Correlation between ultrasonographic features and expression of tumor stem cells and epithelial mesenchymal transition markers in breast cancer
-
摘要:
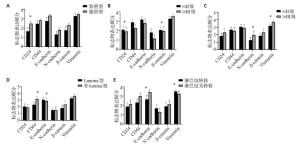
目的探讨乳腺癌超声征象与肿瘤干细胞及上皮间质转化标志物表达水平的相关性及其意义。 方法选择在我院行乳腺癌手术的患者组织标本98例,分析每位患者对应的乳腺超声征象,包括肿块周边是否有毛刺、边缘是否有高回声晕、纵横比、后方回声情况、微小钙化、内部血流显像分级;并采用免疫组化方法检测组织样本中CD24、CD44、E-cadherin、N-cadherin、β-catenin、Vimentin的表达情况,分析其与超声表现的相关性。分析其它可能影响肿瘤干细胞及上皮间质转化标志物表达的因素;对肿瘤干细胞及上皮间质转化标志物表达相关性进行多因素logistic回归分析。 结果肿瘤周边是否有毛刺与CD44、E-cadherin、N-cadherin、β-catenin的表达水平差异有统计学意义(P < 0.05)。肿块边缘是否有高回声晕与CD44和E-cadherin的表达水平差异有统计学意义(P < 0.05)。肿块纵横比、后方回声变化均与E-cadherin的表达水平差异有统计学意义(P < 0.05)。血流分级、腺体类型与CD44的表达水平差异有统计学意义(P < 0.05)。腺体背景类型是CD24表达的独立相关因素(P < 0.05)。血流情况、雌孕激素受体表达情况是CD44表达的独立相关因素(P < 0.05)。有无高回声晕、肿块后方回声特征、腋窝淋巴结是否有转移是E-cadherin表达的独立相关因素(P < 0.05)。临床分期是N-cadherin表达的独立相关因素(P < 0.05)。边缘有无毛刺是β- catenin表达的独立相关因素(P < 0.05)。 结论乳腺癌超声征象与肿瘤干细胞及上皮间质转化标志物表达之间存在联系,超声征象可作为无创性预测乳腺癌患者肿瘤干细胞及上皮间质转化标志物表达水平的方式,并可为预测乳腺癌潜在的侵袭能力提供更多依据。 Abstract:ObjectiveTo investigate the correlation and significance of ultrasonographic signs of breast cancer with the expression level of tumor stem cells and epithelial mesenchymal transformation markers. MethodsA total of 98 tissue specimens from patients undergoing breast cancer surgery in our hospital were selected. The corresponding breast ultrasound signs of each patient were analyzed, including whether there were burrs around the mass, whether there was hyperechoic halo at the edge, aspect ratio, posterior echo, microcalcification, and internal blood flow imaging grade. The expression of CD24, CD44, E-cadherin, N-cadherin, β-catenin, Vimentin were detected by immunohistochemistry. The correlation between these indexes and ultrasound findings were analyzed. Other factors that may affect the expression of tumor stem cells and epithelial mesenchymal transition markers were analyzed. Multivariate logistic regression analysis was used to analyze the correlation between the expression of tumor stem cells and epithelial mesenchymal transition markers. ResultsThe presence of burr around the tumor was significantly different with the expression levels of CD44, E-cadherin, N-cadherin and β-catenin (P < 0.05). The expression level of CD44 and E-cadherin were significantly different with the presence of hyperechoic halos at the edge of the mass (P < 0.05).The changes of aspect ratio and posterior echo of the tumor were significantly different with the expression level of E-cadherin (P < 0.05).There were significant differences in blood flow grade, gland type and CD44 expression level (P < 0.05). Gland background type was an independent factor associated with CD24 expression. Blood flow and grouping according to estrogen receptor expression were independent correlative factors of CD44 expression. The presence of hyperechoic halos, posterior echo characteristics of the mass, and axillary lymph node metastasis were independent related factors of E-cadherin expression. Clinical staging was an independent factor associated with N-cadherin expression. The presence of burr on the edge was an independent factor related to β-catenin expression. ConclusionUltrasound signs of breast cancer are associated with the expression of tumor stem cells and epithelial mesenchymal transformation markers. Ultrasound signs can be used as a way to noninvasively predict the expression level of tumor stem cells and epithelial mesenchymal transformation markers in breast cancer patients, and can provide more evidence for predicting the potential invasion ability of breast cancer. -
Key words:
- breast cancer /
- ultrasonic signs /
- cancer stem cell /
- epithelial mesenchymal transition
-
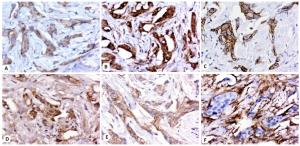
图 2 乳腺癌肿瘤干细胞及上皮间质转化标志物镜下图像
A:CD24在癌细胞细胞质中呈弱阳性表达(+),赋分1分;B:CD44在癌细胞的细胞膜和细胞质中呈强阳性表达(++++),赋分4分;C:E-cadherin在癌细胞细胞膜和细胞质中呈阳性表达(++),赋分2分;D:N-cadherin在癌细胞细胞膜中呈阳性表达(++),赋分2分;E:β-catenin在癌细胞细胞膜中呈强阳性表达(+++),赋分3分;F:Vimentin在癌巢周围组织细胞质中呈强阳性表达(++++),赋分4分.
Figure 2. Here are objective images of breast cancer tumor stem cells and epithelial mesenchymal transformation markers (Original magnification: ×100).
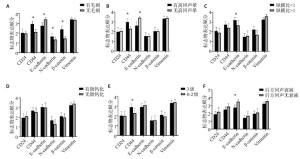
表 1 CD24表达水平多因素分析
Table 1. Multivariate analysis of CD24 expression level
因素 估算 标准误 瓦尔德 自由度 显著性 95% CI 下限 上限 [腺体背景(1.致密型; 2.脂肪型)=1] -1.008 0.375 7.223 1 0.007 -1.744 -0.273 [组织学分级(1.小于等于Ⅱ级; 2.大于等于Ⅲ级)=1] 0.275 0.367 0.561 1 0.454 -0.444 0.993 表 2 CD44表达水平多因素分析
Table 2. Multivariate analysis of CD44 expression level
因素 估算 标准误 瓦尔德 自由度 显著性 95% CI 下限 上限 [高回声晕(1.有; 2.无)=1] 0.513 0.416 1.52 1 0.218 -0.302 1.328 [边缘毛刺(1.有; 2.无)=1] 0.707 0.426 2.752 1 0.097 -0.128 1.543 [血流分组(1.丰富; 2.不丰富)=1] 0.875 0.396 4.886 1 0.027 0.099 1.651 [雌孕激素受体分型(1.非luminal型; 2.luminal型)=1] 1.387 0.429 10.431 1 0.001 0.545 2.229 表 3 E-cadherin表达水平多因素分析
Table 3. Multivariate analysis of E-cadherin expression level
因素 估算 标准误 瓦尔德 自由度 显著性 95% CI 下限 上限 [边缘毛刺(1.有; 2.无)=1] -0.87 0.482 3.258 1 0.071 -1.814 0.075 [有无高回声晕(1.有; 2.无)=1] -1.216 0.46 6.972 1 0.008 -2.118 -0.313 [纵横比(1.小于1; 2.大于1)=1] 0.552 0.423 1.703 1 0.192 -0.277 1.381 [后方回声特征(1.不变; 2.衰减)=1] 1.153 0.469 6.044 1 0.014 0.234 2.072 [雌孕激素受体分型(1.非luminal型; 2.luminal型)=1] -0.108 0.43 0.063 1 0.802 -0.951 0.735 [腋窝淋巴结(1.转移; 2无转移)=1] -1.095 0.429 6.509 1 0.011 -1.937 -0.254 表 4 N-cadherin表达水平多因素分析
Table 4. Multivariate analysis of N-cadherin expression level
因素 估算 标准错误 瓦尔德 自由度 显著性 95% CI 下限 上限 [边缘毛刺(1.有; 2.无)=1] 0.263 0.386 0.463 1 0.496 -0.494 1.019 [临床分期(1.≤Ⅱ期; 2.≥Ⅲ期)=1] -0.827 0.389 4.507 1 0.034 -1.59 -0.063 表 5 β-catenin表达水平多因素分析
Table 5. Multivariate analysis of β-catenin expression level
因素 估算 标准错误 瓦尔德 自由度 显著性 95% CI 下限 上限 [边缘毛刺(1.有; 2.无)=1] 1.248 0.391 10.17 1 0.001 0.481 2.015 [组织学分级(1.≤Ⅱ级; 2.≥Ⅲ级)=1] -0.034 0.371 0.008 1 0.928 -0.76 0.693 表 6 Vimentin表达水平多因素分析
Table 6. Multivariate analysis of vimentin expression level
因素 估算 标准错误 瓦尔德 自由度 显著性 95% CI 下限 上限 [临床分期(1.≤Ⅱ期; 2.≥Ⅲ期)=1] -1.294 0.505 6.581 1 0.01 -2.283 -0.305 [腋窝淋巴结(1.转移; 2无转移)=1] -0.027 0.439 0.004 1 0.95 -0.889 0.834 -
[1] Zujewski JA, Dvaladze AL, Ilbawi A, et al. Knowledge summaries for comprehensive breast cancer control[J]. J Glob Oncol, 2018(4): 1-7. [2] Böger C, Behrens HM, Röcken C. Ki67: an unsuitable marker of gastric cancer prognosis unmasks intratumoral heterogeneity[J]. J Surg Oncol, 2016, 113(1): 46-54. doi: 10.1002/jso.24104 [3] 李河圣. miR-26b下调EphA2表达抑制原发性肝癌细胞增殖、侵袭和转移的机制研究[D]. 济南: 山东大学, 2016. [4] 弥禹, 魏超群, 葛红岩. 细胞因子对三氧化二砷抑制新生血管形成的作用研究进展[J]. 医学综述, 2020, 26(17): 3399-405. doi: 10.3969/j.issn.1006-2084.2020.17.014 [5] Greenburg G, Hay ED. Epithelia suspended in collagen gels can lose polarity and express characteristics of migrating mesenchymal cells[J]. J Cell Biol, 1982, 95(1): 333-9. doi: 10.1083/jcb.95.1.333 [6] Scimeca M, Trivigno D, Bonfiglio R, et al. Breast cancer metastasis to bone: From epithelial to mesenchymal transition to breast osteoblast-like cells[J]. Semin Cancer Biol, 2021, 72: 155-64. doi: 10.1016/j.semcancer.2020.01.004 [7] Emad A, Ray T, Jensen TW, et al. Superior breast cancer metastasis risk stratification using an epithelial-mesenchymal-amoeboid transition gene signature[J]. Breast Cancer Res, 2020, 22(1): 74. doi: 10.1186/s13058-020-01304-8 [8] Sarrió D, Rodriguez-Pinilla SM, Hardisson D, et al. Epithelialmesenchymal transition in breast cancer relates to the basal-like phenotype[J]. Cancer Res, 2008, 68(4): 989-97. doi: 10.1158/0008-5472.CAN-07-2017 [9] Jørgensen CLT, Forsare C, Bendahl PO, et al. Expression of epithelial-mesenchymal transition-related markers and phenotypes during breast cancer progression[J]. Breast Cancer Res Treat, 2020, 181(2): 369-81. doi: 10.1007/s10549-020-05627-0 [10] 周建桥, 詹维伟. 超声乳腺影像报告数据系统及其解读[J]. 中华医学超声杂志: 电子版, 2011, 8(6): 1332-41. doi: 10.3877/cma.j.issn.1672-6448.2011.06.029 [11] Li X, Lewis MT, Huang J, et al. Intrinsic resistance of tumorigenic breast cancer cells to chemotherapy[J]. JNCI J Natl Cancer Inst, 2008, 100(9): 672-9. doi: 10.1093/jnci/djn123 [12] Mani SA, Guo W, Liao MJ, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells[J]. Cell, 2008, 133(4): 704-15. doi: 10.1016/j.cell.2008.03.027 [13] Pinho SS, Oliveira P, Cabral J, et al. Loss and recovery of Mgat3 and GnT-Ⅲ mediated E-cadherin N-glycosylation is a mechanism involved in epithelial-mesenchymal-epithelial transitions[J]. PLoS One, 2012, 7(3): e33191. DOI: 10.1371/journal.pone.0033191. [14] Liu HY, Zhang XF, Li J, et al. The biological and clinical importance of epithelial-mesenchymal transition in circulating tumor cells[J]. J Cancer Res Clin Oncol, 2015, 141(2): 189-201. doi: 10.1007/s00432-014-1752-x [15] Bruner HC, Derksen PWB. Loss of E-cadherin-dependent cell-cell adhesion and the development and progression of cancer[J]. Cold Spring Harb Perspect Biol, 2018, 10(3): a029330. doi: 10.1101/cshperspect.a029330 [16] Choi Y, Lee HJ, Jang MH, et al. Epithelial-mesenchymal transition increases during the progression of in situ to invasive basal-like breast cancer[J]. Hum Pathol, 2013, 44(11): 2581-9. doi: 10.1016/j.humpath.2013.07.003 [17] Cao ZQ, Wang Z, Leng P. Aberrant N-cadherin expression in cancer [J]. Biomed Pharmacother, 2019, 118: 109320. doi: 10.1016/j.biopha.2019.109320 [18] Bindels S, Mestdagt M, Vandewalle C, et al. Regulation of vimentin by SIP1 in human epithelial breast tumor cells[J]. Oncogene, 2006, 25(36): 4975-85. doi: 10.1038/sj.onc.1209511 [19] Prasad CP, Rath G, Mathur S, et al. Expression analysis of Ecadherin, Slug and GSK3beta in invasive ductal carcinoma of breast [J]. BMC Cancer, 2009, 9: 325. doi: 10.1186/1471-2407-9-325 [20] Yamashita M, Ogawa T, Zhang XH, et al. Role of stromal myofibroblasts in invasive breast cancer: stromal expression of alpha-smooth muscle actin correlates with worse clinical outcome[J]. Breast Cancer, 2012, 19(2): 170-6. doi: 10.1007/s12282-010-0234-5 [21] Tamaki K, Sasano H, Ishida T, et al. The correlation between ultrasonographic findings and pathologic features in breast disorders [J]. Jpn J Clin Oncol, 2010, 40(10): 905-12. http://en.cnki.com.cn/Article_en/CJFDTOTAL-ZHCD201106005.htm [22] Yang Q, Liu HY, Liu D, et al. Ultrasonographic features of triplenegative breast cancer: a comparison with other breast cancer subtypes[J]. Asian Pac J Cancer Prev, 2015, 16(8): 3229-32. doi: 10.7314/APJCP.2015.16.8.3229 [23] 卓家伟, 何以敉, 张美恋, 等. 乳腺癌常规超声及剪切波弹性成像表现与淋巴结转移关系的研究[J]. 中华超声影像学杂志, 2018(8): 709-13. doi: 10.3760/cma.j.issn.1004-4477.2018.08.017 [24] Scimeca M, Bonfiglio R, Menichini E, et al. Microcalcifications drive breast cancer occurrence and development by macrophage-mediated epithelial to mesenchymal transition[J]. Int J Mol Sci, 2019, 20(22): 5633. doi: 10.3390/ijms20225633 [25] 向永涛, 甘兵, 赵亮. 乳腺癌原发灶超声特征与腋窝淋巴结转移的关系[J]. 中国免疫学杂志, 2019, 35(10): 1251-4. doi: 10.3969/j.issn.1000-484X.2019.10.021 [26] 项文静, 于鹏丽, 吴敏. 腋窝淋巴结转移与乳腺癌超声征象的相关性分析[J]. 肿瘤学杂志, 2018, 24(2): 156-9. doi: 10.3969/j.issn.1673-5412.2018.02.022 [27] Yaghjyan L, Stoll E, Ghosh K, et al. Tissue-based associations of mammographic breast density with breast stem cell markers[J]. Breast Cancer Res, 2017, 19(1): 100. doi: 10.1186/s13058-017-0889-3 [28] Guo Q, Dong ZW, Zhang L, et al. Ultrasound features of breast cancer for predicting axillary lymph node metastasis [J]. J Ultrasound Med, 2018, 37(6): 1354-3. doi: 10.1002/jum.14469 [29] Chen SL, Zhang WW, Wang J, et al. The role of axillary lymph node dissection in tubular carcinoma of the breast: a population database study[J]. Med Sci Monit, 2019, 25: 880-7. doi: 10.12659/MSM.913077 [30] Lyons TG. Targeted therapies for triple-negative breast cancer[J]. Curr Treat Options Oncol, 2019, 20(11): 82. doi: 10.1007/s11864-019-0682-x -






 下载:
下载: