actinin-4 and promotes colorectal cancer progression[J]. Br J Cancer, 2017, 117(4): 563-71. doi: 10.1038/bjc.2017.193 [11] Nunoue K, Ohashi K, Okano I, et al. LIMK-1 and LIMK-2, two members of a LIM motif-containing protein kinase family[J]. Oncogene, 1995, 11(4): 701-10. [12] Scott RW, Olson MF. LIM kinases: function, regulation and association with human disease[J]. J Mol Med, 2007, 85(6): 555-68. doi: 10.1007/s00109-007-0165-6 [13] Honda K, Yamada T, Endo R, et al. Actinin-4, a novel actinbundling protein associated with cell motility and cancer invasion[J]. J Cell Biol, 1998, 140(6): 1383-93. doi: 10.1083/jcb.140.6.1383 [14] Shao HS, Li SY, Watkins SC, et al. α-actinin-4 is required for amoeboid-type invasiveness of melanoma cells[J]. J Biol Chem, 2014, 289(47): 32717-28. doi: 10.1074/jbc.M114.579185 [15] Honda K, Yamada T, Hayashida Y, et al. Actinin-4 increases cell motility and promotes lymph node metastasis of colorectal cancer[J]. Gastroenterology, 2005, 128(1): 51-62. doi: 10.1053/j.gastro.2004.10.004 [16] Simons M, Wang M, McBride OW, et al. Human nonmuscle myosin heavy chains are encoded by two genes located on different chromosomes[J]. Circ Res, 1991, 69(2): 530-9. doi: 10.1161/01.RES.69.2.530 [17] Katono K, Sato Y, Jiang SX, et al. Prognostic significance of MYH9 expression in resected non-small cell lung cancer[J]. PLoS One, 2015, 10(3): e0121460. doi: 10.1371/journal.pone.0121460 [18] Zhou W, Fan MY, Wei YX, et al. The expression of MYH9 in osteosarcoma and its effect on the migration and invasion abilities of tumor cell[J]. Asian Pac J Trop Med, 2016, 9(6): 597-600. doi: 10.1016/j.apjtm.2016.04.011 [19] Yu MX, Wang JH, Zhu ZJ, et al. Prognostic impact of MYH9 expression on patients with acute myeloid leukemia[J]. Oncotarget, 2017, 8(1): 156-63. doi: 10.18632/oncotarget.10613 [20] Schramek D, Sendoel A, Segal JP, et al. Direct in vivo RNAi screen unveils myosin IIa as a tumor suppressor of squamous cell carcinomas[J]. Science, 2014, 343(6168): 309-13. doi: 10.1126/science.1248627 -







 下载:
下载: