Effect of HSP90 inhibitor on apoptosis in esophageal squamous carcinoma cell TE-1 and its mechanism
-
摘要:
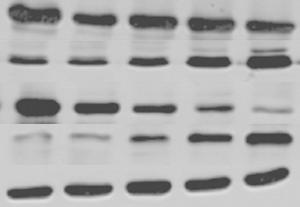
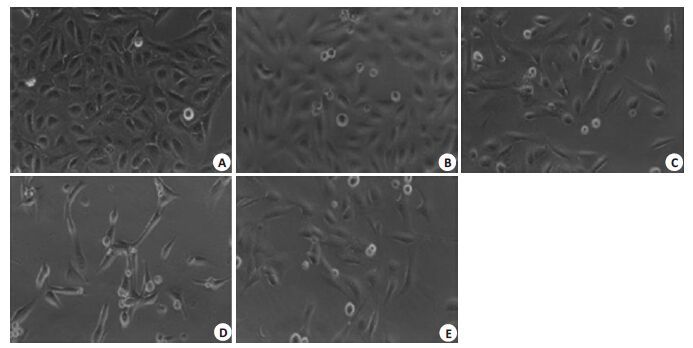
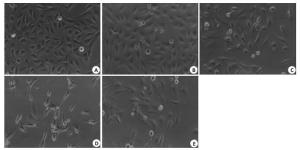
目的探讨抑制热休克蛋白90(Hsp90)对食管鳞癌细胞株TE-1凋亡的影响。 方法采用不同浓度(0.25、0.5、1.0、2.0 μmol/L)、不同时间(24、48、72 h)HSP90抑制剂17-烯丙胺-17-脱甲氧格尔德霉素(17-AAG)处理食管鳞癌细胞株TE-1,分别采用MTT法、流式细胞仪及western blot检测细胞增殖、细胞凋亡及Hsp90、Hsp70、Akt、Fas蛋白表达。 结果17-AAG对TE-1细胞体外增殖抑制率和细胞凋亡率有明显促进作用,并呈现时间-剂量依赖性。17-AAG 处理后TE-1 细胞Hsp90、Akt 蛋白表达明显降低,Hsp70、Fas蛋白表达明显上调。 结论17-AAG可显著抑制TE-1增殖,促进凋亡,其机制可能是通过抑制Hsp90活性、影响其相关信号通路所致。 Abstract:Objective To evaluate the effect of inhibition Heat shock protein 90 (HSP90) on apoptosis in esophageal squamous carcinoma cell TE-1. Methods TE-1 were treated with different time(24 h, 48h or 72 h)and different concentrations(0.25 μmol/L, 0.5 μmol/L, 1.0 μmol/L or 2.0 μmol/L)of 17-allylamio-17-desmethoxygeldanamycin (17-AAG). The cell proliferation, apoptosis and protein expression of Hsp90, Hsp70, Akt and Fas were determined by MTT method, flow cytometry and western blot. Results The 17-AAG on TE-1 cell in vitro proliferation inhibition and apoptosis rate had obvious promoting effect, and time-dose dependent. After curcumin treatment, the protein expression of Hsp90 and Akt significantly reduced, Hsp70 and Fas significantly increased. Conclusion 17-AAG can significantly inhibit the proliferation of TE-1, promote apoptosis, its mechanism may be through inhibiting Hsp90 activity and influence the related signaling pathway. -
Key words:
- HSP90 /
- Esophageal squamous carcinoma /
- 17-AAG /
- Proliferation /
- Apoptosis
-
表 1 不同浓度17-AAG对TE-1增殖抑制率的影响(%,x±s)
Group Concentration of 17-AAG(μmol/L) 24 h 48 h 72 h Control group 0 - - - Experimental group 0.25 0.867±0.021 6.411±1.421* 13.342±2.017*# 0.5 2.534±0.124a 11.357±3.270a* 16.434±3.146a*# 1 4.475±0.529ab 15.321±2.026ab* 17.782±2.588ab*# 2 10.621±0.847abc 22.247±5.024abc* 24.957±3.534abc*# aP<0.05 vs 0.25 μmol/L 17-AAG; bP<0.05 vs 0.5 μmol/L 17-AAG; cP<0.05 vs 1.0 μmol/L 17-AAG; *P<0.05 vs 24 h; #P<0.05 vs 48 h. 表 2 不同浓度17-AAG对TE-1细胞凋亡的影响(x±s)
Group Concentration of 17-AAG(μmol/L) 24 h 48 h 72 h Control group 0 0.32±0.04 0.37±0.05 0.39±0.07 Experimental group 0.25 10.51±2.11Δ 17.41±1.89Δ* 29.36±2.65Δ*# 0.5 19.47±3.24Δa 28.34±2.88Δa* 39.84±3.99Δa*# 1 28.31±3.09Δab 39.25±4.56Δab* 53.15±5.97Δab*# 2 39.94±4.07Δabc 51.14±5.37Δabc* 78.09±8.43Δabc*# ΔP<0.05 vs Control group; aP<0.05 vs 0.25 μmol/L 17-AAG; bP<0.05 vs 0.5 μmol/L 17-AAG; cP<0.05 vs 1.0 μmol/L 17-AAG; *P<0.05 vs 24 h; #P<0.05 vs 48 h. -
[1] Wu C, Kraft P, Zhai K, et al. Genome-wide association analyses of esophageal squamous cell carcinoma in Chinese identify multiple susceptibility loci and gene-environment interactions[J]. Nat Genet, 2012, 44(10): 1090. doi: 10.1038/ng.2411 [2] Tanaka Y, Kamohara H, Kinoshita KA, et al. Clinical impact of serum exosomal microRNA-21 as a clinical biomarker in human esophageal squamous cell carcinoma[J]. Cancer, 2013, 119(6): 1159-67. doi: 10.1002/cncr.27895 [3] Sang J, Acquaviva J, Friedland JC, et al. Targeted inhibition of the molecular chaperone Hsp90 overcomes ALK inhibitor resistance in Non-Small cell lung cancer[J]. Cancer Discov, 2013, 3(4): 430-43. doi: 10.1158/2159-8290.CD-12-0440 [4] Lu X, Xiao L, Wang L, et al. Hsp90 inhibitors and drug resistance in cancer:the potential benefits of combination therapies of Hsp90 inhibitors and other anti-cancer drugs[J]. Biochem Pharmacol, 2012, 83(8): 995-1004. doi: 10.1016/j.bcp.2011.11.011 [5] 赵志敏, 柳晓春, 王秀娥, 等. Hsp90分子抑制剂拮抗肿瘤耐药的研究进展[J]. 现代生物医学进展, 2014(15): 2967-71. http://www.cnki.com.cn/Article/CJFDTOTAL-SWCX201415042.htm [6] Gomez F, Chen L, Wierda W, et al. In vitro effect of AZD1208(Pim Kinase Inhibitor)and 17-AAG(HSP90 Inhibitor)combination in CLL [J]. Cancer Res, 2013, 73(8): 2057-2057. [7] O'malley KJ, Langmann G, Ai J, et al. Hsp90 inhibitor 17‐AAG inhibits progression of LuCaP35 xenograft prostate tumors to castration resistance[J]. Prostate, 2012, 72(10): 1117-23. doi: 10.1002/pros.v72.10 [8] Chen YM, Wang B, Liu D, et al. Hsp90 chaperone inhibitor 17- AAG attenuates a beta-Induced synaptic toxicity and memory impairment [J]. Journal of Neuroscience, 2014, 34(7): 2464-70. doi: 10.1523/JNEUROSCI.0151-13.2014 [9] 宋雪冰. HSP90α在食管鳞癌组织及细胞中的表达及17-AAG对食管鳞癌细胞株的影响[D]. 石家庄: 河北医科大学, 2012. [10] Powers MV, Valenti M, Miranda S, et al. Mode of cell death induced by the HSP90 inhibitor 17-AAG (tanespimycin) is dependent on the expression of pro-apoptotic BAX[J]. Oncotarget, 2013, 4 (11): 1963-75. doi: 10.18632/oncotarget [11] 陈美霓, 许静洪, 赵菊梅. HSP90抑制剂17-AAG对胃癌SGC-7901细胞生长周期和凋亡的影响[J]. 南方医科大学学报, 2013, 33(2): 271-5. http://www.cnki.com.cn/Article/CJFDTOTAL-DYJD201302023.htm [12] 李红梅, 聂丽娟, 霍强, 等. Hsp90 抑制剂槚如酸对乳腺癌 MDA-MB-231细胞增殖、侵袭、迁移的影响[J]. 南方医科大学学报, 2015, 34(3): 355-9. http://youxian.cnki.com.cn/yxdetail.aspx?filename=DYJD20150317007&dbname=CAPJ2015 [13] 刘毅, 王颖, 刘晓丹, 等. 热休克蛋白90抑制剂17-AAG诱导K562 细胞凋亡作用的研究[J]. 中华血液学杂志, 2006, 27(12): 809-12. http://www.cnki.com.cn/Article/CJFDTOTAL-ZHXY200612007.htm -






 下载:
下载: