Expand resection in glioblastoma under magnetic resonance T2Flair
-
摘要:
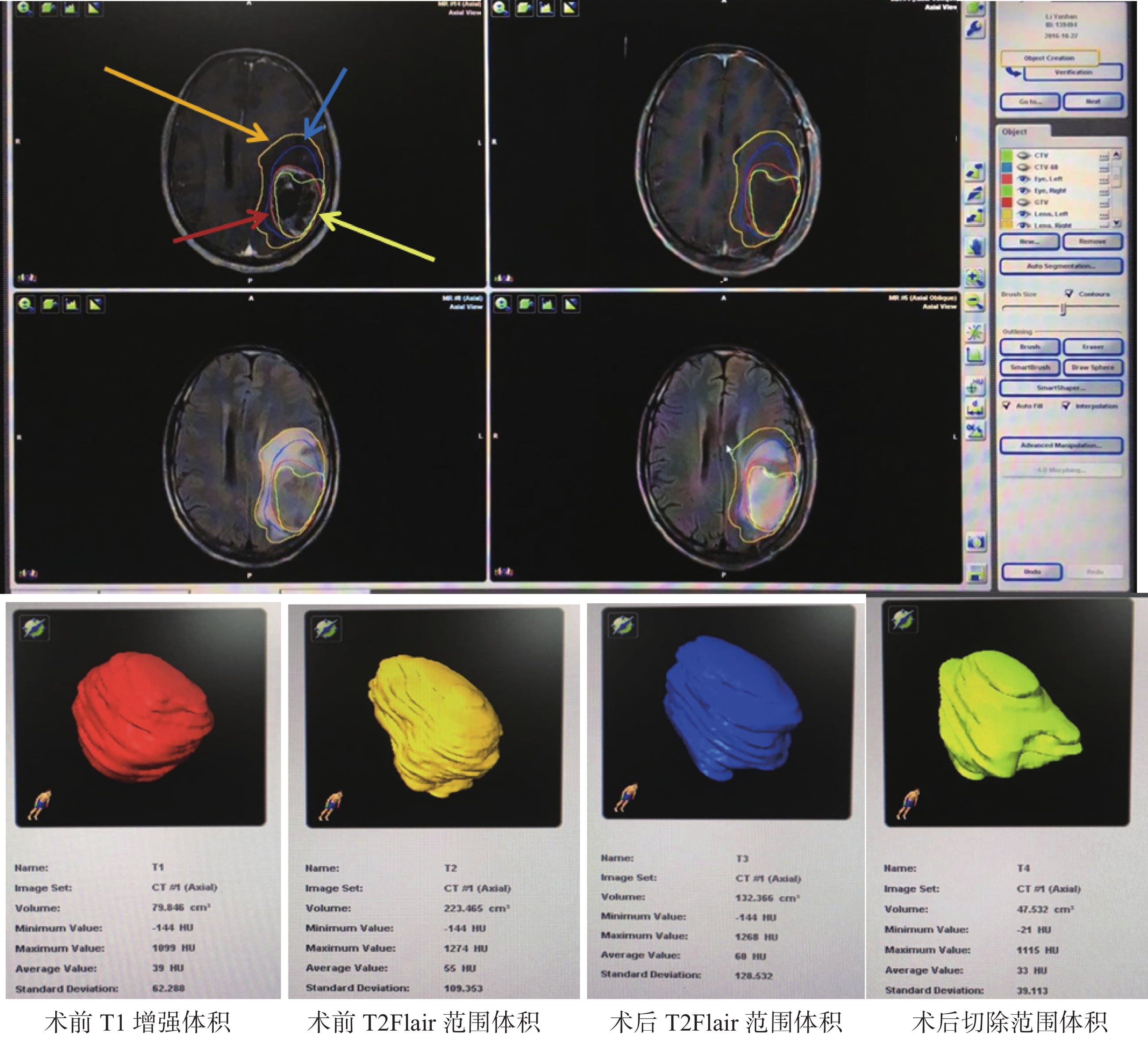
目的 探究在磁共振T2Flair序列范围下扩大切除脑胶质母细胞瘤并辅助放化疗对患者预后的影响。 方法 回顾性分析2010年9月~2016年1月我科41例在唤醒麻醉下进行颅内胶质母细胞瘤患者,其中17例MRI T2Flair序列范围下扩大切除的脑胶质母细胞瘤的患者,剩余24例作对照组为T1增强像下切除的脑胶质母细胞瘤的患者。所有病例手术过程中应用术中神经导航结合DTI、皮层电刺激、术中超声、术中肿瘤荧光造影等多种辅助手段,术后病理均确诊为胶质母细胞瘤(GBM)。采用德国Brainlab放疗计划系统分别勾画出术前、术后MRI T1增强病灶及T2Flair范围,进行图像融合,比较术前术后体积,明确肿瘤T1增强切除范围及T2Flair切除范围,所有患者随访至2017年1月,术后病人均行同步放疗及辅助化疗。 结果 对照组24例病例T1增强病灶均全部切除,术后无患者出现神经功能障碍。另外17例T2Flair像扩大切除患者中,7例T2Flair切除0~10%,4例T2Flair切除10%~25%,6例T2Flair切除25%以上;所有病人随访至2017年1月,17例T2Flair切除GBM患者,中位生存时间为30.45月,对照组24例病例切除GBM患者,中位生存时间为15.37月,两组有统计学差异(χ2=6.16,P=0.013)。T2Flair切除范围0~10%组,10%~25%组,25%以上组,各组中位生存时间分别20.16月,45月,33月,组间无明显统计学差异。 结论 唤醒麻醉下应用术中神经导航结合DTI、皮层电刺激、术中超声、术中肿瘤荧光造影等多种辅助手段有效保障了颅内GBM患者的手术安全性,提高了T1增强像的病灶全切率,可以最大安全范围完成了T2Flair的扩大切除;T2Flair的扩大全切除可明显延长GBM患者的生存期,但并非T2Flair像切除越多越好,切除程度之间无明显生存差异。 Abstract:Objective To explore the effect of extensive resection induced by MRI T2Flair sequence on the prognosis of glioblastoma. Methods Forty-one cases of anesthesia in patients with intracranial glioblastoma between September 2010 and January 2016 in our Hospital were retrospectively analyzed. Seventeen cases were performed with MRI T2Flair sequence under extended resection of brain glioblastoma patients, 24 cases of control group with T1 enhancement were performed with the remaining resection of brain glioblastoma patients. All cases were applied with intraoperative neural navigation application in the process of surgery combined with electrical stimulation DTI, cortex, intraoperative ultrasound, intraoperative tumor fluorescence imaging. Postoperative pathology were diagnosed with glioblastoma (GBM). The preoperative and postoperative MRI T1 enhanced lesions and T2Flair scope were outlined by German making radiotherapy plan system. Image fusion was conducted. The preoperative and postoperative volume were compared. T1 enhance tumor resection range and T2Flair excision scope were definited. The patients were followed up to January 2017. The patient underwent postoperative radiotherapy and chemotherapy. Results T1 enhanced lesions of 24 cases in Control group were excised. No patient had neurological dysfunction after operation. Another 17 cases of T2Flair patients as extended resection, 7 cases T2Flair had a excision of 0%~10%, 4 cases of T2Flair had a resection of 10%~25%, 6 T2Flair removed more than 25%. All patients were followed up to January 2017, 17 patients with GBM T2Flair excision had a median survival time of 30.45 months. In the control group of 24 cases with GBM patients, median survival time was 15.37 months. Difference between two groups was significantly (P=0.013). T2Flair resection were divided into group of 0%~10%, 10%~25% and more than 25%. The median survival time of each group was 20.16, 45, 33 months, respectively, with no significant differences . Conclusion Application of awaken anesthesia combined with intraoperative neural navigation, electrical stimulation DTI, cortex, intraoperative ultrasound, intraoperative tumor fluorescence imaging can effectively guarantee the operation safety of the intracranial GBM patients. It improves the T1 enhanced the rate of total resection of lesions like, promotes the maximum safe range to complete the T2Flair extended resection. T2Flair total excision of the expansion can obviously prolong the survival time of GBM patients, but not T2Flair like to remove the more the better. The difference between the survival degree of resection is not significant. -
Key words:
- glioblastoma /
- T2Flair /
- extended resection /
- awake anesthesia
-
[1] Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2): 115-32. doi: 10.3322/caac.21338 [2] Ostrom QT, Gittleman H, Liao P, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007-2011[J]. Neuro Oncol, 2014, 16(Suppl 4): iv1-63. doi: 10.1093/neuonc/nou223 [3] Schwartzbaum JA, Fisher JL, Aldape KD. Epidemiology and molecular pathology of glioma[J]. Nat Clin Pract Neurol, 2006, 2(9): 494-503. [4] Sanai N, Berger MS. Glioma extent of resection and its impact on patient outcome[J]. Neurosurgery, 2008, 62(4): 753-66. [5] Gorlia T, van den Bent MJ, Hegi ME, et al. Nomograms for predicting survival of patients with newly diagnosed glioblastoma: prognostic factor analysis of EORTC and NCIC trial 26981-22981/CE.3[J]. Lancet Oncol, 2008, 9(1): 29-38. doi: 10.1016/S1470-2045(07)70384-4 [6] 王永志, 杨 帆, 季玉陈, 等. 手术切除程度对胶质母细胞瘤预后预测因素的影响[J].中国微侵袭神经外科杂志,2015(07):289-92. http://www.cnki.com.cn/Article/CJFDTOTAL-ZWQX201507001.htm [7] Pan I W FS, treatment factors associated with survival among adult glioblastoma patients: A USA population-based study from 2000-2010[J]. J Clin Neurosci, 2015, 22(10): 1575-81. [8] Ostrom QT, Gittleman H, Fulop J, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008-2012[J]. Neuro Oncol, 2015, 17(4): 1-62. [9] 张忠, 江涛, 王忠诚. 功能区胶质瘤的手术治疗进展[J]. 中国微侵袭神经外科杂志, 2006(02): 88-90. doi: 10.3969/j.issn.1009-122X.2006.02.019 [10] Mcgirt MJ, Chaichana KL, Gathinji MA, et al. Independent association of extent of resection with survival in patients with malignant brain astrocytoma[J]. J Neurosurg, 2009, 110(1): 156-62. doi: 10.3171/2008.4.17536 [11] Hardesty DA, Sanai N. The value of glioma extent of resection in the modern neurosurgical era[J]. Front Neurol, 2012, 3(6): 140. [12] 马翔宇, 李新钢, 刘佳, 等. 影响胶质母细胞瘤切除术后的预后因素分析[J]. 中华神经外科杂志, 2008, 24(6): 464-7. http://www.cnki.com.cn/Article/CJFDTOTAL-HZZZ201621042.htm [13] Laws ER, Parney IF, Huang W, et al. Survival following surgery and prognostic factors for recently diagnosed malignant glioma: data from the glioma outcomes project[J]. J Neurosurg, 2003, 99(3): 467-73. doi: 10.3171/jns.2003.99.3.0467 [14] Almeida JP, Chaichana KL, Rincon-Torroella J, et al. The value of extent of resection of glioblastomas: clinical evidence and current approach[J]. Curr Neurol Neurosci Rep, 2014, 15(2): 517. [15] 杨伯捷, 吴劲松, 姚成军, 等. 术中MRI监测下行胶质母细胞瘤扩大全切除术的临床研究[J]. 中国临床神经科学, 2010(04): 381-4. doi: 10.3969/j.issn.1008-0678.2010.04.010 [16] De Witte O, Levivier M, Violon P, et al. Quantitative imaging study of extent of surgical resection and prognosis of malignant astrocytomas[J]. Neurosurgery, 1998, 43(2): 398-9. doi: 10.1097/00006123-199808000-00151 [17] Feng C, Yang Y, DM Xiao, et al. Optimizing the extent of resection and minimizing the morbidity in insular high-grade glioma surgery by high-field intraoperative MRI guidance[J]. Turk Neurosurg, 2016, 25(7): 45. [18] 张忠, 江涛, 谢坚, 等. 唤醒麻醉和术中功能定位切除语言区胶质瘤[J]. 中华神经外科杂志, 2007, 23(9): 643-5. http://www.cnki.com.cn/Article/CJFDTOTAL-ZHSW200709006.htm [19] 周辉, 鲁明, 邓心情, 等. 术中唤醒麻醉结合定位技术切除脑深部病灶的临床研究[J]. 中华神经医学杂志, 2015, 14(8): 835-8. http://cpfd.cnki.com.cn/Article/CPFDTOTAL-ZGSK201309001128.htm [20] Grabowski MM, Recinos PF, Nowacki AS, et al. Residual tumor volume versus extent of resection: predictors of survival after surgery for glioblastoma[J]. J Neurosurg, 2014, 121(5): 1115-23. doi: 10.3171/2014.7.JNS132449 [21] 梁进华, 刘湘衡, 白红民, 等. 多形性胶质母细胞瘤的预后影响因素分析[J]. 中华神经外科杂志, 2016, 32(8): 831-5. http://cdmd.cnki.com.cn/Article/CDMD-10089-1013287953.htm -







 下载:
下载: