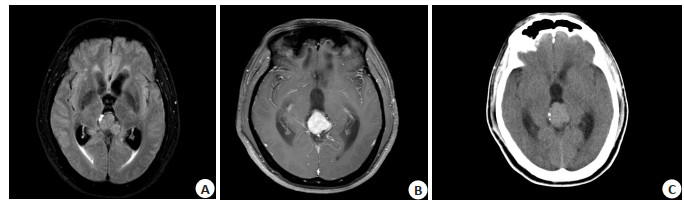
Imaging features and differential diagnosis of pineal parenchymal cell tumors
-
摘要:
目的 探讨松果体实质细胞肿瘤的CT及MRI影像特征。 方法 收集本院34例松果体实质细胞肿瘤的CT、MRI及临床资料,根据病理结果将病例分为3组:松果体细胞瘤(PC组,n=9)、中间分化型松果体实质细胞肿瘤(PPTID组,n=14)和松果体母细胞瘤(PB组,n=11);使用单因素方差分析及卡方检验对3组病例的影像观察指标进行统计学分析。 结果 松果体实质细胞肿瘤以边界清楚、形态规则的T1WI低信号、T2WI高信号、明显强化为主要表现,扩散加权成像呈高信号,磁共振波谱成像示Cho/NAA比值升高。3组肿瘤患者的年龄(P=0.009)及囊变(P=0.047)情况差异有统计学意义。将年龄及囊变情况进一步行两两比较,结果显示PB组患者年龄较PPTID(P=0.007)及PC组(P=0.007)小,而PC组发生囊变较PB组(P=0.019)低。 结论 松果体实质细胞瘤具有一定的临床特征,3种病理类型具有一定影像差异,通过影像学方法有助于鉴别其病理类型。 Abstract:Objective To investigate the CT and MRI features of pineal parenchymal cell tumors. Methods CT, MRI and clinical data of 34 cases of pineal parenchymal cell tumor in our hospital were collected. The patients were divided into three groups according to the pathological results: pineocytoma (PC group), moderately differentiated pineal parenchymal cell tumor (PPTID group) and pineoblastoma (PB group). Results Pineal parenchymal tumors were mainly characterized by well-defined, regular shape of low signal intensity on T1WI, high signal intensity on T2WI, and significant enhancement, high signal intensity on diffusion-weighted imaging, and elevated Cho/NAA ratio on magnetic resonance spectroscopy. Among the three groups cases, there were significant differences in age (P=0.009) and cystic degeneration (P=0.047). We found that the age of patients with PB were younger than that in both PPTID group (P=0.007) and PC group (P=0.007), while cystic changes occurred less frequently in the PC group than in the PB group (P=0.019). Conclusion With certain clinical and imaging features, pathological types of pineal parenchymal tumors can be identify with the help of CT and MRI technology -
表 1 3组病例的一般资料及肿瘤边界、形态、最大径测量对比
Table 1. Comparison of clinical information, boundary, shape and maximum diameter of tumors in cases of three groups (n)
项目 PC(n=9) PPTID(n=14) PB(n=11) F/χ2 P 年龄(岁) 5.536 0.009 < 20 2 3 7 > 20 7 11 4 性別 0.413 0.910 男 5 9 6 女 4 5 5 边界 1.053 1.000 清楚 9 13 11 侵犯周围组织 0 1 0 形态 1.600 0.537 类圆形、分叶状 9 12 9 不规则 0 2 2 肿瘤最大径(mm) 2.389 0.108 < 40 9 11 6 > 40 0 3 5 PC:松果体细胞瘤;PPTID:中分化松果体实质细胞肿瘤;PB:松果体母细胞瘤. 表 2 3组肿瘤的MR信号特征及瘤内特殊MR信号成分对比
Table 2. Comparison of MR signal and specific morphologic components of tumors in three groups (n)
项目 PC(n=9) PPTID(n=14) PB(n=11) χ2 P MR T1WI信号 - - 等 5 3 0 低 3 4 3 高 0 0 0 混杂 1 7 8 MR T2WI信号 - - 等 1 1 0 低 1 0 0 高 7 6 3 混杂 0 7 8 强化程度 0.573 0.887 无-中 3 3 3 重 6 11 8 强化方式 1.138 0.626 均匀 5 6 7 不均匀 4 8 4 囊变 5.826 0.047 是 1 7 7 否 8 7 4 钙化 2.445 0.349 是 4 5 2 否 5 9 9 出血 0.494 0.887 是 2 2 2 否 7 12 9 脑积水 1.391 0.725 是 8 13 11 否 1 1 0 表 3 3组肿瘤DWI、MRS及CT表现对比
Table 3. Comparison of DWI, MRS and CT performance of tumors among the three groups (n)
项目 PC(n=9) PPTID(n=14) PB(n=11) DWI信号 8 13 4 等 1 1 0 低 2 0 0 高 5 9 4 混杂 0 3 0 MRS表现 0 2 1 Cho峰上升 - 2 1 Cho峰不上升 - 0 0 MRS表现 0 2 1 NAA峰降低 - 1 1 NAA峰不降低 - 1 0 CT平扫密度 7 9 5 等 3 2 0 低 0 0 0 高 0 1 1 混杂 4 6 4 表 4 3组肿瘤年龄及囊变情况LSD两两对比
Table 4. Age and cystic change in three groups by LSD statistical method
指标 PC vs PPTID PPTID vs PB PC vs PB 年龄(Mean±SD) 2.2381±8.1014 22.1740±7.6400 24.4121±8.5227 P 0.784 0.007 0.007 囊变(Mean±SD) 0.3889±0.2021 0.1364±0.1906 0.5253±0.2126 P 0.064 0.480 0.019 -
[1] Raleigh DR, Solomon DA, Lloyd SA, et al. Histopathologic review of pineal parenchymal tumors identifies novel morphologic subtypes and prognostic factors for outcome[J]. Neuro Oncol, 2017, 19(1): 78-88. doi: 10.1093/neuonc/now105 [2] Ito T, Kanno H, Sato K, et al. Clinicopathologic study of pineal parenchymal tumors of intermediate differentiation[J]. World Neurosurg, 2014, 81(5/6): 783-9. [3] Li BK, Vasiljevic A, Dufour C, et al. Pineoblastoma segregates into molecular sub-groups with distinct clinico-pathologic features: a Rare Brain Tumor Consortium registry study[J]. Acta Neuropathol, 2020, 139(2): 223-41. doi: 10.1007/s00401-019-02111-y [4] Mandera M, Marcol W, Kotulska K, et al. Childhood pineal parenchymal tumors: clinical and therapeutic aspects[J]. Neurosurg Rev, 2011, 34(2): 191-6. doi: 10.1007/s10143-010-0287-0 [5] Singla N, Kapoor A, Dhandapani S, et al. Revisiting the metastatic potential of childhood pineal parenchymal tumor of intermediate differentiation: a case report[J]. Childs Nerv Syst, 2016, 32(7): 1183-5. doi: 10.1007/s00381-016-3117-z [6] Tong T, Yao ZW, Feng XY. TEMPORARY REMOVAL: MRI and 1H-MRS on diagnosis of pineal region tumors[J]. Clin Imaging, 2012, 36(6): 702-9. doi: 10.1016/j.clinimag.2012.01.039 [7] Kang YJ, Bi WL, Dubuc AM, et al. Integrated genomic characterization of a pineal parenchymal tumor of intermediate differentiation[J]. World Neurosurg, 2016, 85: 96-105. doi: 10.1016/j.wneu.2015.07.032 [8] Chung PED, Gendoo DMA, Ghanbari-Azarnier R, et al. Modeling germline mutations in pineoblastoma uncovers lysosome disruptionbased therapy[J]. Nat Commun, 2020, 11(1): 1825. doi: 10.1038/s41467-020-15585-2 [9] Tian Y, Liu R, Qin J, et al. Retrospective analysis of the clinical characteristics, therapeutic aspects, and prognostic factors of 18 cases of childhood pineoblastoma[J]. World Neurosurg, 2018, 116: e162-8. doi: 10.1016/j.wneu.2018.04.135 [10] 史玉振, 田迎, 童明敏, 等. 松果体实质细胞肿瘤的MRI表现[J]. 放射学实践, 2012, 27(11): 1187-90. doi: 10.3969/j.issn.1000-0313.2012.11.008 [11] Kakigi T, Okada T, Kanagaki M, et al. Quantitative imaging values of CT, MR, and FDG-PET to differentiate pineal parenchymal tumors and germinomas: are they useful?[J]. Neuroradiology, 2014, 56(4): 297-303. doi: 10.1007/s00234-014-1334-2 [12] Fakhran S, Escott EJ. Pineocytoma mimicking a pineal cyst on imaging: true diagnostic dilemma or a case of incomplete imaging? : fig 1[J]. AJNR Am J Neuroradiol, 2008, 29(1): 159-63. doi: 10.3174/ajnr.A0750 [13] Awa R, Campos F, Arita K, et al. Neuroimaging diagnosis of pineal region tumors-quest for pathognomonic finding of germinoma[J]. Neuroradiology, 2014, 56(7): 525-34. doi: 10.1007/s00234-014-1369-4 [14] Mano Y, Kanamori M, Kumabe T, et al. Extremely late recurrence 21 years after total removal of immature teratoma: a case report and literature review[J]. Neurol Med Chir: Tokyo, 2017, 57(1): 51-6. doi: 10.2176/nmc.cr.2016-0241 [15] Suh CH, Kim HS, Jung SC, et al. 2-Hydroxyglutarate MR spectroscopy for prediction of isocitrate dehydrogenase mutant glioma: a systemic review and meta-analysis using individual patient data[J]. Neuro Oncol, 2018, 20(12): 1573-83. doi: 10.1093/neuonc/noy113 -







 下载:
下载: