Application of quantitative analysis of contrast-enhanced ultrasound in the evaluation of prognostic factors of breast invasive ductal carcinoma
-
摘要:
目的探讨超声造影定量分析参数在浸润性乳腺癌预后因素评估中的应用价值。 方法对110例浸润性导管乳腺癌进行术前进行灰阶超声造影,并用定量分析QLAB软件分析,生成时间-强度曲线,对周围正常组织与肿瘤组织血流灌注参数进行比较,并分析浸润性乳腺癌超声造影灌注定量参数与预后因素关系。最后分别对各个超声造影定量灌注参数值做ROC曲线分析。 结果与正常组织相比,肿瘤组织峰值强度(PI)明显升高,达峰时间(TTP)、平均渡越时间(MTT)明显短,差异有统计学意义(P < 0.05);上升时间段(RT)相比差异无统计学意义(P>0.05)。与病理分级为高-中分化浸润性乳腺癌患者相比,低分化浸润性乳腺癌患者的RT、TTP明显延长,PI明显升高,差异有统计学意义(P < 0.05),而MTT相比差异无统计学意义(P>0.05)。肿瘤直径>20 mm的浸润性乳腺癌患者的PI明显高于肿瘤直径≤20 mm患者(P < 0.05),而TTP、MTT、RT相比差异无统计学意义。与无淋巴结转移浸润性乳腺癌患者相比,有淋巴结转移患者的PI升高,RT延长,差异有统计学意义(P < 0.05),而TTP、MTT相比差异无统计学意义(P>0.05);ER阴性、PR阴性的浸润性乳腺癌患者PI明显高于ER阳性、PR阳性患者,差异有统计学意义(P < 0.05),而TTP、MTT、RT相比差异无统计学意义(P>0.05);有无Her-2与Ki-67的表达患者的PI、TTP、MTT、RT相比差异无统计学意义(P>0.05)。ROC曲线分析结果显示,PI在ER阴性表达、PR阴性表达鉴别诊断中具有中-低等的诊断效能,PI的截断点均为12.975 dB,标准误分别为0.073(95%CI: 0.562~0.849)、0.074(95%CI: 0.540~0.828),敏感度和特异性分别为77.40%、72.20%,65.20%、66.70%,曲线下面积分别为0.705、0.688。 结论PI、TTP、RT能反映病灶内血流灌注情况,对浸润性乳腺癌患者预后评估有一定的帮助。 Abstract:ObjectiveTo explore the application value of ultrasound contrast quantitative analysis parameters in evaluating prognostic factors of invasive breast cancer. Methods110 cases of invasive ductal breast cancer were pre-operatively performed gray-scale ultrasound contrast. QLAB software was used to analyzed to generate time-intensity curve. The blood perfusion parameters of surrounding normal tissue and tumor tissue were compared. The relationship between quantitative parameters of ultrasound contrast perfusion of invasive breast cancer and prognostic factors were analyzed. Finally, ROC curve analysis was performed on the quantitative perfusion parameters of each ultrasound contrast. ResultsCompared with normal tissues, the peak intensity (PI) of tumor tissues was significantly increased, the peak time (TTP) and the mean transit time (MTT) were significantly shorter, the difference was statistically significant (P < 0.05). There is no statistically significant difference in the rise time (RT) (P>0.05). Compared with patients with high-to-medium differentiated invasive breast cancer, the RT and TTP of patients with poorly differentiated invasive breast cancer were significantly prolonged, and PI was significantly increased. The difference was statistically significant (P < 0.05). Compared with MTT, the difference was not statistically significant (P>0.05). The PI of invasive breast cancer patients with tumor diameter >20 mm was significantly higher than that of patients with tumor diameter ≤20 mm (P < 0.05), and there was no statistically significant difference between TTP, MTT and RT. Compared with patients with invasive breast cancer without lymph node metastasis, patients with lymph node metastasis had elevated PI and prolonged RT. The difference was statistically significant (P < 0.05), while the difference between TTP and MTT was not statistically significant (P>0.05). ER-negative and PR-negative invasive breast cancer patients were significantly higher PI than ER-positive and PR-positive patients, the difference was statistically significant (P < 0.05), but there was no statistically significant difference between TTP, MTT and RT (P>0.05). There is no statistically significant difference between PI-2, TTP, MTT, RT of patients with and without Her-2 expression (P>0.05). ROC curve analysis results show that PI has a medium-low diagnostic efficiency in the differential diagnosis of ER negative expression and PR negative expression. The cutoff point of PI is 12.975dB, and the standard errors are 0.073 (95%CI: 0.562-0.849), 0.074 (95%CI: 0.540-0.828), sensitivity and specificity were 77.40%, 72.20%, 65.20%, 66.70%, and the area under the curve was 0.705, 0.688, respectively. ConclusionPI, TTP, RT can reflect the blood perfusion in the lesion, which is helpful to the prognosis assessment of patients with invasive breast cancer. -
Key words:
- invasive breast cancer /
- contrast-enhanced ultrasound /
- quantitative analysis /
- prognosis
-
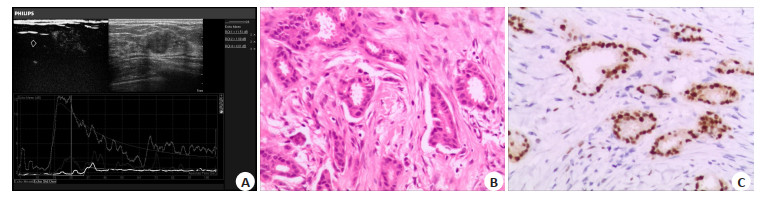
图 1 乳腺癌病灶超声造影时间-强度曲线图和病理、免疫组化图
A: 超声造影图可见高增强、造影后范围扩大; 时间-强度曲线图,灰色曲线代表肿瘤边缘区域,白色曲线代表周围正常乳腺组织; 与周围正常乳腺组织相比, 边缘区域具有快进高增强的特点,峰值强度为12.5 dB, 达峰时间为22.04 s, 平均渡越时间为12.79 s,周围正常乳腺组织峰值强度为4.94 dB, 达峰时间为28.89 s, 平均渡越时间为26.22 s; B: 浸润性乳腺癌病理图(HE染色, ×200), 组织学分级Ⅰ级; C: 免疫组化示ER阳性高表达(SP染色, ×200)
Figure 1. Time-intensity curve of contrast-enhanced ultrasound and pathological and immunohistochemical diagrams of breast cancer lesions
表 1 浸润性乳腺癌边缘带及周围正常组织血流灌注参数对比
Table 1. Comparison of blood perfusion parameters in the marginal zone of invasive breast cancer and surrounding normal tissues (Mean±SD)
组別 PI(dB) RT(s) TTP(s) MTT(s) 肿瘤边缘带 12.302±5.844 4.153±2.869 20.270±9.769 26.222±15.661 正常组织 5.547±4.067 4.855±3.665 30.785±26.362 46.389±29.434 t 5.692 -0.905 -2.244 -3.629 P 0.000 0.368 0.030 0.001 PI: 峰值强度; RT: 上升时间段; TTP: 达峰时间: MTT: 平均渡越时间. 表 2 浸润性乳腺癌超声造影定量参数与预后的关系
Table 2. Relationship between quantitative parameters of contrast-enhanced ultrasound and prognosis of invasive breast cancer (Mean±SD)
预后因素 PI(dB) RT(s) TTP(s) MTT(s) 病灶大小 > 20 mm(n=76) 15.665±4.795 5.484±3.596 19.213±6.840 30.035±15.205 ≤20 mm(n=34) 11.095±5.355 4.346±3.133 18.800±5.274 29.001±20.341 t -3.136 -1.122 -0.220 -0.208 P 0.003 0.267 0.827 0.836 组织学分级 低分化组(n=28) 16.506±4.476 7.009±4.518 21.601±8.341 30.586±18.325 高-中分化组(n=82) 12.775±5.451 3.927±1.832 17.480±4.039 28.332±14.394 t -2.622 -2.974 -2.433 0.477 P 0.011 0.007 0.018 0.635 淋巴结转移 有(n=36) 15.346±4.941 6.391±4.149 20.713±1.353 29.155±21.975 无(n=74) 11.281±4.985 3.797±1.710 17.545±4.360 27.621±12.871 t -2.685 -2.964 -1.613 0.263 P 0.010 0.005 0.114 0.795 ER +(n=69) 12.174±4.994 4.370±2.025 20.041±7.461 24.717±12.693 -(n=41) 15.749±5.205 6.143±4.637 18.371±5.384 33.413±18.630 t 2.540 1.716 0.956 -1.929 P 0.014 0.097 0.343 0.059 PR +(n=57) 11.812±4.093 4.462±2.002 19.122±5.417 26.950±13.961 -(n=53) 15.279±5.639 5.777±4.371 19.172±7.238 33.156±19.475 t 2.215 -1.433 -0.028 0.218 P 0.031 0.160 0.977 0.185 Ki-67 +(n=79) 14.511±5.507 4.793±0.965 18.020±6.231 29.935±17.309 -(n=31) 12.180±4.831 5.087±3.433 18.909±6.090 28.413±14.429 t -0.713 -0.146 -0.245 0.235 P 0.479 0.884 0.808 0.815 Her-2 +(n=42) 14.570±5.314 5.063±3.136 18.597±6.124 27.843±19.717 -(n=68) 11.906±5.010 5.837±4.499 19.739±6.704 26.045±15.455 t 1.480 0.581 0.651 -0.287 P 0.149 0.565 0.518 0.776 表 3 不同参数对浸润性乳腺癌病理预后因素的诊断效能
Table 3. The diagnostic efficacy of different parameters on the pathological prognostic factors of invasive breast cancer
定量参数 峰值强度 达峰时间 上升时间段 病理预测因素 病灶大小(> 20 mm) 组织学分级(低分化) 淋巴结转移(有转移) ER(-) PR(-) 组织学分级(低分化) 组织学分级(低分化) 淋巴结转移(有转移) 截断值 13.155 13.500 13.500 12.975 12.975 18.630 3.460 3.105 敏感度(%) 75.70 81.00 69.00 77.40 72.20 61.90 71.40 75.90 特异性(%) 82.40 60.60 76.50 65.20 66.70 51.50 57.60 58.80 曲线下面积 0.774 0.709 0.738 0.705 0.688 0.620 0.719 0.694 P 0.001 0.010 0.007 0.010 0.031 0.141 0.007 0.030 -
[1] Yan J, Liu XL, Han LZ, et al. Relation between ki-67, ER, PR, Her2/ neu, p21, EGFR, and TOP Ⅱ-α expression in invasive ductal breast cancer patients and correlations with prognosis[J]. Asian Pac J Cancer Prev, 2015, 16(2): 823-9. doi: 10.7314/APJCP.2015.16.2.823 [2] Szabó BK, Aspelin P, Kristoffersen Wiberg M, et al. Invasive breast cancer: correlation of dynamic MR features with prognostic factors [J]. Eur Radiol, 2003, 13(11): 2425-35. doi: 10.1007/s00330-003-2000-y [3] Yaghan R, Stanton PD, Robertson KW, et al. Oestrogen receptor status predicts local recurrence following breast conservation surgery for early breast cancer[J]. Eur J Surg Oncol, 1998, 24(5): 424-6. doi: 10.1016/S0748-7983(98)92341-1 [4] Scholzen T, Gerdes J. The Ki-67 protein: from the known and the unknown[J]. J Cell Physiol, 2000, 182(3): 311-22. doi: 10.1002/(SICI)1097-4652(200003)182:3<311::AID-JCP1>3.0.CO;2-9 [5] Ji CL, Li XL, He YP, et al. Quantitative parameters of contrastenhanced ultrasound in breast invasive ductal carcinoma: The correlation with pathological prognostic factors[J]. Clin Hemorheol Microcirc, 2017, 66(4): 333-45. doi: 10.3233/CH-170251 [6] Zhao YX, Liu S, Hu YB, et al. Diagnostic and prognostic values of contrast-enhanced ultrasound in breast cancer: a retrospective study [J]. Onco Targets Ther, 2017, 10: 1123-9. doi: 10.2147/OTT.S124134 [7] Cao XL, Bao W, Zhu SG, et al. Contrast-enhanced ultrasound characteristics of breast cancer: correlation with prognostic factors [J]. Ultrasound Med Biol, 2014, 40(1): 11-7. doi: 10.1016/j.ultrasmedbio.2013.08.014 [8] Szabó BK, Saracco A, Tánczos E, et al. Correlation of contrastenhanced ultrasound kinetics with prognostic factors in invasive breast cancer [J]. Eur Radiol, 2013, 23(12): 3228-36. doi: 10.1007/s00330-013-2960-5 [9] Wan CF, Du J, Fang H, et al. Enhancement patterns and parameters of breast cancers at contrast-enhanced US: correlation with prognostic factors [J]. Radiology, 2012, 262(2): 450-9. doi: 10.1148/radiol.11110789 [10] Vraka I, Panourgias E, Sifakis E, et al. Correlation between contrastenhanced ultrasound characteristics (qualitative and quantitative) and pathological prognostic factors in breast cancer[J]. In Vivo, 2018, 32(4): 945-54. doi: 10.21873/invivo.11333 [11] Caproni N, Marchisio F, Pecchi A, et al. Contrast-enhanced ultrasound in the characterisation of breast masses: utility of quantitative analysis in comparison with MRI[J]. Eur Radiol, 2010, 20(6): 1384-95. doi: 10.1007/s00330-009-1690-1 [12] Wan C, Du J, Fang H, et al. Evaluation of breast lesions by contrast enhanced ultrasound: qualitative and quantitative analysis[J]. Eur J Radiol, 2012, 81(4): e444-50. http://europepmc.org/abstract/MED/21612882 [13] Zhao HJ, Xu R, Ouyang QF, et al. Contrast-enhanced ultrasound is helpful in the differentiation of malignant and benign breast lesions [J]. Eur J Radiol, 2010, 73(2): 288-93. doi: 10.1016/j.ejrad.2009.05.043 [14] Li XK, Li YQ, Zhu Y, et al. Association between enhancement patterns and parameters of contrast-enhanced ultrasound and microvessel distribution in breast cancer [J]. Oncol Lett, 2018, 15(4): 5643-9. http://europepmc.org/abstract/MED/29556301 [15] Pinder SE, Ellis IO, Galea M, et al. Pathological prognostic factors in breast cancer. Ⅲ. Vascular invasion: relationship with recurrence and survival in a large study with long-term follow-up[J]. Histopathology, 1994, 24(1): 41-7. doi: 10.1111/j.1365-2559.1994.tb01269.x [16] Wang XH, Xu P, Wang Y, et al. Contrast-enhanced ultrasonographic findings of different histopathologic types of breast cancer[J]. Acta Radiol, 2011, 52(3): 248-55. doi: 10.1258/ar.2010.100279 [17] 冷晓玲, 黄国福, 马富成. 乳腺癌不同病灶大小、病理类型及组织学分级的超声造影差异[J]. 中国临床医学影像杂志, 2015, 26(8): 561-5. https://www.cnki.com.cn/Article/CJFDTOTAL-LYYX201508009.htm [18] 张琨, 张雄, 杨晓丽, 等. 乳腺癌超声造影表现与病理组织分型及分级的关系[J]. 南昌大学学报: 医学版, 2014, 54(10): 55-8. https://www.cnki.com.cn/Article/CJFDTOTAL-JXYB201410017.htm [19] Colpaert C, Vermeulen P, van Beest P, et al. Intratumoral hypoxia resulting in the presence of a fibrotic focus is an independent predictor of early distant relapse in lymph node-negative breast cancer patients [J]. Histopathology, 2001, 39(4): 416-25. doi: 10.1046/j.1365-2559.2001.01238.x [20] Olszewski WP, Szumera-Ciećkiewicz A, Piechocki J, et al. The characteristics of the sentinel lymph node metastasis in predicting the axillary lymph node status in patients with breast carcinoma [J]. Pol J Pathol, 2009, 60(3): 138-43. http://www.ncbi.nlm.nih.gov/pubmed/20069508 [21] Putti TC, Abd El-Rehim DM, Rakha EA, et al. Estrogen receptornegative breast carcinomas: a review of morphology and immunophenotypical analysis [J]. Mod Pathol, 2005, 18(1): 26-35. doi: 10.1038/modpathol.3800255 [22] 王婧婕, 胡田, 贾春梅. 乳腺癌超声造影边缘带增强特征及定量参数与其免疫组化指标的相关性分析[J]. 临床超声医学杂志, 2020, 22(5): 331-4. doi: 10.3969/j.issn.1008-6978.2020.05.004 [23] Iwase H, Ando Y, Ichihara S, et al. Immunohistochemical analysis on biological markers in ductal carcinoma in situ of the breast[J]. Breast Cancer, 2001, 8(2): . http://test.europepmc.org/abstract/MED/11342981 [24] Bos R, van Diest PJ, van der Groep P, et al. Expression of hypoxiainducible factor-1alpha and cell cycle proteins in invasive breast cancer are estrogen receptor related[J]. Breast Cancer Res, 2004, 6(4): R450-9. doi: 10.1186/bcr813 [25] Buadu LD, Murakami J, Murayama S, et al. Breast lesions: correlation of contrast medium enhancement patterns on MR images with histopathologic findings and tumor angiogenesis [J]. Radiology, 1996, 200(3): 639-49. doi: 10.1148/radiology.200.3.8756909 [26] Liu gui-feng, Wang zong-qiang, Zhang shu-hua, et al. Diagnostic and prognostic values of contrast-enhanced ultrasound combined with diffusion-weighted magnetic resonance imaging in different subtypes of breast cancer [J]. Int J Mol Med, 2018, 42(1): 105-14. http://europepmc.org/abstract/MED/29620140 [27] Floros KV, Lochmann TL, Hu B, et al. Coamplification of miR-4728 protects HER2-amplified breast cancers from targeted therapy[J]. Proc NatlAcad Sci USA, 2018, 115(11): E2594-603. http://europepmc.org/articles/PMC5856537/ [28] Du R, Zhang H, Shu WW, et al. Correlation between ki-67 expression and hemodynamics of contrast-enhanced ultrasound in patients with breast infiltrative ductal carcinoma[J]. Am Surg, 2018, 84(6): 856-61. doi: 10.1177/000313481808400632 [29] 徐平, 杨敏, 刘勇, 等. C-erbB-2和Ki-67表达阳性及阴性的乳腺癌超声造影定性及定量参数对比观察[J]. 山东医药, 2020, 60(30): 57-9. doi: 10.3969/j.issn.1002-266X.2020.30.014 [30] Hu MZ, Li K. Comparing of two clinical diagnosis by receiver operating characteristic curves[J]. J Mathemat Med, 2005, 18(4): 293-6. -






 下载:
下载: