Clinical significance of multimodal contrast ultrasonography for evaluating the efficacy of radiotherapy and chemotherapy in patients with radical mastectomy
-
摘要:
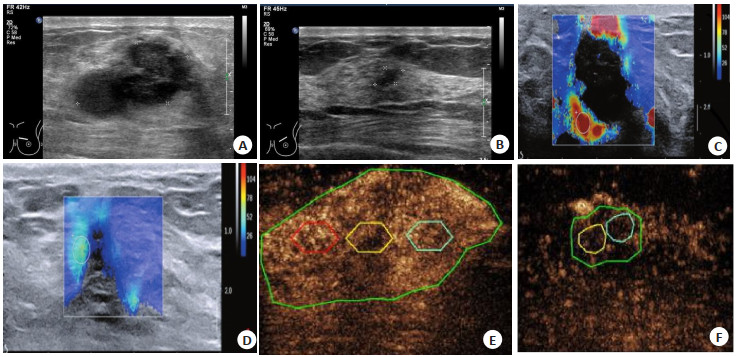
目的 探究多模态超声造影技术对乳腺根治术患者放化疗疗效评估的临床意义。 方法 回顾性分析2015年7月~2019年1月我院收治的140例乳腺癌放化疗患者临床资料,患者在化疗前后行超声造影、弹性超声检查,以术后病理作为金标准,分析超声造影对放化疗疗效的评估价值。 结果 常规超声评估放化疗疗效的敏感度为52.63%,特异性为87.50%,准确度68.57%,经Kappa检验常规造影与病理检测一致性较差(Kappa=0.388)。超声造影评估化疗疗效的敏感度为94.7%,特异性为98.4%,准确度为96.4%;超声造影与病理的一致性分析显示Kappa=0.928,一致性较好。放化疗前所有患者肿瘤超声图像表现为高增强,放化疗后有效患者主要表现为低增强,无效患者主要表现为中、高增强;无效患者化疗前多见灌注区缺损和放射血管,而在有效患者中少见;放化疗后有效患者区域血流量、时间-强度的曲线下面积和弹性应变率比值均低于无效患者,弹性评分、达峰时间高于无效患者,差异具有统计学意义(P < 0.05)。 结论 多模态超声造影技术的部分定性和定量指标对乳腺根治术患者放化疗疗效具有一定评估价值。 Abstract:Objective To investigate the clinical significance of multimodal contrast ultrasonography for evaluating the efficacy of radiotherapy and chemotherapy in patients with radical mastectomy. Methods The clinical data of 140 patients with breast cancer undergoing radiotherapy and chemotherapy admitted to our hospital from July 2015 to January 2019 was retrospectively analyzed. The patients underwent contrast ultrasonography and ultrasonic elasticity-before and after chemotherapy. The postoperative pathology was used as the gold standard. The diagnostic value of the ultrasound contrast in radiotherapy and chemotherapy was analyzed. Results The curative effect of radiotherapy and chemotherapy evaluated by conventional ultrasound was 52.63%, the specificity was 87.50%, and the accuracy was 68.57%. The consistency between conventional radiography and pathology was poor by Kappa test (Kappa=0.388). The sensitivity, specificity and accuracy of contrast-enhanced ultrasound in evaluating chemotherapy efficacy were 94.7%, 98.4% and 96.4% respectively; The consistency analysis of contrast- enhanced ultrasound and pathology showed that Kappa=0.928, with good consistency. The ultrasound images of all patients showed high enhancement before radiotherapy and chemotherapy, while the effective patients after radiotherapy and chemotherapy showed low enhancement, the ineffective patients showed moderate and high enhancement (P < 0.05). After chemoradiotherapy, the regional blood flow, area under curve and strain ratio in effective patientswere were lower than those in ineffective patients, whereas the time to peak and elasticity scorewerehigher than the ineffective patients (P < 0.05). Conclusion Partial qualitative and quantitative indicators of multimodal contrast ultrasonography have some certain evaluation value for the efficacy of radiotherapy and chemotherapy in patients undergoing radical mastectomy. -
表 1 常规超声与术后病理评估放化疗效果比较
Table 1. Comparison between conventional ultrasound and postoperative pathological evaluation of radiochemotherapy effect (n)
常规超声评估 病理检测 合计 有效 无效 有效 40 8 48 无效 36 56 92 合计 76 64 140 表 2 不同病理结果的多模态超声造影参数比较
Table 2. Comparison of multimodal contrast- enhanced ultrasound parameters with different pathological results (Mean±SD)
参数 病理检查 t P 有效(n=76) 无效(n=64) PI(dB) 6.22±0.41 6.31±0.55 1.108 0.27 TTP(s) 13.74±0.52 10.20±0.65 35.793 0.001 MTT(s) 36.16±5.13 35.92±5.09 0.277 0.783 AUC 85.12±10.53 136.56±22.61 17.694 < 0.001 RBV(mL) 2732.45±451.60 3546.41±361.30 11.621 < 0.001 弹性评分 3.51±0.73 2.61±0.78 2.315 < 0.001 SR 1.40±0.32 2.17±0.45 3.846 < 0.001 PI: 峰值强度; TTP: 达峰时间; MTT: 平均渡越时间; AUC: 曲线下面积; RBV: 区域血流量; SR: 应变率比值. 表 3 超声造影与术后病理评估放化疗效果比较
Table 3. Comparison of contrast-enhanced ultrasound and postoperative pathology in evaluating the effect of radiotherapy and chemotherapy (n)
超声造影评估 病理检测 合计 有效 无效 有效 72 1 73 无效 4 63 67 合计 76 64 140 -
[1] 高伟, 周阳. 超声造影在乳腺癌诊断及新辅助化疗疗效评估中的应用价值[J]. 解放军预防医学杂志, 2017, 35(2): 168-70. https://www.cnki.com.cn/Article/CJFDTOTAL-JYYX201702024.htm [2] 杨月杰. 超声造影技术在乳腺癌新辅助化疗疗效评估中的应用分析[J]. 山西医药杂志, 2019, 48(2): 166-7. doi: 10.3969/j.issn.0253-9926.2019.02.011 [3] Song L, Li L, Liu B, et al. Diagnostic evaluations of ultrasound and magnetic resonance imaging in mammary duct ectasia and breast cancer [J]. Oncol Lett, 2018, 15(2): 1698-706. [4] 万财凤, 王琳, 刘雪松, 等. 超声造影评估乳腺癌新辅助化疗疗效的临床价值[J]. 临床超声医学杂志, 2017, 19(10): 652-5. doi: 10.3969/j.issn.1008-6978.2017.10.002 [5] 《乳腺癌HER检测指南》编写组. 乳腺癌HER2检测指南(2014版)[J]. 中华病理学杂志, 2014, 43(4): 262-7. doi: 10.3760/cma.j.issn.0529-5807.2014.04.012 [6] 蒋朝霞, 彭卫军, 许玲辉, 等. RECIST标准在乳腺癌转移治疗疗效评价中的应用与WHO标准比较[J]. 中国癌症杂志, 2008, 18(2): 106-9. doi: 10.3969/j.issn.1007-3639.2008.02.006 [7] 郭轩, 唐甜甜, 王新乐, 等. 超声光散射成像技术对乳腺癌新辅助化疗疗效早期评价的研究[J]. 中华超声影像学杂志, 2018, 27(2): 160-3. doi: 10.3760/cma.j.issn.1004-4477.2018.02.014 [8] 刘立卷, 姜晓聪, 张高高. 氢质子磁共振波谱与细针穿刺活检在乳腺癌诊断中的价值比较[J]. 肿瘤基础与临床, 2017, 30(6): 512-4. doi: 10.3969/j.issn.1673-5412.2017.06.015 [9] Kobayashi T, Hayashi M, Arai M. Current status of ultrasonic tissue characterization in breast cancer [J]. J UOEH, 1984, 6(4): 397-410. doi: 10.7888/juoeh.6.397 [10] 贾志莺, 张连花, 马富成. 超声联合技术在乳腺癌新辅助化疗疗效评价中的应用价值[J]. 医学影像学杂志, 2017, 27(9): 1723-5. https://www.cnki.com.cn/Article/CJFDTOTAL-XYXZ201709030.htm [11] 汤晓艳. 超声造影在肝癌介入治疗中的应用价值[J]. 实用癌症杂志, 2018, 33(5): 781-3. doi: 10.3969/j.issn.1001-5930.2018.05.025 [12] Fukui K, Masumoto N, Shiroma N, et al. Novel tumor- infiltrating lymphocytes ultrasonography score based on ultrasonic tissue findings predicts tumor-infiltrating lymphocytes in breast cancer[J]. Breast Cancer, 2019, 26(5): 573-80. [13] 梁永平, 刘景萍, 王海霞, 等. 超声造影对乳腺癌肝转移小结节的诊断价值[J]. 天津医药, 2017, 45(1): 91-4. https://www.cnki.com.cn/Article/CJFDTOTAL-TJYZ201701024.htm [14] 郭凤娟, 徐金锋, 刘莹莹, 等. 靶向超声造影在乳腺癌诊断与治疗中的研究进展[J]. 中国医学影像技术, 2019, 35(3): 459-62. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYXX201903052.htm [15] Shanmugam S, Govindasamy G, Hussain SA, et al. Axillary dissection for breast cancer using electrocautery versus ultrasonic dissectors: a prospective randomized study[J]. Indian J Cancer, 2017, 54(3): 543-6. [16] 林周谊, 张群霞, 冉海涛. 超声联合超声造影诊断乳腺癌转移性前哨淋巴结的应用价值[J]. 中国超声医学杂志, 2018, 34(5): 407-9. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGCY201805007.htm [17] 吕文豪, 聂芳, 张雪云, 等. 超声造影评价乳腺癌病灶增强区域扩大程度与免疫指标的相关性分析[J]. 中国超声医学杂志, 2018, 34(6): 485-7. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGCY201806002.htm [18] Nie M, Qin YC, Zhu JF, et al. Correlation between ultrasonic featuresand expression levels of C-erbB-2, VEGF and nm23 in breast cancer[J]. Oncol Lett, 2018, 16(2): 1701-7. -







 下载:
下载: