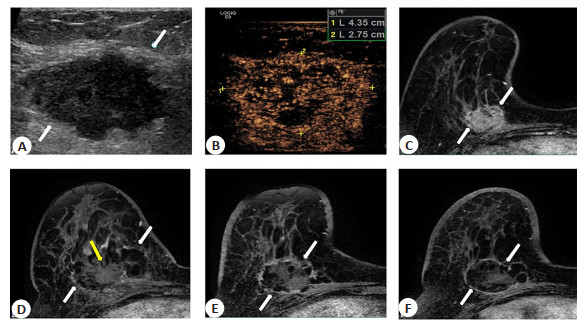
Microwave ablation versus modified radical treatment with preserved nipple sparing mastectomy for early breast cancer: a propensity score-matched analysis
-
摘要:
目的 比较超声引导下微波消融(MWA)与保留乳头乳晕复合体改良根治术(NSM)治疗早期乳腺癌(T0/1/2 N0/1 M0)的可行性和有效性。 方法 回顾性分析本院2014年1月~2020年1月进行MWA或NSM治疗的83例早期乳腺癌患者,其中MWA组30例,NSM组53例。通过倾向性评分匹配按1∶1平衡两组基线特征。主要评价指标为肿瘤进展,次要评价指标包括总生存、美容满意度和并发症。 结果 倾向性评分匹配前平均肿瘤大小2.3 cm(0.3~5.0 cm);倾向性评分匹配后两组患者各19例,基线特征平衡。中位随访时间34.1(四分位数23.1~41.1)月。所有肿瘤均取得技术成功。转移性淋巴结处理,两组各消融/切除腋窝淋巴结4例(4/19,21.0%)。MWA组术后辅助全身性治疗比NSM组少(36.8% vs 73.7%,P=0.02)。共有6例发生肿瘤进展,其中MWA组3例,NSM组3例。MWA组术后42月发生1例局部肿瘤进展,28月发生1例同侧乳房转移,32.8月发生1例脑转移;NSM术后10.2月、31.2月各发生1例同侧乳房转移,34月发生1例骨转移,两组肿瘤进展差异无统计学意义(P=0.68)。两组均无乳腺癌相关死亡和严重并发症(P>0.05)。但MWA组手术时间短(32 min vs 133 min,P < 0.001)、术后住院时间短(3 d vs 5 d,P < 0.001)、美容满意度更好(P < 0.001)。 结论 在早期乳腺癌患者中,中位随访期间MWA取得了与NSM相似的治疗效果及更好的美容满意度,MWA为不愿或不耐受手术患者提供了微创安全可耐受的局部治疗选择。 Abstract:Objective To compare the feasibility and effectiveness of ultrasound-guided microwave ablation (MWA) and nipple sparing mastectomy (NSM) for early breast cancer (T0/1/2 N0/1 M0). Methods Retrospective analysis of 83 patients with early breast cancer who underwent MWA or NSM from January 2014 to January 2020 in our hospital. including 30 patients in the MWA group and the other 53 patients in the NSM group. Propensity score matching was performed at 1∶1 to balance the baseline characteristics. The primary evaluation metric was tumor progression in the intention-to-treat population, and secondary evaluation metrics included overall survival, cosmetic satisfaction and complications. Results Before propensity score matching The mean tumor size was 2.3 cm (0.3-5.0 cm). After propensity score matching, each group had 19 patients and the baseline characteristic were balanced. Median follow-up was 34.1 months (IQR 23.1-41.1 months). All the tumors achieved technique effectiveness. For the management of metastatic lymph nodes, 4 axillary lymph nodes were ablated/resected in both groups (4/19, 21.0%). There were fewer adjuvant systemic postoperative treatments in the MWA group than in the NSM group (36.8% vs 73.7%, P=0.02). A total of 6 cases of tumour progression occurred, including 3 in the MWA and 3 in the NSM. One local tumor progression occurred in the MWA group at 42 mouths, one ipsilateral breast recurrence occurred at 28 months and 1 case of brain metastasis at 32.8 months after MWA. 1 case of ipsilateral breast metastasis at 10.2 months and 1 case of bone metastasis at 31.2 months and 1 case of bone metastasis at 34 months postoperatively in the NSM group. Two groups had no significant difference in tumor progression (P=0.68). No participants in both groups developed cancer related death and major complications (P>0.05). MWA achieves similar outcomes to NSM and better cosmetic satisfaction during median follow-up in patients with early-stage breast cancer, and that who do not want or are intolerant of surgery.However, MWA had a shorter operation time (32 min vs 133 min, P < 0.001), a shorter postoperative hospital stay (3 d vs 5 d, P < 0.001) and better beauty satisfaction (P < 0.001). Conclusion Preliminary study showed that in selected early breast cancer patients, MWA has achieved similar outcomes to early breast cancer treatment and better aesthetic satisfaction as NSM during an intermediate follow-up. MWA offers a minimally invasive, safe and tolerable local treatment option for patients who reluctant or intolerant to breastconserving surgery. -
图 2 MWA和NSM治疗乳腺癌肿瘤进展的KM曲线
A: 倾向性评分匹配前两组肿瘤进展差异无统计学意义(P=0.49, Log-rank检验),MWA和NSM组1、2、3年的肿瘤进展率分别为0%、0%、16.1%和1.9%、14.5%、14.5%; B: 倾向性评分匹配后两组肿瘤进展差异无统计学意义(P=0.86, Log-rank检验), MWA和NSM组1、2、3年的肿瘤进展率分别为0%、0%、16.1% 和5.3%、11.6%、21.4%.
Figure 2. Kaplan- Meier estimates for tumor progression between breast cancer patients who underwent MWA and NSM.
表 1 两组患者的一般资料特征
Table 1. Baseline characteristics for patients of two groups [n(%)]
指标 倾向性评分匹配前 倾向性评分匹配后 MWA(n=30) NSM (n=53) P MWA(n=19) NSM (n=19) P 年龄(岁) < 0.001 1.00 < 45 3 (10.0) 37(69.8) 3 (15.8) 3 (15.8) ≥45 27(90.0) 16(30.2) 16 (84.2) 16(84.2) 直径(cm, Mean±SD) 2.7±1.2 2.0±1.0 0.01 2.3±1.0 1.8±1.0 0.16 ≤2.0 10(33.3) 27(51.0) 9 (47.3) 12(63.2) 2.1~3.0 10(33.3) 19(35.8) 5 (26.3) 6 (31.6) 3.1~4.0 5 (16.7) 5(9.4) 4 (21.1) 1(5.3) 4.1~5.0 5 (16.7) 2(3.8) 1 (5.3) 0(0.0) 肿瘤数量 0.47 单个 23(76.7) 45(84.9) 17 (89.5) 16(84.2) 1.00 多个 7 (23.3) 8 (15.1) 2(10.5) 3 (15.8) 肿瘤位置 0.24 0.51 左乳 13(43.3) 30(56.6) 7 (36.8) 9 (47.4) 右乳 17(56.7) 23(43.4) 12 (63.2) 10(52.6) 初发肿瘤 是 23(86.8) 46(76.7) 15 (78.9) 17(89.5) 否 7 (13.2) 7 (23.3) 4 (21.1) 2 (10.5) TNM分期 0.37 0.52 T1N0M0 5 (16.7) 15(28.3) 4 (21.1) 5 (26.3) T1N1M0 6 (20.0) 7 (13.2) 4 (21.1) 5 (26.3) T1N2M0 1(3.3) 3(5.7) 1 (5.3) 1(5.3) T2N0M0 8 (26.7) 18(34.0) 5 (26.3) 4 (21.1) T2N1M0 4 (13.3) 5(9.4) 3 (15.8) 3 (15.8) T2N2M0 6 (20.0) 5(9.4) 2 (10.5) 1(5.3) 分子亚型 0.007 0.24 管腔A 11(36.7) 12(22.6) 9 (47.4) 8 (42.1) 管腔B 官腔B1 4 (13.3) 27(50.9) 2 (10.5) 4 (21.1) 官腔B2 3 (10.0) 6 (11.3) 3 (15.8) 2 (10.5) HER2阴性 2(6.7) 4(7.5) 1 (5.3) 3 (15.8) 三阴性 6 (20.0) 3(5.7) 2 (10.5) 1(5.3) 未确定 4 (13.3) 1(1.9) 2 (10.5) 1(5.3) MWA: 微波消融; NSM: 保留乳头乳晕复合体改良根治术. 表 2 辅助治疗
Table 2. Adjuvant Treatment [n(%)]
指标 倾向性匹配评分前 倾向性匹配评分后 MWA (n=30) NSM (n=53) P MWA (n=19) NSM (n=19) P 辅助性全身疗法 9 (32.1) 43 (81.1) < 0.001 7 (36.8) 14 (73.7) 0.02 内分泌治疗 3 (10.7) 10 (18.9) 0.34 3 (15.8) 3 (15.8) 1.00 化疗 4 (14.3) 17 (32.1) 0.11 2 (10.5) 7 (36.8) 0.06 内分泌治疗+化疗 2(7.1) 16 (30.2) 0.02 2 ((10.5)) 4 (21.1) 0.66 辅助性放疗 4 (14.3) 14 (26.4) 0.31 4 (21.1) 7 (36.8) 0.28 淋巴结放疗 2(7.1) 0 0.12 2 (10.5) 0 (0.0) 0.15 乳腺+淋巴结放疗 2(7.1) 14 (26.4) 0.04 2 (10.5) 7 (36.8) 0.049 表 3 术后结果及随访
Table 3. Postoperative outcomes and follow-up [n(%)]
指标 倾向性评分匹配前 倾向性评分匹配后 MWA (n=30) NSM (n=53) P MWA (n=19) NSM (n=19) P 术后住院时间(d)* 3.0(1.5~3.0) 5.0(3.2~6.0) < 0.001 3.0(1.5~3.0) 5.0(3.0~6.0) < 0.001 手术时间(min, Mean±SD) 31.2±16.3 131.5±47.3 < 0.001 32.9±15.4 133.2 ±28.2 < 0.001 出血量(mL, Mean±SD) 2.0±0.5 139.0±100.0 < 0.001 2.0±0.5 102.6±77.2 < 0.001 体温>38 ℃ 1(3.3) 3(5.7) 0.68 0 (0.0) 0(0.0) >0.99 严重并发症 0(0.0) 0(0.0) >0.99 0 (0.0) 0(0.0) >0.99 全因死亡 2(6.7) 0(0.0) 0.99 1 (5.3) 0(0.0) 0.31 乳腺癌相关死亡 0(0.0) 0(0.0) >0.99 0 (0.0) 0(0.0) >0.99 局部复发 1(3.3) 0(0.0) 0.15 1 (5.3) 0(0.0) 0.15 同侧乳腺复发 1(3.3) 2(3.8) 0.13 1 (5.3) 1(5.3) 1.0 乳腺外转移 1(3.3) 4(7.5) 0.34 1 (5.3) 2 (10.5) 0.66 花费(元) * 24512.2
(21963.1~36591.8)21048.7
(18118.9~24507.3)0.005 24366.2
(21438.2~25931.8)22584.0
(18609.2~27304.8)0.23 消融/切除腋窝淋巴结 8 (26.7) 10(18.9) 0.26 4 (21.0) 4 (21.0) 1.0 美容满意度 < 0.001 < 0.001 不满意 0(0.0) 1(1.9) 0 (0.0) 1(5.3) 一般 0(0.0) 11(20.8) 0 (0.0) 4 (21.1) 较满意 3 (10.0) 36(67.9) 0 (0.0) 11(57.9) 非常满意 27(90.0) 5(9.4) 19 (100.0) 3 (15.8) *以中位数(四分位数间距表示). 表 4 Cox分析MWA和NSM与肿瘤进展的相关性
Table 4. Cox associations between MWA and NSM with tumor progression
治疗方式 PSM前 PSM后 模型1a 模型2b 模型1a 模型2b HR (95% CI) P HR (95% CI) P HR (95% CI) P HR (95% CI) P NSM 参照组 参照组 参照组 参照组 MWA 1.6 (0.4, 6.7) 0.49 1.7(0.1, 15.1) 0.72 1.2 (0.2, 5.8) 0.86 1.6 (0.2, 13.7) 0.68 a未调整; b调整年龄、肿瘤直径、肿瘤数量、肿瘤位置、初发肿瘤、术后放疗、术后化疗、术后内分泌治疗. -
[1] Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394-424. doi: 10.3322/caac.21492 [2] de la Cruz L, Blankenship SA, Chatterjee A, et al. Outcomes after oncoplastic breast-conserving surgery in breast cancer patients: a systematic literature review[J]. Ann Surg Oncol, 2016, 23(10): 3247-58. doi: 10.1245/s10434-016-5313-1 [3] van Maaren MC, de Munck L, de Bock GH, et al. 10 year survival after breast-conserving surgery plus radiotherapy compared with mastectomy in early breast cancer in the Netherlands: a populationbased study[J]. Lancet Oncol, 2016, 17(8): 1158-70. doi: 10.1016/S1470-2045(16)30067-5 [4] Zarba Meli E, Cattin F, Curcio A, et al. Surgical delay may extend the indications for nipple-sparing mastectomy: a multicentric study[J]. Eur J Surg Oncol, 2019, 45(8): 1373-7. doi: 10.1016/j.ejso.2019.02.014 [5] Fleming MM, Holbrook AI, Newell MS. Update on image-guided percutaneous ablation of breast cancer[J]. AJR Am J Roentgenol, 2017, 208(2): 267-74. doi: 10.2214/AJR.16.17129 [6] Brem RF. Radiofrequency ablation of breast cancer: a step forward[J]. Radiology, 2018, 289(2): 325-6. doi: 10.1148/radiol.2018181784 [7] Lanza E, Palussiere J, Buy X, et al. Percutaneous image-guided cryoablation of breast cancer: a systematic review[J]. J Vasc Interv Radiol, 2015, 26(11): 1652-7. e1. doi: 10.1016/j.jvir.2015.07.020 [8] Peek MC, Ahmed M, Napoli A, et al. Systematic review of highintensity focused ultrasound ablation in the treatment of breast cancer[J]. Br J Surg, 2015, 102(8): 873-82. doi: 10.1002/bjs.9793 [9] van Hillegersberg R, van Staveren HJ, Roggan A, et al. Interstitial laser photocoagulation as a treatment for breast cancer[J]. Br J Surg, 1995, 82(6): 856. [10] Zhou WB, Jiang YN, Chen L, et al. Image and pathological changes after microwave ablation of breast cancer: a pilot study[J]. Eur J Radiol, 2014, 83(10): 1771-7. doi: 10.1016/j.ejrad.2014.06.015 [11] Yu J, Liang P, Yu XL, et al. A comparison of microwave ablation and bipolar radiofrequency ablation both with an internally cooled probe: results in ex vivo and in vivo porcine livers[J]. Eur J Radiol, 2011, 79(1): 124-30. doi: 10.1016/j.ejrad.2009.12.009 [12] Giorgio A, Gatti P, Montesarchio L, et al. Microwave ablation in intermediate hepatocellular carcinoma in cirrhosis: an Italian multicenter prospective study[J]. J Clin Transl Hepatol, 2018, 6(3): 251-7. [13] Liang P, Yu J, Yu XL, et al. Percutaneous cooled-tip microwave ablation under ultrasound guidance for primary liver cancer: a multicentre analysis of 1363 treatment-naive lesions in 1007 patients in China[J]. Gut, 2012, 61(7): 1100-1. doi: 10.1136/gutjnl-2011-300975 [14] Cil TD, McCready D. Modern approaches to the surgical management of malignant breast disease: the role of breast conservation, complete mastectomy, skin-and nipple-sparing mastectomy[J]. Clin Plast Surg, 2018, 45(1): 1-11. doi: 10.1016/j.cps.2017.07.002 [15] Mauri G, Sconfienza LM, Pescatori LC, et al. Technical success, technique efficacy and complications of minimally-invasive imaging-guided percutaneous ablation procedures of breast cancer: a systematic review and meta-analysis[J]. Eur Radiol, 2017, 27(8): 3199-210. doi: 10.1007/s00330-016-4668-9 [16] Field TS, Bosco JLF, Prout MN, et al. Age, comorbidity, and breast cancer severity: impact on receipt of definitive local therapy and rate of recurrence among older women with early-stage breast cancer[J]. JAm Coll Surg, 2011, 213(6): 757-65. doi: 10.1016/j.jamcollsurg.2011.09.010 [17] Galimberti V, Cole BF, Viale G, et al. Axillary dissection versus no axillary dissection in patients with breast cancer and sentinel-node micrometastases (IBCSG 23-01): 10-year follow-up of a randomised, controlled phase 3 trial[J]. Lancet Oncol, 2018, 19(10): 1385-93. doi: 10.1016/S1470-2045(18)30380-2 [18] Jacobson JA, Danforth DN, Cowan KH, et al. Ten-year results of a comparison of conservation with mastectomy in the treatment of stage I and Ⅱ breast cancer[J]. N Engl J Med, 1995, 332(14): 907-11. doi: 10.1056/NEJM199504063321402 [19] Romanoff A, Zabor EC, Stempel M, et al. A comparison of patientreported outcomes after nipple-sparing mastectomy and conventional mastectomy with reconstruction[J]. Ann Surg Oncol, 2018, 25(10): 2909-16. doi: 10.1245/s10434-018-6585-4 [20] Veronesi U, Salvadori B, Luini A, et al. Conservative treatment of early breast cancer. Long-term results of 1232 cases treated with quadrantectomy, axillary dissection, and radiotherapy[J]. Ann Surg, 1990, 211(3): 250-9. [21] Ashikari AY, Kelemen PR, Tastan B, et al. Nipple sparing mastectomy techniques: a literature review and an inframammary technique[J]. Gland Surg, 2018, 7(3): 273-87. doi: 10.21037/gs.2017.09.02 [22] Galimberti V, Morigi C, Bagnardi V, et al. Oncological outcomes of nipple-sparing mastectomy: a single-center experience of 1989 patients[J]. Ann Surg Oncol, 2018, 25(13): 3849-57. doi: 10.1245/s10434-018-6759-0 [23] Chen JY, Zhang C, Li F, et al. A meta-analysis of clinical trials assessing the effect of radiofrequency ablation for breast cancer[J]. Onco Targets Ther, 2016, 9: 1759-66. [24] Zhou WB, Zha XM, Liu XA, et al. US-guided percutaneous microwave coagulation of small breast cancers: a clinical study[J]. Radiology, 2012, 263(2): 364-73. doi: 10.1148/radiol.12111901 [25] Yu J, Han ZY, Li T, et al. Microwave ablation versus nipple sparing mastectomy for breast cancer ≤ 5 cm: a pilot cohort study[J]. Front Oncol, 2020, 10: 546883. doi: 10.3389/fonc.2020.546883 -







 下载:
下载: