Efficacy of neoadjuvant chemotherapy in breast cancer patients and its ultrasonographic BI-RADS characteristics
-
摘要:
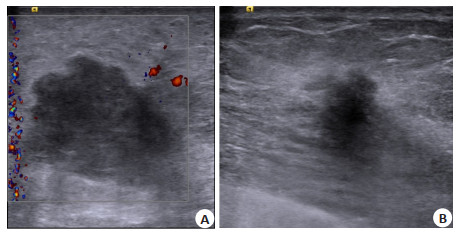
目的 分析乳腺癌患者新辅助化疗疗效与超声乳腺图像报告和数据系统(BI-RADS)征象的关系。 方法 选择我院2019年6月~2020年12月收治的86例符合标准的乳腺癌患者,根据疗效将其分为无效组(n=23)及有效组(n=63)。所有患者均采用标准新辅助化疗方案治疗;对患者行超声检查,采用BI-RADS方法记录、评估病灶危险程度。 结果 治疗后有效组患者肿瘤体积和肿瘤面积均明显低于无效组(P < 0.05);治疗后有效组患者后方回声增强、弹性评分低和边缘清晰患者占比均明显高于无效组(P < 0.05);治疗后两组患者BI-RADS分级明显优于治疗前(P < 0.05),治疗后有效组患者BI-RADS分级均明显优于无效组(P < 0.05)。 结论 采用超声BI-RADS征象分析可有效提高乳腺癌患者新辅助化疗疗效的评估价值。 Abstract:Objective To analyze the relationship between the efficacy of neoadjuvant chemotherapy and ultrasound breast image reporting and the BI-RADS signs in breast cancer patients. Methods Eighty-six patients who admitted to our hospital between June 2019 and December 2020 were selected and divided into ineffective group (n=23) and effective group (n=63) according to their efficacy. All patients were treated with standard neoadjuvant chemotherapy regimens. The patients were examined by ultrasonography, and the risk level of lesions was recorded and assessed using BI-RADS method. Results After treatment, the tumor volume and tumor area of patients in the effective group were significantly lower than those in the ineffective group (P < 0.05). The proportion of patients with clear edge, low elastic score and posterior echo enhancement in the effective group was significantly higher than that in the ineffective group (P < 0.05). The BI-RADS grading of patients in both groups was significantly better than that before treatment (P < 0.05), and the BI-RADS grading of patients in the effective group was significantly better than that of the ineffective group (P < 0.05). Conclusion ultrasound BI- RADS sign analysis can effectively improve the evaluation of the efficacy neoadjuvant chemotherapy in breast cancer patients. -
Key words:
- breast cancer /
- neoadjuvant chemotherapy /
- curative effect /
- ultrasound /
- BI-RADS signs
-
表 1 两组患者肿瘤病灶大小检测结果
Table 1. Detection results of tumor focus size in two groups (Mean±SD)
组別 肿瘤休积(cm3) 肿瘤面积(cm2) 治疗前 治疗后 治疗前 治疗后 有效组(n=63) 17.64±1.97 5.32±0.71 9.67±1.13 4.23±1.21 无效组(n=23) 17.68±2.19 8.15±0.65 9.63±1.12 7.94±1.01 t 0.056 10.017 0.089 7.831 P 0.956 < 0.001 0.929 < 0.001 表 2 两组患者超声检查影像图检测结果
Table 2. Ultrasonic imaging results of two groups[n(%)]
组別 边缘清晰 弹性评分低 后方回声增强 有效组(n=63) 治疗前 16(25.40) 20(31.75) 10(15.87) 治疗后 58(92.06) 45(71.43) 5(80.95) 无效组(n=23) 治疗前 5(21.74) 8(34.78) 5(21.74) 治疗后 14(60.87) 12(52.17) 13(56.52) 表 3 患者BI-RADS分级调查结果
Table 3. Survey results of BI- RADS classification of patients [n (%)]
组别 0~3级 4~6级 有效组(n=63) 治疗前 3(4.76) 60(95.24) 治疗后 63(100.00) 0 无效组(n=23) 治疗前 1(4.35) 22(95.65) 治疗后 20(86.96) 3(13.05) -
[1] Fernandes J, Sannachi L, Tran WT, et al. Monitoring breast cancer response to neoadjuvant chemotherapy using ultrasound strain elastography[J]. Transl Oncol, 2019, 12(9): 1177-84. doi: 10.1016/j.tranon.2019.05.004 [2] Peng J, Pu H, Jia Y, et al. Early prediction of response to neoadjuvant chemotherapy using contrast-enhanced ultrasound in breast cancer[J]. Medicine, 2021, 100(19): e25908. doi: 10.1097/MD.0000000000025908 [3] Zheng YR, Ding XW, Zou DH, et al. The treatment option of progressive disease in breast cancer during neoadjuvant chemotherapy: a single-center experience[J]. J Clin Oncol, 2019, 37 (15_suppl): e12082. doi: 10.1200/JCO.2019.37.15_suppl.e12082 [4] 中国乳腺癌新辅助治疗专家组. 中国乳腺癌新辅助治疗专家共识(2019年版)[ J]. 中国癌症杂志, 2019, 29(5): 390-400. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGAZ201905012.htm [5] Lee BH, Lee DS, Cho CW, et al. Role and limitation of neoadjuvant hepatic arterial infusion chemotherapy in advanced hepatocelluar carcinoma patients with Child-Pugh class A[J]. World J Surg Oncol, 2019, 17(1): 143. doi: 10.1186/s12957-019-1685-6 [6] Alhamad K, Alrifai N, Attah AA, et al. Neoadjuvant chemotherapy and disease recurrence after curative-intent surgery in patients with pancreatic adenocarcinoma: a single-center retrospective review[J]. J Clin Oncol, 2021, 39(15_suppl): e18610. doi: 10.1200/JCO.2021.39.15_suppl.e18610 [7] Shah MA, Almhanna K, Iqbal S, et al. Multicenter, randomized phase Ⅱ study of neoadjuvant pembrolizumab plus chemotherapy and chemoradiotherapy in esophageal adenocarcinoma (EAC)[J]. J Clin Oncol, 2021, 39(15_suppl): 4005. doi: 10.1200/JCO.2021.39.15_suppl.4005 [8] 安小筱, 毛大华, 龚城, 等. 乳腺癌新辅助化疗后病理完全缓解的意义[J]. 临床医药文献电子杂志, 2019, 6(30): 83. https://www.cnki.com.cn/Article/CJFDTOTAL-LCWX201930074.htm [9] Miron B, Ross EA, Anari F, et al. Defects in DNA repair genes and long-term survival in cisplatin-based neoadjuvant chemotherapy for muscle invasive bladder cancer (MIBC)[J]. J Clin Oncol, 2019, 37 (15): 4536. [10] Blair AB, Sorber R, Rozich NS, et al. A qualitative review of neoadjuvant chemotherapy in resectable pancreatic adenocarcinoma [J]. Pancreas, 2019, 48(8): 973-84. doi: 10.1097/MPA.0000000000001376 [11] Kumar S, Saikia J, Kumar V Jr, et al. Neoadjuvant chemotherapy followed by surgery in lung cancer: Indian scenario[J]. Curr Probl Cancer, 2020, 44(3): 100563. doi: 10.1016/j.currproblcancer.2020.100563 [12] 王仲臣, 王广征, 谭亭昭. 表柔比星与多西他赛不同联合方案对乳腺癌患者新辅助化疗效果的影响[J]. 实用医院临床杂志, 2019, 16(5): 87-91. https://www.cnki.com.cn/Article/CJFDTOTAL-YYLC201905027.htm [13] 赵永存, 高秀琴, 李惠娟, 等. 高频超声应用乳腺超声影像报告和数据系统分类在乳腺癌筛查中的临床价值[J]. 中国妇幼保健, 2021, 36 (22): 5334-7. https://www.cnki.com.cn/Article/CJFDTOTAL-ZFYB202122065.htm [14] 陈轶洁, 唐丽娜, 刘健, 等. 超声造影评估乳腺癌新辅助化疗疗效的效能分析[J]. 中华内分泌外科杂志, 2019, 13(5): 383-6. doi: 10.3760/cma.j.issn.1674-6090.2019.05.007 [15] 张岳宇, 孔繁云, 陈成辉. 彩色多普勒超声检查评价乳腺癌新辅助化疗疗效及复发的价值[J]. 癌症进展, 2019, 17(13): 1519-21. https://www.cnki.com.cn/Article/CJFDTOTAL-AZJZ201913010.htm [16] 杨梅, 李伟萍, 王坤. 乳腺癌新辅助化疗疗效评估及治疗方案的多学科讨论[J]. 循证医学, 2019, 19(2): 126-8. https://www.cnki.com.cn/Article/CJFDTOTAL-YEBM201902020.htm [17] 张小平, 杨军, 吴文飞. 部分乳腺肿瘤BI-RADS分类术前评估在临床治疗中的应用价值[J]. 中外医学研究, 2019, 17(33): 78-80. https://www.cnki.com.cn/Article/CJFDTOTAL-YJZY201933031.htm [18] 杨月杰. 超声造影技术在乳腺癌新辅助化疗疗效评估中的应用分析[J]. 山西医药杂志, 2019, 48(2): 166-7. https://www.cnki.com.cn/Article/CJFDTOTAL-SXYY201902013.htm [19] 杨立光, 周倩, 李启霖, 等. 功能成像技术对乳腺癌新辅助化疗后疗效评估的研究进展[J]. 分子影像学杂志, 2019, 42(3): 285-9. doi: 10.12122/j.issn.1674-4500.2019.03.01 [20] Sohal D, Duong MT, Chang R, et al. Immunologic predictors of therapeutic response to neoadjuvant chemotherapy for pancreatic ductal adenocarcinoma (PDA) in SWOG S1505[J]. J Clin Oncol, 2021, 39(3_suppl): 419. -







 下载:
下载: