Application of magnetic susceptibility weighted imaging in differential diagnosis, preoperative pathological grading and surgical guidance of glioma
-
摘要:
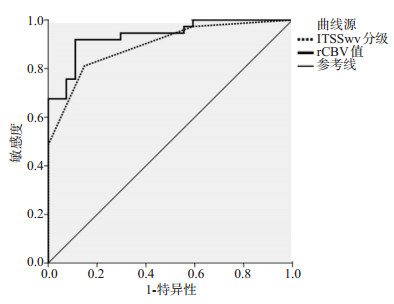
目的 探讨磁敏感加权成像(SWI)在脑胶质瘤鉴别诊断、术前病理分级及手术指导中的应用。 方法 回顾性分析新疆维吾尔自治区人民医院2018年10月~2021年10月经手术及组织病理学检查证实为脑胶质瘤(n=64)、单发脑转移瘤(n=15)和颅内淋巴瘤(n=15)的患者的临床资料。以病理结果作为金标准,将脑胶质瘤患者按照世界卫生组织肿瘤分类标准分为低级别组(n= 27)和高级别组(n=37),所有患者均通过SWI检测,进行瘤内磁敏感信号强度(ITSS)分级评估以及肿瘤实质区与瘤周水肿区相对脑血容量(rCBV)值测定,比较不同肿瘤类型差异,并通过ROC曲线评估其在高级别与低级别脑胶质瘤中的鉴别价值。 结果 高级别脑胶质瘤与单发脑转移瘤ITSS分级、肿瘤实质区rCBV值的差异无统计学意义(P > 0.05);高级别脑胶质瘤周水肿区rCBV值高于单发脑转移瘤(P < 0.05);高级别脑胶质瘤ITSS分级程度低于淋巴瘤,血管评分高于淋巴瘤(P < 0.05);高级别胶质瘤ITSS分级高于低级别胶质瘤,肿瘤实质区rCBV值高于低级别胶质瘤(P < 0.05);Spearman相关性分析结果显示,胶质瘤分级与ITSS分级、rCBV值呈正相关关系(r=0.728、0.851,P < 0.05);ITSS分级评估脑胶质瘤高级别ROC曲线下面积为0.894,敏感度、特异性、准确度分别为81.08%、85.19%、82.81%;rCBV值评估脑胶质瘤高级别ROC曲线下面积为0.937,敏感度、特异性、准确度分别为91.89%、88.89%、90.63%;64例脑胶质瘤患者,和常规序列相比,SWI序列对于肿瘤边界的显示更为清晰,同时可呈现肿瘤微小血管病变信息,胶质瘤肿瘤实质区rCBV值均高于瘤周水肿区rCBV值(P < 0.05)。 结论 SWI有助于高级别胶质瘤的鉴别诊断,对于胶质瘤低级别与高级别的评估效能较高,可在一定程度上指导术中脑胶质瘤切除。 Abstract:Objective To investigate the application of susceptibility weighted imaging (SWI) in the differential diagnosis, preoperative pathological grading and surgical guidance of glioma. Methods The clinical data of 64 patients with glioma, 15 patients with single brain metastases and 15 patients with intracranial lymphoma confirmed by surgery and histopathological examination in the People's Hospital of Xinjiang Uygur Autonomous Region from October 2018 to October 2021 were retrospectively analyzed. The pathological results were used as the gold standard. The patients with glioma were divided into low-grade group (n=27) and high-grade group (n=37) according to the WHO tumor classification criteria. All patients were detected by SWI, and the intratumoral susceptibility signal intensity (ITSS) grading evaluation and the relative cerebral blood volume (rCBV) value of the tumor parenchymal area and the peritumoral edema area were measured. The differences of different tumor types were compared, and the ROC curve was used to evaluate its differential value between high-grade and low-grade gliomas. Results Comparison of ITSS grade and rCBV value in tumor parenchyma between high-grade gliomas and solitary metastases (P > 0.05); the rCBV value in peritumoral edema area of high-grade glioma was higher than that of single brain metastasis (P < 0.05). The ITSS grade of high-grade glioma was lower than that of lymphoma, and the vascular score was higher than that of lymphoma (P < 0.05). ITSS grade of high grade glioma was higher than that of low grade glioma, rCBV value of tumor parenchyma was higher than that of low grade glioma (P < 0.05). Spearman correlation analysis showed that glioma grade was positively correlated with ITSS grade and rCBV value (r=0.728, 0.851, P < 0.005). The area under the high-level ROC curve of ITSS grading evaluation of glioma was 0.894, and the sensitivity, specificity and accuracy were 81.08%, 85.19% and 82.81%, respectively. The area under the ROC curve of rCBV was 0.937, and the sensitivity, specificity and accuracy were 91.89%, 88.89 % and 90.63%, respectively. Compared with conventional sequence, SWI sequence showed tumor boundary more clearly and showed tumor microangiopathy information in 64 patients with glioma. The rCBV values in glioma parenchyma were higher than those in peritumoral edema (P < 0.05). Conclusion SWI is helpful for the differential diagnosis of high-grade gliomas, and has high evaluation efficiency for low-grade and high-grade gliomas, which can guide intraoperative glioma resection to a certain extent. -
表 1 高级别脑胶质瘤与单发脑转移瘤ITSS分级、肿瘤实质区rCBV值比较
Table 1. Comparison of ITSS grade and rCBV value in tumor parenchyma between high-grade glioma and single brain metastasis
肿瘤类别 ITSS分级(n) 肿瘤实质区rCBV(Mean±SD) 瘤周水肿区rCBV(Mean±SD) 0级 Ⅰ级 Ⅱ级 Ⅲ级 高级别脑胶质瘤(n=37) 1 5 12 19 4.620±1.009 1.914±0.405 单发脑转移瘤(n=15) 1 1 2 11 3.998±1.120 0.817±0.206 Z/t 1.195 1.952 9.941 P 0.232 0.057 <0.001 ITSS: 瘤内磁敏感信号强度; rCBV: 相对脑血容量. 表 2 高级别脑胶质瘤与淋巴瘤ITSS分级、出血率、血管结构半定量评分比较
Table 2. Comparison of high-grade glioma and lymphoma in ITSS grade, bleeding rate and semi-quantitative score of vascular structure (n)
肿瘤类别 ITSS分级 血管评分 0级 Ⅰ级 Ⅱ级 Ⅲ级 0分 1分 2分 3分 高级别脑胶质瘤(n=37) 1 5 12 19 0 1 5 31 淋巴瘤(n=15) 0 1 1 13 4 8 2 1 Z/t 2.216 5.668 P 0.027 < 0.001 表 3 高、低级别胶质瘤ITSS分级与肿瘤实质区rCBV值比较
Table 3. Comparison of ITSS grade and rCBV value in tumor parenchyma of high and low grade gliomas
病理分级 ITSS分级(n) 肿瘤实质区rCBV值(Mean±SD) 0级 Ⅰ级 Ⅱ级 Ⅲ级 低级别 Ⅰ级(n=8) 7 1 0 0 1.179±0.238 Ⅱ级(n=19) 4 11 4 0 1.815±0.405 高级别 Ⅲ级(n=22) 1 4 9 8 4.387±1.005 Ⅳ级(n=15) 0 2 3 10 4.961±1.114 Z/t 5.876 13.900 P <0.001 <0.001 表 4 ITSS分级与rCBV值对脑胶质瘤分级的诊断评估价值
Table 4. Diagnostic value of ITSS grading and rCBV value for brain glioma grading
时间 临界值 AUC 95% CI 敏感度(%) 特异性(%) 准确度(%) P ITSS分级 Ⅱ级 0.894 0.818~0.970 81.08 85.19 82.81 <0.001 rCBV值 2.940 0.937 0.880~0.994 91.89 88.89 90.63 <0.001 表 5 脑胶质瘤肿瘤实质区、瘤周水肿区rCBV值比较
Table 5. Comparison of rCBV values in tumor parenchyma and peritumoral edema of glioma (Mean±SD)
病理分级 肿瘤实质区rCBV 瘤周水肿区rCBV t P 低级别(n=27) 1.627±0.367 0.510±0.124 14.282 <0.001 高级别(n=37) 4.620±1.072 1.914±0.405 14.133 <0.001 t 13.900 17.395 P <0.001 <0.001 -
[1] 王伟, 沈龙山, 程雯, 等. 多模态磁共振成像在脑胶质瘤分级诊断及预后评估中的研究进展[J]. 分子影像学杂志, 2022, 45(5): 795-9. doi: 10.12122/j.issn.1674-4500.2022.05.31 [2] 莫家鹏, 刘群会, 章慧, 等. miR-671-3p靶向调控CKAP4对人脑胶质瘤细胞增殖及迁移的影响[J]. 中南医学科学杂志, 2022, 50(3): 341-4, 353. https://www.cnki.com.cn/Article/CJFDTOTAL-HYYY202203007.htm [3] 李欣, 谢继承, 王静, 等. 磁共振MRS、DWI及SWI序列在脑胶质瘤分级诊断中的应用价值[J]. 中华全科医学, 2022, 20(9): 1541-4. https://www.cnki.com.cn/Article/CJFDTOTAL-SYQY202209026.htm [4] Li GZ, Li L, Li YM, et al. An MRI radiomics approach to predict survival and tumour-infiltrating macrophages in gliomas[J]. Brain, 2022, 145(3): 1151-61. doi: 10.1093/brain/awab340 [5] 田东旭. 磁敏感加权成像及3D-ASL在各级脑胶质瘤分级中的效果[J]. 中国医疗器械信息, 2022, 28(13): 59-61. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGQX202213008.htm [6] Jiang RF, Hu XM, Deng KJ, et al. Neurite orientation dispersion and density imaging in evaluation of high-grade glioma-induced corticospinal tract injury[J]. Eur J Radiol, 2021, 140: 109750. doi: 10.1016/j.ejrad.2021.109750 [7] 胡宁芮, 李传亭. 磁共振新技术鉴别脑胶质瘤真假性进展及放射性损伤的研究进展[J]. 医学影像学杂志, 2022, 32(4): 685-7. https://www.cnki.com.cn/Article/CJFDTOTAL-XYXZ202204032.htm [8] Haller S, Haacke EM, Thurnher MM, et al. Susceptibility-weighted imaging: technical essentials and clinical neurologic applications[J]. Radiology, 2021, 299(1): 3-26. doi: 10.1148/radiol.2021203071 [9] 崔振华. 磁敏感加权成像检查+氢质子磁共振波谱成像对脑胶质瘤分级诊断效能的影响[J]. 黑龙江医学, 2022, 46(6): 743-4, 747. https://www.cnki.com.cn/Article/CJFDTOTAL-HLYX202206024.htm [10] 梅邹, 毕俊英. 三种MRI灌注技术术前评估脑胶质瘤分级的价值对比[J]. 磁共振成像, 2022, 13(2): 83-6, 95. https://www.cnki.com.cn/Article/CJFDTOTAL-CGZC202202017.htm [11] Louis DN, Ohgaki H, Wiestler OD, et al. The 2007 WHO classification of tumours of the central nervous system[J]. Acta Neuropathol, 2007, 114(2): 97-109. [12] Park MJ, Kim HS, Jahng GH, et al. Semiquantitative assessment of intratumoral susceptibility signals using non-contrast-enhanced high-field high-resolution susceptibility-weighted imaging in patients with gliomas: comparison with MR perfusion imaging[J]. AJNR Am J Neuroradiol, 2009, 30(7): 1402-8. [13] 张庆浩, 段文超, 刘献志, 等. 无症状WHOⅡ级脑胶质瘤的疾病特征和术后生存分析[J]. 中华神经外科杂志, 2020, 36(4): 405-9. [14] 沈烨, 谢红, 恽健, 等. 探讨磁共振SWI序列对脑胶质瘤术前分级的应用价值[J]. 中国CT和MRI杂志, 2020, 18(8): 13-5, 59. https://www.cnki.com.cn/Article/CJFDTOTAL-CTMR202008005.htm [15] Gaudino S, Marziali G, Pezzullo G, et al. Role of susceptibility-weighted imaging and intratumoral susceptibility signals in grading and differentiating pediatric brain tumors at 1.5 T: a preliminary study[J]. Neuroradiology, 2020, 62(6): 705-13. [16] 王良东. DWI与SWI对脑胶质瘤患者分级诊断效能的影响[J]. 医学理论与实践, 2019, 32(6): 884-5. https://www.cnki.com.cn/Article/CJFDTOTAL-YXLL201906058.htm [17] Cluceru J, Interian Y, Phillips JJ, et al. Improving the noninvasive classification of glioma genetic subtype with deep learning and diffusion-weighted imaging[J]. Neuro-oncology, 2022, 24(4): 639-52. [18] 鲍燕, 陈国平, 丁朝鹏. DWI联合SWI检查在不同级别脑胶质瘤诊断中的应用价值[J]. 河南医学研究, 2020, 29(2): 329-30. https://www.cnki.com.cn/Article/CJFDTOTAL-HNYX202002073.htm [19] 张晓星, 李跃华. 磁共振技术在脑胶质瘤术前分级中的应用进展[J]. 中国医学计算机成像杂志, 2019, 25(3): 326-31. https://www.cnki.com.cn/Article/CJFDTOTAL-YJTY201903026.htm [20] 王林魁, 温琥玲, 杨凡慧. SWI对急性缺血性脑卒中出血性转化的检测及其对预后的影响[J]. 川北医学院学报, 2021, 36(8): 968-70, 998. https://www.cnki.com.cn/Article/CJFDTOTAL-NOTH202108004.htm [21] 刘海, 陈士新, 付伟, 等. DWI、DTI联合常规MRI可提高高级别脑胶质瘤的诊断准确度[J]. 分子影像学杂志, 2021, 44(3): 546-51. doi: 10.12122/j.issn.1674-4500.2021.03.26 [22] 杜常月, 苗娜, 齐旭红, 等. 磁敏感加权成像及3D-ASL在各级脑胶质瘤分级中的价值[J]. 磁共振成像, 2019, 10(9): 645-9. https://www.cnki.com.cn/Article/CJFDTOTAL-CGZC201909003.htm [23] Xi YB, Kang XW, Wang N, et al. Differentiation of primary central nervous system lymphoma from high-grade glioma and brain metastasis using arterial spin labeling and dynamic contrast-enhanced magnetic resonance imaging[J]. Eur J Radiol, 2019, 112: 59-64. [24] 曹中华. DCE-MRI联合SWI对脑胶质瘤分级诊断效能的影响[J]. 深圳中西医结合杂志, 2019, 29(14): 81-2. https://www.cnki.com.cn/Article/CJFDTOTAL-SZZX201914040.htm [25] Ikeda K, Kolakshyapati M, Takayasu T, et al. Diffusion-weighted imaging-gadolinium enhancement mismatch sign in diffuse midline glioma[J]. Eur J Radiol, 2022, 147: 110103. [26] 孔令伟, 李守巍. 磁敏感加权成像在脑胶质瘤诊断和治疗中的应用进展[J]. 中华神经外科杂志, 2020, 36(2): 214-6. -







 下载:
下载: