Therapeutic effect and influencing factors of combined radiotherapy and chemotherapy on germ cell tumors in basal ganglia
-
摘要:
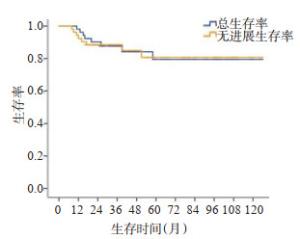
目的研究联合放化疗对基底节区生殖细胞肿瘤的疗效及影响因素。 方法对2009年1月~2019年1月首诊于广东三九脑科医院肿瘤中心的52例基底节区生殖细胞肿瘤患者进行生存分析,将放化疗后存在明显残留与无明显残留的两组患者进行组间比较,以分析影响疗效的临床因素。 结果至2020年6月15日,中位随访时间38月(范围13~125月),随访49例,3例失访。其中40人生存,9例死亡。3年总生存率83%,3年无进展生存率84%。6例患者在接受放化疗后存在明显残留(肿瘤稳定或进展),46例患者无明显残留(部分缓解或完全缓解)。诊断为生殖细胞瘤的患者为30例(57.7%),非生殖细胞瘤性生殖细胞肿瘤患者22例(42.3%)。非生殖细胞瘤性生殖细胞肿瘤较生殖细胞瘤更易出现治疗后残留,两组差异有统计学意义(P=0.013),伴随肿瘤标记物异常增高(甲胎蛋白>500 ng/mL或人绒毛膜促性腺激素>1000 mU/mL)患者较低危组患者更易出现治疗后残留(P= 0.000)。单因素及多因素分析均发现治疗结束时是否存在肿瘤明显残留是影响生存的独立预后因素(P=0.024,HR=0.306,95% CI:0.024~1.488)。 结论基底节区生殖细胞肿瘤随肿瘤类型、肿瘤标记物的不同对治疗反应不同,治疗后存在明显残留是影响生存的独立预后因素。对于明显残留的非生殖细胞瘤性生殖细胞肿瘤建议行再次手术治疗以利于局部控制。 -
关键词:
- 生殖细胞肿瘤 /
- 生殖细胞瘤 /
- 非生殖细胞瘤性生殖细胞肿瘤 /
- 基底节 /
- 预后
Abstract:ObjectiveTo explore the efficacy of combined radiotherapy and chemotherapy on germ cell tumors in the basal ganglia and analysis of influencing factors. MethodsFrom January 2009 to January 2019, 52 cases of germ cell tumors in the basal ganglia were first diagnosed in the Cancer Center of Guangdong Sanjiu Brain Hospital. Survival analysis was performed on the whole group of patients; the residual and non-residual after radiotherapy and chemotherapy were divided to 2 groups. The clinical factors which would affect the efficacy were analyzed between 2 groups. ResultsOn June 15, 2020, the median follow-up time was 38 months (range 13-125 months), 49 cases were followed up, and 3 cases were lost. Of these, 40 survived and 9 died. The 3-year overall survival rate was 83%, and the 3-year progression-free survival rate was 84%. Six patients had significant residues (tumor stability or progression) after receiving chemotherapy and radiotherapy, and 46 patients had no significant residues (partial remission or complete remission). There were 30 patients (57.7%) diagnosed with germ cell tumors, and 22 patients (42.3%) with non-germinomaus germ cell tumors. Non-germinomaus germ cell tumors were more likely to remain after treatment than germ cell tumors. There was a significant statistical difference between the two groups (P=0.013), accompanied by abnormally increased tumor markers (AFP greater than 500 ng/mL or HCG greater than 1000 mU/mL). High risk patients were more likely to have residual after treatment than the low-risk group (P=0.000). Both univariate and multivariate analysis found that the presence or absence of significant tumor residue at the end of treatment was an independent prognostic factor affecting survival, P=0.024 (HR: 0.306, 95%CI: 0.024-1.488). ConclusionGerm cell tumors in the basal ganglia have different response to treatment with different tumor types and tumor markers. Obvious residual after treatment is an independent prognostic factor affecting survival. For the obvious residual non-germinomaus germ cell tumors, surgical treatment is recommended again to facilitate local control. -
Key words:
- germ cell tumors /
- germinoma /
- non-germinomaus germ cell tumors /
- basal ganglia /
- prognosis
-
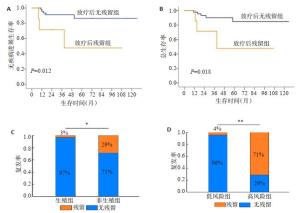
图 3 治疗后是否存在病灶残留是影响无进展生存率及总生存率的独立预后因素(A~B),以及肿瘤种类及是否治疗前存在较高的肿瘤标记物影响治疗后效果(C~D)
Figure 3. Whether there is residual disease after treatment is an independent prognostic factor that affects progression-free survival and overall survival (A-B), and tumor types and with high tumor markers before treatment affect the post-treatment effect (C-D). *P < 0.05; **P < 0.01.
表 1 基底节区治疗后残留组与对照组间患者临床特征差异
Table 1. Clinical characteristics of germ cell tumors in the basal ganglia (n=52)
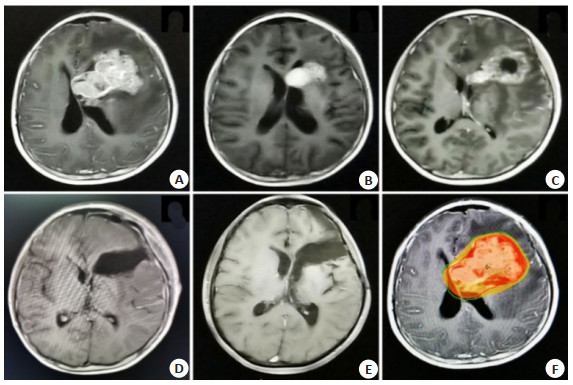
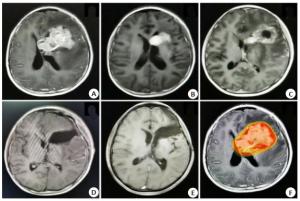
临床特征 明显残留组Δ(n=6) 无明显残留组a(n=46) P 年龄 ≤14岁 6(100) 29(63.0) 0.07 > 14岁 0(0) 17(37) 中位年龄(范围) 9(7~14) 12(5~32) 0.013 性别 男 5(83.3) 40(87.0) 女 1(16.7) 6(13.0) 0.807 肿瘤位置 单侧基底节 5(83.3) 35(76.1) 双侧基底节 1(16.7) 11(23.9) 0.692 诊断 生殖细胞瘤 0(0) 30(65.2) 0.003 非生殖性生殖细胞肿瘤 6(100) 16(34.8) 治疗前血清AFP值(ng/mL) 阴性* 1(16.7) 40(87.0) 0.000 8.78~100 0(0) 3(6.5) > 100 5(83.3) 3(6.5) 均值(范围) 28104(1.25~139000) 1154.1(0~52432.4) 0.005 治疗前血清HCG值(mU/mL) 阴性# 5(83.3) 24(52.2) 0.281 5~100 0(0) 20(43.5) > 100 1(16.7) 2(4.3) 均值(范围) 117(1.2~692.28) 215.2(0~9161.46) 0.572 中位发病时间(月) 2(1~24) 6(1~54) 0.121 放疗情况 中位BED(范围) 72(46.73~89.92) 63.72(42.48~79.47) 0.294 化疗情况 中位化疗疗程(范围) 4(0~8) 6(0~8) 0.096 *AFP血清阴性: 在我院检测值为0~8.78 ng/mL; #HCG血清阴性: 在我院检测值为0~5 mU/mL; Δ明显残留: 首程治疗后疗效为SD及PD; a无明显残留: 首程治疗和疗效为CR及PR. 表 2 影响基底节区生殖细胞肿瘤生存的多因素预后分析
Table 2. Univariate and multivariate survsval analysis of prognostic factors
预后因素 单因素分析 多因素分析 风险率(95%CI) P 风险率(95%CI) P 年龄 ≤14岁vs > 14岁 0.326(0.040~2.658) 0.271 0.616(0.061~6.237) 0.681 病理分型 生殖vs非生殖 0.412(0.098~1.725) 0.210 0.452(0.081~2.522) 0.113 治疗前中枢播散 无播散vs播散 0.601(0.073~4.931) 0.632 0.751(0.073~7.727) 0.809 危险分层 非高危组vs高危组* 0.384(0.077~1.923) 0.027 0.189(0.076~2.622) 0.515 治疗后肿瘤残留 无残留vs有残留 0.206(0.049~0.874) 0.018 0.206(0.024~1.488) 0.024 *高危组: 伴随肿瘤标记物异常增高(AFP > 500 ng/mL或HCG > 1000 mU/mL) -
[1] Matsutani M, Sano K, Takakura K, et al. Primary intracranial germ cell tumors: a clinical analysis of 153 histologically verified cases [J]. J Neurosurg, 1997, 86(3): 446-55. doi: 10.3171/jns.1997.86.3.0446 [2] Nagata K, Nikaido Y, Yuasa T, et al. Germinoma causing wallerian degeneration[J]. J Neurosurg, 1998, 88(1): 126-8. doi: 10.3171/jns.1998.88.1.0126 [3] Nixon J, Brown S, Smith IL, et al. Comparing alternating pressure mattresses and high-specification foam mattresses to prevent pressure ulcers in high-risk patients: the PRESSURE 2 RCT[J]. Health TechnolAssess, 2019, 23(52): 1-176. http://www.researchgate.net/publication/336101799_Comparing_alternating_pressure_mattresses_and_high-specification_foam_mattresses_to_prevent_pressure_ulcers_in_high-risk_patients_the_PRESSURE_2_RCT [4] Allen J, Chacko J, Donahue B, et al. Diagnostic sensitivity of serum and lumbar CSF bHCG in newly diagnosed CNS germinoma[J]. Pediatr Blood Cancer, 2012, 59(7): 1180-2. doi: 10.1002/pbc.24097 [5] Joo JH, Park JH, Ra YS, et al. Treatment outcome of radiation therapy for intracranial germinoma: adaptive radiation field in relation to response to chemotherapy[J]. Anticancer Res, 2014, 34 (10): 5715-21. http://europepmc.org/abstract/med/25275079 [6] Wang YY, Bian HH, Chen W, et al. The diagnosis and treatment of acute, subacute and chronic radiation syndrome from external exposure[J]. Chin J Radiol Health, 2019, 28(4): 355-60. [7] Ozelame RV, Shroff M, Wood B, et al. Basal Ganglia germinoma in children with associated ipsilateral cerebral and brain stem hemiatrophy[J]. Pediatr Radiol, 2006, 36(4): 325-30. doi: 10.1007/s00247-005-0063-4 [8] 孙晓非, 杨群英. 儿童原发中枢神经系统生殖细胞肿瘤多学科诊疗专家共识[J]. 中国小儿血液与肿瘤杂志, 2018, 23(6): 281-6. doi: 10.3969/j.issn.1673-5323.2018.06.001 [9] Fangusaro J, Wu SJ, MacDonald S, et al. Phase Ⅱ trial of responsebased radiation therapy for patients with localized CNS nongerminomatous germ cell tumors: a children's oncology group study[J]. J Clin Oncol, 2019, 37(34): 3283-90. doi: 10.1200/JCO.19.00701 [10] De B, Cahlon O, Dunkel IJ, et al. Reduced-volume radiotherapy for patients with localized intracranial nongerminoma germ cell tumors [J]. J Neurooncol, 2017, 134(2): 349-56. doi: 10.1007/s11060-017-2532-7 [11] National Institute of Cancer. Common Terminology Criteria for Adverse Events (CTCAE) v4.0[EB/OL]. [2020-10-10]. http://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm. [12] 邱晓光, 罗世祺, 马振宇, 等. 28例基底节区生殖细胞瘤诊断性放疗的评价[J]. 中华神经外科杂志, 2006, 22(5): 290-2. doi: 10.3760/j.issn:1001-2346.2006.05.010 [13] Tamaki N, Lin T, Shirataki K, et al. Germ cell tumors of the thalamus and the basal Ganglia[J]. Childs Nerv Syst, 1990, 6(1): 3-7. doi: 10.1007/BF00262257 [14] Liang L, Korogi Y, Sugahara T, et al. MRI of intracranial germ-cell tumours[J]. Neuroradiology, 2002, 44(5): 382-8. doi: 10.1007/s00234-001-0752-0 [15] Villani A, Bouffet E, Blaser S, et al. Inherent diagnostic and treatment challenges in germinoma of the basal ganglia: a case report and review of the literature[J]. J Neurooncol, 2008, 88(3): 309-14. doi: 10.1007/s11060-008-9568-7 [16] Rosenblum MK, Nakazato Y, Matsutani M. WHO Classification of Tumours of the Central Nervous System. 4th rev. ed[M]. Lyon, France: IARC Press, 2016. [17] Souweidane MM, Krieger MD, Weiner HL, et al. Surgical management of primary central nervous system germ cell tumors: proceedings from the second international symposium on central nervous system germ cell tumors[J]. J Neurosurg Pediatr, 2010, 6 (2): 125-30. doi: 10.3171/2010.5.PEDS09112 [18] Calaminus G, Kortmann R, Worch J, et al. SIOP CNS GCT 96: final report of outcome of a prospective, multinational nonrandomized trial for children and adults with intracranial germinoma, comparing craniospinal irradiation alone with chemotherapy followed by focal primary site irradiation for patients with localized disease[J]. Neuro Oncol, 2013, 15(6): 788-96. doi: 10.1093/neuonc/not019 [19] Calaminus G, Bamberg M, Jürgens H, et al. Impact of surgery, chemotherapy and irradiation on long term outcome of intracranial malignant non-germinomatous germ cell tumors: results of the German Cooperative Trial MAKEI 89[J]. Klin Padiatr, 2004, 216 (3): 141-9. doi: 10.1055/s-2004-822626 [20] Fujimaki T. Central nervous system germ cell tumors: classification, clinical features, and treatment with a historical overview[J]. J Child Neurol, 2009, 24(11): 1439-45. doi: 10.1177/0883073809342127 [21] Breen WG, Blanchard MJ, Rao AN, et al. Optimal radiotherapy target volumes in intracranial nongerminomatous germ cell tumors: Long-term institutional experience with chemotherapy, surgery, and dose- and field-adapted radiotherapy[J]. Pediatr Blood Cancer, 2017, 64(11): 26637. doi: 10.1002/pbc.26637 [22] Kim CY, Choi JW, Lee JY, et al. Intracranial growing teratoma syndrome: clinical characteristics and treatment strategy[J]. J Neurooncol, 2011, 101(1): 109-15. doi: 10.1007/s11060-010-0238-1 [23] Goldman S, Bouffet E, Fisher PG, et al. Phase Ⅱ trial assessing the ability of neoadjuvant chemotherapy with or without second-look surgery to eliminate measurable disease for nongerminomatous germ cell tumors: a children's oncology group study[J]. J Clin Oncol, 2015, 33(22): 2464-71. doi: 10.1200/JCO.2014.59.5132 [24] Calaminus G, Frappaz D, Kortmann RD, et al. Outcome of patients with intracranial non-germinomatous germ cell tumors-lessons from the SIOP-CNS-GCT-96 trial[J]. NeuroOncol, 2017, 19(12): 1661-72. -






 下载:
下载: