Value of DCE-MRI, DTI and their combination in the differential diagnosis of central prostate nodules
-
摘要:
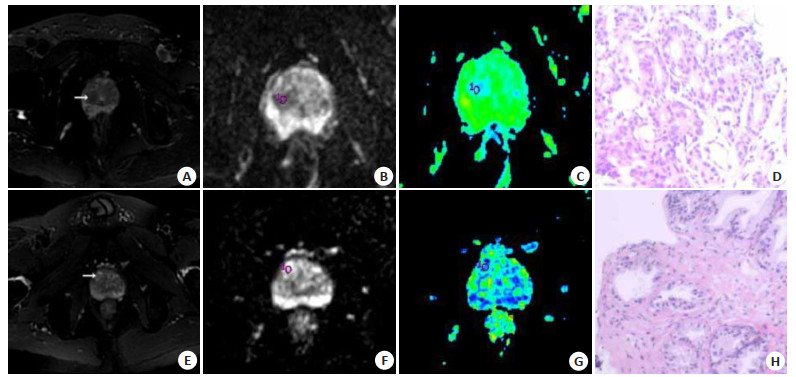
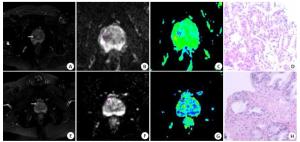
目的探讨MRI动态增强扫描(DCE-MRI)、扩散张量成像(DTI)及两者联合在前列腺中央区结节鉴别诊断中的价值。 方法选取2019年1月~2020年6月在我院就诊的前列腺中央区结节患者79例,共计91个结节,给予DCE-MRI、DTI检查,分析良恶性结节DCE-MRI、DTI参数差异。 结果经病理学诊断,91个结节中,恶性结节39个,良性结节52个;恶性结节DCE-MRI参数峰值时间为103.36±32.50 s,明显低于良性结节(P < 0.05),而强化率和容量转移常数分别为(4.90±1.10)%和12.20±4.11 min-1,明显高于良性结节(P < 0.05);恶性结节时间-强度曲线(TIC)类型Ⅲ比例为84.62%,明显多于良性结节(P < 0.05);良性结节TIC类型Ⅱ比例为80.77%,明显多于恶性结节(P < 0.05);良恶性结节TIC类型Ⅰ比例比较差异无统计学意义(P>0.05);恶性结节DTI参数表观扩散系数值为(1.03±0.22)×10-3 mm2/s,明显低于良性结节(P < 0.05),而各向异性分数为0.32±0.10,明显高于良性结节(P < 0.05);峰值时间、强化率、容量转移常数、TIC类型、表观扩散系数、各向异性分数及联合诊断前列腺中央区恶性结节的ROC曲线下面积分别为0.870、0.883、0.868、0.838、0.903、0.885和0.933(P < 0.05)。 结论DCE-MRI、DTI及联合诊断在前列腺中央区结节鉴别诊断中有较好的应用价值,值得临床使用。 Abstract:ObjectiveTo investigate the value of dynamic contrast-enhanced MRI (DCE-MRI), diffusion tensor imaging (DTI) and their combination in the differential diagnosis of central prostate nodules. MethodsFrom January 2019 to June 2020, 79 patients with central prostate nodules in our hospital, 91 nodules, were examined by DCE-MRI and DTI, and the differences of parameters between benign and malignant nodules were analyzed. ResultsThe pathological diagnosis was made, among the 91 nodules; 39 were malignant and 52 were benign. The Tmax of DCE-MRI parameters of malignant nodules was 103.36 ± 32.50s, which was significantly lower than that of benign nodules (P < 0.05), while the enhancement rate and Ktrans were (4.90± 1.10)% and 12.20±4.11 min-1, which were significantly higher than those of benign nodules (P < 0.05). The proportion of TIC type Ⅲ in malignant nodules was 84.62%, which was significantly higher than that in benign nodules (P < 0.05). The proportion of TIC type Ⅱ in benign nodules was 80.77%, which was significantly higher than that in malignant nodules (P < 0.05). There was no significant difference in the proportion of TIC type Ⅰ between benign and malignant nodules (P>0.05). The ADC value of malignant nodules was (1.03±0.22)×10-3 mm2/s, which was significantly lower than that of benign nodules (P < 0.05), while the FA was 0.32 ± 0.10, which was significantly higher than that of benign nodules (P < 0.05). The area under ROC curve of Tmax, enhancement rate, Ktrans, TIC type, ADC, FA and combined diagnosis of central prostate malignant nodules were 0.870, 0.883, 0.868, 0.838, 0.903, 0.885 and 0.933, respectively (P < 0.05). ConclusionDCE-MRI, DTI and combined diagnosis have good application value in the differential diagnosis of central prostate nodules and is worthy of clinical use. -
表 1 良恶性结节DCE-MRI参数比较
Table 1. Comparison of DCE-MRI parameters of benign and malignant nodules
组别 SImax(%) Tmax(s) R(%) Ktrans(min-1) TIC类型 Ⅰ Ⅱ Ⅲ 恶性(n=39) 142.20±21.65 103.36±32.50 4.90±1.10 12.20±4.11 2(5.13) 4(10.26) 33(84.62) 良性(n=52) 137.69±27.80 144.46±30.15 3.12±1.09 9.89±1.20 3(5.77) 42(80.77) 7(13.46) t/χ2 0.840 -6.224 7.679 3.846 0.000 44.328 45.801 P 0.403 0.000 0.000 0.000 1.000 0.000 0.000 SImax: 强化率; Tmax: 峰值时间; FA: 各向异性分数; Ktrans:容量转移常数. 表 2 良恶性结节DTI参数比较
Table 2. Comparison of DTI parameters of benign and malignant nodules
组别 ADC(×10-3 mm2/s) FA 恶性(n=39) 1.03±0.22 0.32±0.10 良性(n=52) 1.49±0.27 0.17±0.05 t -8.690 9.377 P 0.000 0.000 表 3 DCE-MRI、DTI参数诊断价值
Table 3. Diagnostic Value of DCE-MRI, DTI Parameters
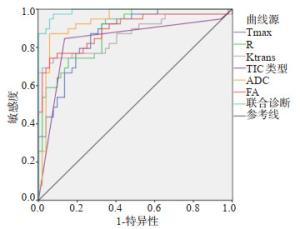
指标 曲线下面积 P 截断值 敏感度(%) 特异性(%) Tmax 0.870 0.000 110.20s 88.00 87.00 R 0.883 0.000 3.98% 88.50 86.90 Ktrans 0.868 0.000 11.50 min-1 87.60 86.20 TIC类型 0.838 0.000 Ⅲ 84.62 86.54 ADC 0.903 0.000 1.15×10-3 mm2/s 89.50 88.00 FA 0.885 0.000 0.27 88.20 87.00 联合诊断 0.933 0.000 - 92.00 89.00 R: 斜率; TIC: 时间-强度曲线; ADC: 表观弥散系数. -
[1] Jae Y. 前列腺癌外科病理新进展[M]. 北京: 人民卫生出版社, 2014. [2] 顾方六. 我国良性前列腺增生和前列腺癌发病调查[J]. 北京大学学报: 医学版, 2000, 32(1): 30-3. doi: 10.3321/j.issn:1671-167X.2000.01.009 [3] 冯亮, 师琳, 蒋益民. 经直肠超声联合前列腺特异抗原密度诊断前列腺癌的临床应用价值研究[J]. 影像科学与光化学, 2020, 38(2): 323-7. https://www.cnki.com.cn/Article/CJFDTOTAL-GKGH202002027.htm [4] 王涛, 马丹丹, 杨明, 等. 经直肠超声引导下徒手经会阴前列腺穿刺在前列腺病变中的应用[J]. 分子影像学杂志, 2020, 43(2): 221-4. doi: 10.12122/j.issn.1674-4500.2020.02.09 [5] 张婷玉, 胡亚萍, 黄晓莹. 动态增强磁共振成像在诊断前列腺癌中的应用观察[J]. 中国药物与临床, 2020, 20(13): 2155-6. https://www.cnki.com.cn/Article/CJFDTOTAL-YWLC202013021.htm [6] 冯虎. DCE-MRI联合DTI检查对前列腺中央区良恶性结节的诊断效能的影响[J]. 现代医用影像学, 2019, 28(4): 766-7. https://www.cnki.com.cn/Article/CJFDTOTAL-XDYY201904019.htm [7] 陈健群, 周秀梅, 孙立辉. MRI动态增强扫描联合DTI对前列腺中央区良恶性结节的诊断价值[J]. 中国中西医结合影像学杂志, 2019, 17(1): 48-51. https://www.cnki.com.cn/Article/CJFDTOTAL-JHYX201901015.htm [8] Sabouri S, Chang SD, Goldenberg SL, et al. Comparing diagnostic accuracy of luminal water imaging with diffusion-weighted and dynamic contrast-enhanced MRI in prostate cancer: a quantitative MRI study[J]. NMR Biomed, 2019, 32(2): e4048. DOI:10.1002/ nbm.4048. [9] 熊青青, 牛翔科, 高月琴, 等. 动态增强MRI鉴别诊断前列腺中央腺体T2WI低信号病变[J]. 中国医学影像技术, 2018, 34(7): 1063-7. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYXX201807036.htm [10] Muglia VF, Reis RB, Rocha TO, et al. Hypoenhancing prostate cancers on dynamic contrast-enhanced MRI are associated with poor outcomes in high-risk patients: results of a hypothesis generating study[J]. Abdom Radiol (NY), 2019, 44(2): 723-31. doi: 10.1007/s00261-018-1771-1 [11] Zhou Y, Wang XL, Xu C, et al. Hepatic pseudolymphoma: imaging features on dynamic contrast-enhanced MRI and diffusion-weighted imaging[J]. Abdom Radiol (NY), 2018, 43(9): 2288-94. doi: 10.1007/s00261-018-1468-5 [12] Peled S, Vangel M, Kikinis R, et al. Selection of fitting model and arterial input function for repeatability in dynamic contrastenhanced prostate MRI [J]. Acad Radiol, 2019, 26(9): e241-51. http://www.sciencedirect.com/science/article/pii/S1076633218304823 [13] Lefrançois P, Zummo-Soucy M, Olivié D, et al. Diagnostic performance of intravoxel incoherent motion diffusion-weighted imaging and dynamic contrast-enhanced MRI for assessment of anal fistula activity[J]. PLoS One, 2018, 13(1): e0191822. doi: 10.1371/journal.pone.0191822 [14] Wei C, Jin BW, Szewczyk-Bieda M, et al. Quantitative parameters in dynamic contrast-enhanced magnetic resonance imaging for the detection and characterization of prostate cancer[J]. Oncotarget, 2018, 9(22): 15997-6007. doi: 10.18632/oncotarget.24652 [15] Zheng N, Li R, Liu WJ, et al. The diagnostic value of combining conventional, diffusion-weighted imaging and dynamic contrastenhanced MRI for salivary gland tumors[J]. Br J Radiol, 2018, 91(1089): 20170707. http://www.ncbi.nlm.nih.gov/pubmed/29902075 [16] Li T, Yu T, Li L, et al. Use of diffusion kurtosis imaging and quantitative dynamic contrast-enhanced MRI for the differentiation of breast tumors [J]. J Magn Reson Imaging, 2018, 48(5): 1358-66. doi: 10.1002/jmri.26059 [17] Ebrahim YGS, Louis MR, Ali E. Multi-parametric dynamic contrast enhanced MRI, diffusion-weighted MRI and proton-MRS in differentiation of benign and malignant breast lesions: Imaging interpretation and radiology-pathology correlation[J]. Egypt J Radiol Nucl Med, 2018, 49(4): 1175-81. doi: 10.1016/j.ejrnm.2018.07.018 [18] Yuan Y, Jiang MD, Wu LZ, et al. Differential diagnostic value of diffusion-weighted and dynamic contrast-enhanced MR imaging in non-cystic lesions in floor of the mouth[J]. Dentomaxillofac Radiol, 2019, 48(3): 20180240. doi: 10.1259/dmfr.20180240 [19] Klawer EME, van Houdt PJ, Pos FJ, et al. Impact of contrast agent injection duration on dynamic contrast-enhanced MRI quantification in prostate cancer [J]. NMR Biomed, 2018, 31(9): e3946. doi: 10.1002/nbm.3946 [20] Li J, He W, Wang J, et al. Quantitative analysis of dynamic contrastenhanced MRI in distinguishing different grades of prostate cancer [J]. Chin J Med Imaging Technol, 2018, 34(6): 901-5. http://www.researchgate.net/publication/331976138_Quantitative_analysis_of_dynamic_contrast-enhanced_MRI_in_distinguishing_different_grades_of_prostate_cancer -






 下载:
下载: