Value of coronary artery dilatation in prognosis fetal growth restriction
-
摘要:
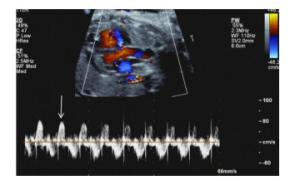
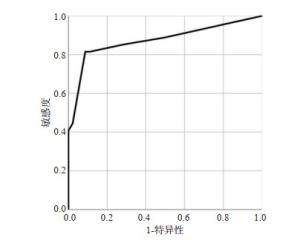
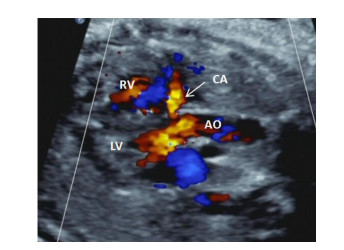
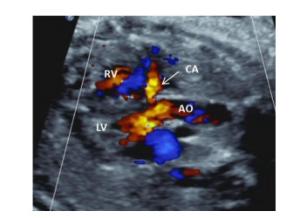
目的探讨冠状动脉扩张在胎儿生长受限(FGR)中的预后评估价值。 方法收集2019年1月~2020年12月我院超声诊断并出生后证实FGR胎儿73例,超声观察冠状动脉(CA)扩张、脐动脉、大脑中动脉(MCA)、静脉导管的血流频谱特征。根据胎儿妊娠结局分为预后良好组和预后不良组,比较胎儿的一般情况及胎儿不同血流异常情况,以超声变量特征建立Logistic回归模型,并绘制受试者工作特征曲线,评价Logistic回归模型对胎儿不良预后的预测价值。 结果FGR胎儿预后不良组27例,预后良好组46例,在脐动脉异常、静脉导管异常、CA扩张组比较差异有统计学意义(P < 0.05),MCA异常组未见明显统计学意义(P>0.05)。Logistic回归分析显示CA扩张、脐动脉异常、静脉导管异常组OR值分别为9.715、4.956、11.291,为胎儿不良预后的独立的危险因素。以CA扩张、脐动脉异常、静脉导管异常3组变量建立的Logistic回归模型预测FGR胎儿不良预后(回归值>0.356)的曲线下面积为0.874,敏感度为81.48%,特异性为91.30%。 结论冠状动脉扩张是FGR预后不良的独立的危险因素。以CA扩张、脐动脉异常、静脉导管异常3个血流异常变量建立的Logistic回归模型能够有效预测FGR不良预后。 Abstract:ObjectiveTo explore the value of coronary artery dilatation in prognosis of fetal growth restriction (FGR). MethodsSeventy-three fetuses with fetal growth restriction (FGR) confirmed by ultrasound in our center from January 2019 to December 2020 were collected. The blood flow spectrum characteristics included coronary artery (CA) dilatation, umbilical artery (UA), middle cerebral artery (MCA) and venous duct (DV) were observed by prenatal ultrasound. The fetuses were divided into two parts: poor prognosis group and good prognosis group according to fetal pregnancy outcome. After comparing the baseline data and blood flow parameters, a logistic regression model based on the characteristics of ultrasonic variableas was established. Receiver operating characteristic curve (ROC) was drawn to evaluate the predictive value of logistic regression model for fetal poor prognosis. ResultsThere were 46 cases in good prognosis group and 27 cases in poor prognosis group. The blood flow parameters including abnormal UA, abnormal DV, CA dilatation had significant differences (P < 0.05), whereas abnormal MCA did not (P>0.05) Logistic regression analysis show that CA dilatation, abnormal UA, and abnormal DV were independent risk factors for fetal poor prognosis(OR values=9.715, 4.956, 11.291, respectively). The Logistic regression model based on CA dilation, abnormal UA and abnormal DV predicted that the area under the curve, the sensitivity and specificity of poor prognosis of FGR were 0.874, 81.8% and 91.30%, respectively. ConclusionCoronary artery dilatation was an independent risk factor for poor prognosis in FGR fetuses. Logistic regression model based on three abnormal blood flow variables, including CA dilation, abnormal UA and abnormal DV could effectively predict the poor prognosis of FGR. -
Key words:
- coronary artery /
- umbilical artery /
- venous duct /
- fetal growth restriction
-
表 1 FGR胎儿不良结局单因素分析
Table 1. Univariate analysis of fetal adverse outcomes in FGR[n(%)]
因素 预后良好组(n=46) 预后不良组(n=27) χ2 P 年龄(岁) 28.50(21~40) 30.00(20~44) 508.50 0.197 首诊孕周(周) 35.50(25.1~39.0) 30.6(23.4~39.2) 325.00 0.001 出生或胎儿死亡孕周(周) 36.60(28.20~39.60) 31.50(24.10~40.10) 315.00 0.001 出生体质量(g) 1950(880~2480) 1050(214~2460) 264.00 0.001 Apgar 1 min 9(8~10) 0(0~9) 88.50 0.001 Apgar 5 min 10(8~10) 0(0~9) 58.00 0.001 Apgar 10 min 10(9~10) 0(0~10) 91.00 0.001 MCA异常 27(0.59) 17(0.63) 0.13 0.719 UA异常 14(0.30) 20(0.74) 13.02 0.001 DV异常 2(0.04) 12(0.44) 17.65 0.001 CA扩张 11(0.24) 22(0.82) 22.76 0.001 MCA: 大脑中动脉; UA: 脐动脉; DV: 静脉导管; CA: 冠状动脉 表 2 FGR胎儿不良结局影响因素的多因素Logistic回归分析结果
Table 2. Multivariate Logistic regression analysis of factors affecting fetal adverse outcomes in FGR
项目 B S.E. Wald值 OR值(95%CI) P UA异常 1.513 0.68 4.956 4.539(1.198~17.193) 0.026 DV异常 2.424 0.942 6.621 11.291(1.782~71.543) 0.01 CA扩张 2.274 0.691 10.827 9.715(2.508~37.636) 0.001 常数项 -3.017 0.702 18.492 - 0.001 -
[1] 中华医学会围产医学分会胎儿医学学组, 中华医学会妇产科学分会产科学组. 胎儿生长受限专家共识(2019版[) J]. 中华围产医学杂志, 2019, 22(6): 361-80. doi: 10.3760/cma.j.issn.1007-9408.2019.06.001 [2] ACOG practice bulletin no. 204 summary: fetal growth restriction [J]. Obstet Gynecol, 2019, 133(2): 390-2. doi: 10.1097/AOG.0000000000003071 [3] Gynecologists ACOOA. ACOG Practice bulletin no. 134: fetal growth restriction[J]. Obstet Gynecol, 2013, 121(5): 1122-33. doi: 10.1097/01.AOG.0000429658.85846.f9 [4] Lausman A, Kingdom J. Maternal fetal medicine committee. Intrauterine growth restriction: screening, diagnosis, and management[J]. J Obstet Gynaecol Can, 2013, 35(8): 741-8. doi: 10.1016/S1701-2163(15)30865-3 [5] Briana DD, Malamitsi-Puchner A. Coronary intimal thickening begins in fetuses: proof of concept for the "fetal origins of adult disease"hypothesis[J]. Angiology, 2020, 71(1): 89. doi: 10.1177/0003319719857384 [6] Guerri-Guttenberg R, Castilla R, Cao G, et al. Coronary intimal thickening begins in fetuses and progresses in pediatric population and adolescents to atherosclerosis[J]. Angiology, 2020, 71(1): 62-9. doi: 10.1177/0003319719849784 [7] Riise HKR, Sulo G, Tell GS, et al. Incident coronary heart disease after preeclampsia: role of reduced fetal growth, preterm delivery, and parity[J]. JAm HeartAssoc, 2017, 6(3): e004158. http://europepmc.org/articles/PMC5523993/ [8] Baschat AA. Fetal responses to placental insufficiency: an update[J]. BJOG, 2004, 111(10): 1031-41. doi: 10.1111/j.1471-0528.2004.00273.x [9] Crispi F, Hernandez-Andrade E, Pelsers MM, et al. Cardiac dysfunction and cell damage across clinical stages of severity in growth-restricted fetuses[J]. Am J Obstet Gynecol, 2008, 199(3): 254. e1-8. http://www.ncbi.nlm.nih.gov/pubmed/18771973 [10] Garcia-Canadilla P, de Vries T, Gonzalez-Tendero A, et al. Structural coronary artery remodelling in the rabbit fetus as a result of intrauterine growth restriction[J]. PLoS One, 2019, 14(6): e0218192. doi: 10.1371/journal.pone.0218192 [11] Bui YK, Howley LW, Ambrose SE, et al. Prominent coronary artery flow with normal coronary artery anatomy is a rare but ominous harbinger of poor outcome in the fetus[J]. J Matern Fetal Neonatal Med, 2016, 29(10): 1536-40. doi: 10.3109/14767058.2015.1057492 [12] Rizzo G, Capponi A, Pietrolucci ME, et al. The significance of visualising coronary blood flow in early onset severe growth restricted fetuses with reverse flow in the ductus venosus[J]. J Matern Fetal Neonatal Med, 2009, 22(7): 547-51. doi: 10.1080/14767050902801777 [13] 刘建君, 郭燕丽, 薛雅方, 等. 产前超声诊断胎儿冠状动脉代偿性扩张[J]. 中国医学影像技术, 2020, 36(7): 1057-60. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYXX202007030.htm [14] Wang Y, Wei J, Liu GL, et al. Effect of regular third-trimester ultrasound examination on antenatal detection and perinatal outcomes of small for gestational age infants[J]. J Int Med Res, 2021, 49(2): 300060521989204. http://www.researchgate.net/publication/349059536_Effect_of_regular_third-trimester_ultrasound_examination_on_antenatal_detection_and_perinatal_outcomes_of_small_for_gestational_age_infants [15] Kanagawa T, Ishii K, Yamamoto R, et al. Fetal outcomes associated with the sequence of Doppler deterioration in severely growthrestricted fetuses[J]. J Ultrasound Med, 2021. DOI: 10.1002/ jum.15614. [16] Afonso S, Bandow GT, Rowe GG. Indomethacin and the prostaglandin hypothesis of coronary blood flow regulation[J]. J Physiol, 1974, 241(2): 299-308. doi: 10.1113/jphysiol.1974.sp010657 [17] Jonker SS, Giraud GD, Chang EI, et al. Coronary vascular growth matches IGF-1-stimulated cardiac growth in fetal sheep[J]. FASEB J, 2020, 34(8): 10041-55. doi: 10.1096/fj.202000215R [18] Davis L, Musso J, Soman D, et al. Role of adenosine signaling in coordinating cardiomyocyte function and coronary vascular growth in chronic fetal Anemia[J]. Am J Physiol Regul Integr Comp Physiol, 2018, 315(3): R500-R508. doi: 10.1152/ajpregu.00319.2017 [19] Cohn HE, Sacks EJ, Heymann MA, et al. Cardiovascular responses to hypoxemia and acidemia in fetal lambs[J]. Am J Obstet Gynecol, 1974, 120(6): 817-24. doi: 10.1016/0002-9378(74)90587-0 [20] Baschat AA, Gembruch U. Evaluation of the fetal coronary circulation[J]. Ultrasound Obstet Gynecol, 2002, 20(4): 405-12. doi: 10.1046/j.1469-0705.2002.00798.x [21] Chaoui R. Coronary arteries in fetal life: physiology, malformations and the "heart-sparing effect"[J]. Acta Paediatr Suppl, 2004, 93 (446): 6-12. http://europepmc.org/abstract/MED/15702664 [22] Baschat AA, Gembruch U, Reiss I, et al. Demonstration of fetal coronary blood flow by Doppler ultrasound in relation to arterial and venous flow velocity waveforms and perinatal outcome-the 'heartsparing effect'[J]. Ultrasound Obstet Gynecol, 1997, 9(3): 162-72. doi: 10.1046/j.1469-0705.1997.09030162.x [23] Fujimori K, Murata Y, Quilligan EJ, et al. Distribution of oxygenated blood flow at three different routes of extracorporeal membrane oxygenation in exteriorized fetal lambs[J]. J Obstet Gynaecol Res, 2001, 27(2): 103-9. doi: 10.1111/j.1447-0756.2001.tb01229.x [24] Reller MD, Morton MJ, Giraud GD, et al. Maximal myocardial blood flow is enhanced by chronic hypoxemia in late gestation fetal sheep[J]. Am J Physiol, 1992, 263(4 Pt 2): H1327-9. http://www.ncbi.nlm.nih.gov/pubmed/1415781 [25] Alfirevic Z, Stampalija T, Dowswell T. Fetal and umbilical Doppler ultrasound in high-risk pregnancies[J]. Cochrane Database Syst Rev, 2017, 6: CD007529. http://pubmedcentralcanada.ca/pmcc/articles/PMC4167858/ [26] Vergani P, Roncaglia N, Ghidini A, et al. Can adverse neonatal outcome be predicted in late preterm or term fetal growth restriction? [J]. Ultrasound Obstet Gynecol, 2010, 36(2): 166-70. doi: 10.1002/uog.7583 [27] Wolf H, Stampalija T, Lees CC, et al. Fetal cerebral blood flow redistribution: an analysis of Doppler reference charts and the association of different thresholds with adverse perinatal outcome [J]. Ultrasound Obstet Gynecol, 2021. DOI: 10.1002/uog.23615. [28] Lees CC, Marlow N, van Wassenaer- Leemhuis A, et al. 2 year neurodevelopmental and intermediate perinatal outcomes in infants with very preterm fetal growth restriction (TRUFFLE): a randomised trial[J]. Lancet, 2015, 385(9983): 2162-72. doi: 10.1016/S0140-6736(14)62049-3 [29] McCowan LM, Harding JE, Roberts AB, et al. A pilot randomized controlled trial of two regimens of fetal surveillance for small-forgestational-age fetuses with normal results of umbilical artery Doppler velocimetry[J]. Am J Obstet Gynecol, 2000, 182(1 pt 1): 81-6. http://europepmc.org/abstract/MED/10649160 [30] Yagel S, Kivilevitch Z, Cohen SM, et al. The fetal venous system, Part Ⅱ: ultrasound evaluation of the fetus with congenital venous system malformation or developing circulatory compromise[J]. Ultrasound Obstet Gynecol, 2010, 36(1): 93-111. doi: 10.1002/uog.7622 [31] Magann EF, Chauhan SP, Hitt WC, et al. Borderline or marginal amniotic fluid index and peripartum outcomes[J]. J Ultrasound Med, 2011, 30(4): 523-8. doi: 10.7863/jum.2011.30.4.523 [32] Baschat AA, Muench MV, Gembruch U. Coronary artery blood flow velocities in various fetal conditions[J]. Ultrasound Obstet Gynecol, 2003, 21(5): 426-9. doi: 10.1002/uog.82 -






 下载:
下载: