Correlation between MSCT features and pathological risk in primary duodenal stromal tumor
-
摘要:
目的 分析不同病理危险度的原发性十二指肠间质瘤(PDST)的多层螺旋CT(MSCT)强化表现,以提高PDST的术前不同危险度分级诊断率。 方法 回顾性分析20例经手术病理证实的原发性十二指肠间质瘤的临床及影像资料,包括肿瘤的部位、形态、大小、密度、边界、增强扫描动脉期、静脉期及延迟期特点及多层面重建,依据病理分级分为低危组和高危2组,进行上述征象的秩和检验。 结果 0例PDST中,位于球部3例,降部11例,水平部6例;病变呈囊实性8例,实性12例;病变向腔内生长1例,腔内外生长8例,腔外生长11例。3例极低危险度,9例低危险度,0例中度危险度,8例高度危险度。增强动脉期明显均匀或不均匀强化,13例呈血管样强化,病变越大,强化越不均匀。8例为高危组,大小5.06±1.64 cm;12例为低危组,大小2.83±1.79 cm。低危组的大小、静脉期差值与高危组的差异具有统计学意义(P < 0.05)。 结论 2结论MSCT增强及多层面重建有利于诊断PDST,肿瘤大小及静脉期差值能有效评估原发性PDST危险度分级,低危组与高危组间质瘤在临床特征与CT强化表现上存在差异。 Abstract:Objective To improve the preoperative diagnosis rate of Primary duodenal stromal tumor (PDST) by analyzing the MSCT enhancement of PDST with different pathological risks. Methods The clinical and imaging data of 20 cases confirmed by surgery pathology of primary duodenal stromal tumor were retrospectivly analyzed. The tumor location, shape, size, density, boundary, enhanced scan, arterial and venous phase and delay characteristic were observed. According to the pathological grading into low-risk and high-risk group, the signs of rank and inspection were analyzed. Results Three of the 20 cases with PDST were located in the bulb, 11 cases were descending, and 6 cases were horizontal.The lesions were solid-cystic in 8 cases and solid in 12 cases.The lesions grew into the lumen in 1 case, inside and outside the lumen in 8 cases, and outside the lumen in 11 cases.There were 3 cases of very low risk, 9 cases of low risk, 0 cases of moderate risk, and 8 cases of high risk. At the enhancement stage, the enhancement was obviously uniform or non-uniform, 13 cases presented vascular enhancement, the bigger the lesion and the more non-uniform the enhancement. Eight were in the high-risk group with an average size of 5.06 ± 1.64 cm. 12 were in the low-risk group with an average size of 2.83 ± 1.79 cm. The size and venous difference of the low-risk group were significantly differrent from the high-risk group (P < 0.05). Conclusions MSCT enhancement and multilevel reconstruction are conducive to the diagnosis of PDST. Tumor size and venous phase difference can effectively evaluate the primary PDST risk grading. The clinical characteristics and CT enhancement of the low-risk group and high-risk group of interstitial tumors are different. -
Key words:
- duodenum /
- stromal tumor /
- pathological risk /
- MSCT
-
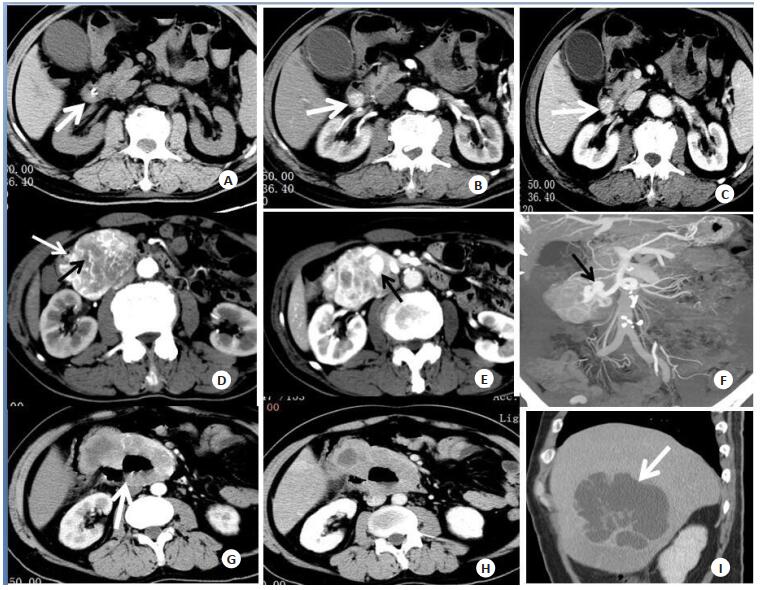
图 1 3位患者十二指肠间质瘤影像表现
A-C: 70岁男性, 极低危险度, 黑便7 d; A:平扫降段等密度结节,平扫易漏诊; B-C:静脉及延迟期腔内结节均匀明显强化; D-F: 71岁男性, 高危险度, 中度贫血, 黑便20 d; D:十二指肠降部外生性肿块, 与内侧壁关系密切(白色箭头), 肿瘤内见迂曲动脉显影(黑色箭头), 肿瘤边界尚清晰, 不均匀强化; E:静脉期显示肿块渐进性强化, 内见静脉瘤形成(黑色箭头); F:最大密度投影显示肿瘤增粗静脉回流于肠系膜上静脉; G-I: 38岁男性, 高危险度, 发现肝占位就诊; G:水平段腔内外占位, 内坏死、积气, 与水平段相通(白色箭头), 动脉期边缘明显强化, 迂曲小动脉显影; H:肿瘤边缘实性成分渐进性强化, 坏死囊变无强化; I:矢状位显示肝脏转移, 呈囊实性占位, 易误诊为肝脓肿.
Figure 1. Imaging findings of primary duodenal stromal tumor of three patients.
表 1 低危险组与高危险组大小及CT三期强化最高与最低值差值
Table 1. Size of low-risk group and high-risk group and the difference between the highest and lowest values of CT third-stage enhancement (Mean±SD)
类别 低危险组(n=12) 高危险组(n=8) P 年龄(岁) 63.08±12.98 54.63±12.02 0.115 大小(cm) 2.83±1.79 5.06±1.64 0.012 平扫(Hu) 38.42±6.73 36.3±3.58 0.473 A-HU* 37.67±23.24 58.37±42.08 0.305 V-HU* 19.58±17.86 38.12±26.88 0.047 D-HU* 13.25±8.44 24.00±15.83 0.057 A-HU*:动脉期差值(软组织最高强化值-软组织最低强化值)CT值;V-HU*:门静脉期差值(软组织最高强化值-软组织最低强化值)CT值; D-HU*:延迟期差值(软组织最高强化值-软组织最低强化值)CT值. -
[1] Emory TS, Sobin LH, Lukes L, et al. Prognosis of gastrointestinal smooth-muscle (stromal) tumors: dependence on anatomic site[J]. Am J Surg Pathol, 1999, 23(1): 82-7. doi: 10.1097/00000478-199901000-00009 [2] Xu XL, Cao ZL, Zhu HH. Capsule endoscopy in the diagnosis of an exophytic gastrointestinal stromal tumor in the small intestine of a young adult woman: a case report[J]. Mol Clin Oncol, 2016, 4(2): 268-70. https://www.ncbi.nlm.nih.gov/pubmed/26893873 [3] 李烦繁, 胡雪阳.晚期肠源性间质瘤临床特征和伊马替尼疗效分析[J].安徽医药, 2016, 20(9): 1739-43. doi: 10.3969/j.issn.1009-6469.2016.09.035 [4] Miettinen M, Kopczynski J, Makhlouf HR, et al. Gastrointestinal stromal tumors, intramural leiomyomas, and leiomyosarcomas in the duodenum: a clinicopathologic, immunohistochemical, and molecular genetic study of 167 cases[J]. Am J Surg Pathol, 2003, 27(5): 625-41. doi: 10.1097/00000478-200305000-00006 [5] 张月浪, 鱼博浪, 李晨霞, 等.小肠间质瘤MSCT征象与病理危险度分级的研究[J].实用放射学杂志, 2011, 27(8): 1171-9. doi: 10.3969/j.issn.1002-1671.2011.08.013 [6] 徐耀, 胡蓉, 候金鹏, 等.小肠间质瘤的MSCT表现与生物学危险度相关分析[J].实用放射学杂志, 2018, 34(7): 1058-62. doi: 10.3969/j.issn.1002-1671.2018.07.019 [7] Lin CW, Chang YT, Zhang Y, et al. Small duodenal gastrointestinal stromal tumor presenting with acute bleeding misdiagnosed as hemobilia: Two case reports[J]. Oncol Lett, 2012, 4(5): 1069-71. http://d.old.wanfangdata.com.cn/OAPaper/oai_pubmedcentral.nih.gov_3499609 [8] Johnston FM, Kneuertz PJ, Cameron JL, et al. Presentation and management of gastrointestinal stromal tumors of the duodenum: a multi-institutional analysis[J]. Ann Surg Oncol, 2012, 19(11): 3351-60. https://www.ncbi.nlm.nih.gov/pubmed/22878613 [9] Kim HC, Lee JM, Son KR, et al. Gastrointestinal stromal tumors of the duodenum: CT and Barium study findings [J]. Am J Roentgenol, 2004, 183(2): 415-9. doi: 10.2214/ajr.183.2.1830415 [10] 戴玲玲, 刘永康, 吕良靓, 等.胰头区域十二指肠间质瘤的影像表现及其与病理的对照研究[J].医学影像学杂志, 2014, 24(5): 791-6. http://d.old.wanfangdata.com.cn/Periodical/yxyxxzz201405029 [11] Xie YB, Du J, Li Q, et al. Treatment and prognosis of patients with duodenal gastrointestinal stromal tumors[J]. National Med J Chin, 2012, 92(24): 1694-7. http://d.old.wanfangdata.com.cn/Periodical/zgptwkzz201710014 [12] 张洪业. MSCT对十二指肠间质瘤的诊断与鉴别诊断价值[J].中国中西医结合影像学杂志, 2014, 12(5): 521-2, 542. doi: 10.3969/j.issn.1672-0512.2014.05.029 [13] Miettinen M, Lasota J. Gastrointestinal stromal tumors-definition, clinical, histological, immunohistochemical, and molecular genetic features and differential diagnosis[J]. J Surg Oncol, 2011, 104(8): 865-73. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ021929681/ [14] 谢齐, 顾乐锋, 郑汉朋, 等.肠系膜间质瘤的CT征象与恶性危险度相关性研究[J].临床放射学杂志, 2015, 34(12): 1934-8. http://d.old.wanfangdata.com.cn/Periodical/lcfsxzz201512020 [15] Kim HC, Kim TK, Sung KB, et al. CT during hepatic arteriography and portography: an illustrative review[J]. Radiographics, 2002, 22(5): 1041-51. doi: 10.1148/radiographics.22.5.g02se071041 [16] 夏国金, 胡珍珍, 何玉麟, 等.原发性小肠间质瘤MSCT强化表现与病理危险度对照分析[J].实用放射学杂志, 2017, 33(6): 571-80. doi: 10.3969/j.issn.1002-1671.2017.06.014 [17] 彭志远, 阳明, 施崇敏, 等. CT与MRI诊断胃肠道外间质瘤的临床分析[J].医学影像学杂志, 2012, 22(9): 1496-9. doi: 10.3969/j.issn.1006-9011.2012.09.027 [18] 张国祥, 杜智, 王毅军, 等.十二指肠间质瘤五例临床分析[J].中华肝胆外科杂志, 2010, 16(11): 839-41. doi: 10.3760/cma.j.issn.1007-8118.2010.11.014 [19] Hou YY, Lu SH, Zhou Y, et al. Predictive values of clinical and pathological parameters for malignancy of gastrointestinal stromal tumors[J]. Histol Histopathol, 2009, 24(6): 737-47. https://digitum.um.es/xmlui/bitstream/10201/36031/1/Predictive%20values%20of%20clinical%20and%20pathological%20parameters%20for%20malignancy%20of%20gastrointestinal%20stromal%20tumors.pdf [20] DeMatteo RP, Lewis JJ, Leung D, et al. Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival[J]. Ann Surg, 2000, 231(1): 51-8. doi: 10.1097/00000658-200001000-00008 [21] Agaimy A, Wünsch PH. Lymph node metastasis in gastrointestinal stromal tumours (GIST) occurs preferentially in young patients < or=40 years: an overview based on our case material and the literature[J]. Langenbecks Arch Surg, 2009, 394(2): 375-81. doi: 10.1007/s00423-008-0449-5 [22] 杨峰, 金忱, 王强, 等.十二指肠间质瘤29例诊治分析[J].中华肝胆外科杂志, 2013, 19(8): 572-3. doi: 10.3760/cma.j.issn.1007-8118.2013.08.005 [23] 雷志毅, 祁明珠, 孟巴音. 27例小肠间质瘤原发及复发转移的CT征象分析[J].医学影像学杂志, 2017, 27(7): 1303-6. http://d.old.wanfangdata.com.cn/Periodical/yxyxxzz201707028 [24] 孙付喜. 128层螺旋CT对小肠淋巴瘤与小肠间质瘤的诊断鉴别研究[J].现代医用影像学, 2018, 27(3): 739-40. http://d.old.wanfangdata.com.cn/Periodical/xdyyyxx201803014 -







 下载:
下载: