Quantitative evaluation of hepatic sinusoidal obstruction syndrome using intravoxel incoherent motion imaging
-
摘要:
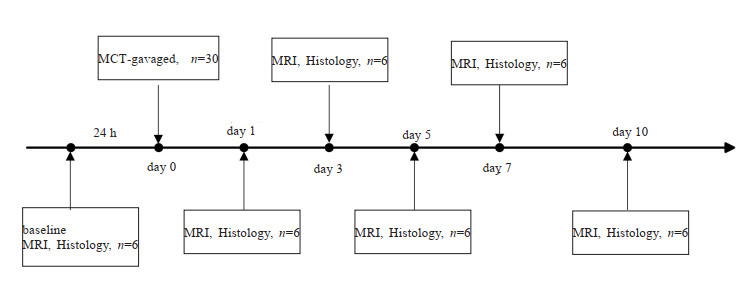
目的 探讨体素内不相干运动(IVIM)MRI定量参数评估野百合碱诱导的大鼠肝窦阻塞综合征(SOS)微循环障碍的可行性。 方法 选取36只SD大鼠,随机分成SOS组(n=30)和基准组(n=6)。SOS组大鼠用野百合碱溶液灌胃,剂量为160 mg/kg,分别在灌胃后第1、3、5、7、10天随机选取6只大鼠行IVIM MRI检查,测量肝脏的水分子扩散(D)、灌注分数(f)、伪扩散系数(D*)、表观扩散系数(ADC),待扫描完成后处死该组大鼠行肝脏取样,测量病理评分。基准组大鼠不做任何干预,在SOS组开始灌胃前1 d行IVIM MRI检查,扫描完成后处死该组大鼠行肝脏病理检查。根据肝纤维化程度将大鼠肝脏分为正常、早期、晚期SOS。分析IVIM MRI定量参数与病理评分及受试者工作特征曲线的相关性。 结果 D、f、D*、ADC的定量参数值在第1、3、5天下降,在第7、10天升高,与病理评分呈负相关关系。与正常肝脏比较,“早期”SOS的D(P < 0.01)、f (P < 0.01)、D*(P < 0.001)、ADC(P < 0.001)均降低,“晚期”SOS的D(P < 0.001)、f (P < 0.001)、D*(P < 0.001)、ADC(P < 0.001)进一步降低。f (r=-0.723)与病理评分的相关系数高于D(r=-0.539)、D*(r=-0.550)和ADC(r=-0.554)。鉴别诊断“早期”SOS和“晚期”SOS时D和f的ROC曲线下面积值(分别为0.8和0.85)高于D*和ADC(分别为0.74和0.73)。 结论 IVIM MRI可以定量评估不同分期SOS肝脏功能和结构的变化,随着SOS损伤的进展,IVIM MRI定量参数出现不同程度的改变,可以为临床监测SOS进展提供重要信息。 Abstract:Objective To investigate the feasibility of introvoxel incoherent motion (IVIM) MRI parameters in evaluating the progression of microcirculatory disturbances in a monocrotaline-induced sinusoidal obstruction syndrome (SOS) rat model. Methods Thirty-six Sprague-Dawley rats were randomly divided into SOS group (n=30) and baseline group (n=6). Rats in SOS group were gavaged with monocrotaline at a dose of 160 mg/kg. Six rats were chosen to perform IVIM MRI scan at 1st, 3rd, 5th, 7th and 10th days after monocrotaline administration, respectively. Pure molecular diffusion (D), perfusion fraction (f), pseudodiffusion (D*), apparent diffusion coefficient (ADC) were measured. After MRI scan, the rats in SOS group were sacrificed for histological evaluation. Rats in baseline group without any intervention, and IVIM MRI was performed one day before MCT administration in SOS group. After the IVIM MRI scan, the rats in baseline group were sacrificed for histological evaluation. The liver samples were categorized into normal, "early", and"late"SOS according to the fibrotic degree. The correlation between MRI parameters and pathological scores as well as receiver operating characteristic curves were analyzed. Results D, f, D* and ADC values decreased on days 1, 3 and 5, increased on days 7 and 10 during SOS progression, and were negatively correlated with pathological scores. Compared with those of normal livers, the D (P < 0.01), f (P < 0.01), D* (P < 0.001) and ADC (P < 0.001) of"early"SOS significantly decreased, and those of"late"SOS significantly decreased (P < 0.001). The correlation coefficient between f (r=-0.732) and pathological score was better than that of D (r=-0.539), D* (r=-0.550), and ADC (r=-0.554). The AUC of D and f (AUC=0.8 and 0.85) were much higher than those of D* and ADC (AUC=0.74 and 0.73) in detecting"late"SOS. Conclusion IVIM MRI analysis can quantitatively characterize the different stages of hepatic SOS. In the progression of SOS injury, IVIM MRI quantitative parameters can provide important information for clinical monitoring of SOS. -
表 1 不同SOS分期下的肝脏MRI量化参数值
Table 1. MRI quantitative parameter values in different SOS stages (Mean±SD)
MRI参数 正常肝脏(n=6) 早期SOS(n=16) 晚期SOS(n=14) P D(mm2/s) 1.0±0.06 0.92±0.04 0.88±0.38 < 0.001 f (%) 25.64±0.79 22.21±1.5 19.29±1.98 < 0.001 D*(mm2/s) 43.78±4.57 26.79±5.27 18.44±3.96 < 0.001 ADC(mm2/s) 1.37±0.04 1.15±0.07 1.08±0.06 < 0.001 SOS: 肝窦阻塞综合征; D: 真扩散系数; f : 灌注分量; D*: 伪扩散系数; ADC: 表观扩散系数. 表 2 病理评分和MRI参数之间的相关系数和95% CI
Table 2. Correlation coefficient and 95%CIs between pathological scores and diffusion parameters
模型 MRI参数 相关系数(95% CI) P IVIM D -0.539 (-0.737, -0.255) < 0.001 f -0.723 (-0.850, -0.518) < 0.001 D* -0.550 (-0.744, -0.270) < 0.001 单指数衰减模型 ADC -0.554 (-0.747, -0.276) < 0.001 表 3 扩散参数诊断不同分期SOS的准确度
Table 3. Diagnostic accuracy of D, f, D* and ADC values for differentiating SOS stages
不同分期及参数 AUC 95% CI 截断值 敏感度(%) 特异性(%) Youden指数 鉴别诊断正常肝脏和SOS D 0.98 0.87~1.00 ≤0.94 96.7 100 0.97 f 0.99 0.88~1.00 ≤24.26 96.7 100 0.97 D* 0.99 0.89~1.00 ≤36.95 96.7 100 0.97 ADC 0.99 0.89~1.00 ≤1.272 96.7 100 0.97 鉴别诊断“晚期”SOS D 0.86 0.69~0.95 ≤0.91 85.7 72.7 0.58 f 0.89 0.74~0.97 ≤22.0 92.9 81.8 0.75 D* 0.81 0.65~0.92 ≤26.47 100 54.6 0.55 ADC 0.81 0.64~0.91 ≤1.164 92.9 54.5 0.47 鉴别诊断“早期”SOS和“晚期”SOS D 0.80 0.62~0.93 ≤0.91 85.7 62.5 0.48 f 0.85 0.67~0.95 ≤21.07 92.9 75.0 0.68 D* 0.74 0.55~0.88 ≤22.52 78.6 62.5 0.41 ADC 0.73 0.54~0.87 ≤1.063 42.9 87.5 0.43 -
[1] Dominique-Charles V, Dominique C-H. Sinusoidal obstruction syndrome[J]. Clin Res Hepatol Gastroenterol, 2016, 40(4): 378-85. doi: 10.1016/j.clinre.2016.01.006 [2] DeLeve LD, Shulman HM, McDonald GB. Toxic injury to hepatic sinusoids: sinusoidal obstruction syndrome (veno-occlusive disease)[J]. Semin Liver Dis, 2002, 22(1): 27-42. doi: 10.1055/s-2002-23204 [3] Rubbia-Brandt L, Audard V, Sartoretti P, et al. Severe hepatic sinusoidal obstruction associated with oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer[J]. Ann Oncol, 2004, 15(3): 460-6. doi: 10.1093/annonc/mdh095 [4] Vauthey JN, Pawlik TM, Ribero D, et al. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases[J]. J Clin Oncol, 2006, 24(13): 2065-72. doi: 10.1200/JCO.2005.05.3074 [5] Ryan P, Nanji S, Pollett A, et al. Chemotherapy-induced liver injury in metastatic colorectal cancer: semiquantitative histologic analysis of 334 resected liver specimens shows that vascular injury but not steatohepatitis is associated with preoperative chemotherapy[J]. Am J Surg Pathol, 2010, 34(6): 784-91. doi: 10.1097/PAS.0b013e3181dc242c [6] Aloia T, Sebagh M, Plasse M, et al. Liver histology and surgical outcomes after preoperative chemotherapy with fluorouracil plus oxaliplatin in colorectal cancer liver metastases[J]. J Clin Oncol, 2006, 24(31): 4983-90. doi: 10.1200/JCO.2006.05.8156 [7] Akıncı B, Atay D, Demir Yenigürbüz F, et al. Therapeutic plasma exchange in pediatric patients with sinusoidal obstruction syndrome/veno-occlusive disease after hematopoietic stem cell transplantation: a single-center experience[J]. Exp Clin Transplant, 2022, 20(7): 680-6. doi: 10.6002/ect.2021.0475 [8] 中华医学会消化病学分会肝胆疾病协作组. 吡咯生物碱相关肝窦阻塞综合征诊断和治疗专家共识意见(2017年, 南京)[J]. 临床肝胆病杂志, 2017, 33(9): 1627-37. doi: 10.3969/j.issn.1001-5256.2017.09.003 [9] Fan CQ, Crawford JM. Sinusoidal obstruction syndrome (hepatic veno-occlusive disease)[J]. J Clin Exp Hepatol, 2014, 4(4): 332-46. doi: 10.1016/j.jceh.2014.10.002 [10] 王露露, 孙璟. 吡咯生物碱导致的肝窦阻塞综合征的诊治进展[J]. 国际消化病杂志, 2020, 40(2): 96-9. doi: 10.3969/j.issn.1673-534X.2020.02.008 [11] Arotçarena R, Calès V, Berthelémy P, et al. Severe sinusoidal lesions: a serious and overlooked complication of oxaliplatin-containing chemotherapy?[J]. Gastroentérologie Clinique Biol, 2006, 30(11): 1313-6. doi: 10.1016/S0399-8320(06)73542-4 [12] Tisman G, MacDonald D, Shindell N, et al. Oxaliplatin toxicity masquerading as recurrent colon cancer[J]. J Clin Oncol, 2004, 22(15): 3202-4. [13] Viganò L, Rubbia-Brandt L, De Rosa G, et al. Nodular regenerative hyperplasia in patients undergoing liver resection for colorectal metastases after chemotherapy: risk factors, preoperative assessment and clinical impact[J]. Ann Surg Oncol, 2015, 22(13): 4149-57. doi: 10.1245/s10434-015-4533-0 [14] Hubert C, Fervaille C, Sempoux C, et al. Prevalence and clinical relevance of pathological hepatic changes occurring after neoadjuvant chemotherapy for colorectal liver metastases[J]. Surgery, 2010, 147(2): 185-94. doi: 10.1016/j.surg.2009.01.004 [15] Ward J, Guthrie JA, Sheridan MB, et al. Sinusoidal obstructive syndrome diagnosed with superparamagnetic iron oxide-enhanced magnetic resonance imaging in patients with chemotherapy-treated colorectal liver metastases[J]. J Clin Oncol, 2008, 26(26): 4304-10. doi: 10.1200/JCO.2008.16.1893 [16] Shin NY, Kim MJ, Lim JS, et al. Accuracy of gadoxetic acid-enhanced magnetic resonance imaging for the diagnosis of sinusoidal obstruction syndrome in patients with chemotherapy-treated colorectal liver metastases[J]. Eur Radiol, 2012, 22(4): 864-71. doi: 10.1007/s00330-011-2333-x [17] Han NY, Park BJ, Sung DJ, et al. Chemotherapy-induced focal hepatopathy in patients with gastrointestinal malignancy: gadoxetic acid: enhanced and diffusion-weighted MR imaging with clinical-pathologic correlation[J]. Radiology, 2014, 271(2): 416-25. doi: 10.1148/radiol.13131810 [18] Donati F, Cioni D, Guarino S, et al. Chemotherapy-induced liver injury in patients with colorectal liver metastases: findings from MR imaging[J]. Diagnostics (Basel), 2022, 12(4): 867. doi: 10.3390/diagnostics12040867 [19] Le Bihan D, Breton E, Lallemand D, et al. MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders[J]. Radiology, 1986, 161(2): 401-7. doi: 10.1148/radiology.161.2.3763909 [20] Chow AM, Gao DS, Fan SJ, et al. Liver fibrosis: an intravoxel incoherent motion (IVIM) study[J]. J Magn Reson Imaging, 2012, 36(1): 159-67. doi: 10.1002/jmri.23607 [21] Baliyan V, Das CJ, Sharma R, et al. Diffusion weighted imaging: technique and applications[J]. World J Radiol, 2016, 8(9): 785-98. doi: 10.4329/wjr.v8.i9.785 [22] Leitão HS, Doblas S, Garteiser P, et al. Hepatic fibrosis, inflammation, and steatosis: influence on the MR viscoelastic and diffusion parameters in patients with chronic liver disease[J]. Radiology, 2017, 283(1): 98-107. doi: 10.1148/radiol.2016151570 [23] Lefebvre T, Hébert M, Bilodeau L, et al. Intravoxel incoherent motion diffusion-weighted MRI for the characterization of inflammation in chronic liver disease[J]. Eur Radiol, 2021, 31(3): 1347-58. doi: 10.1007/s00330-020-07203-y [24] Yang JX, Meng MZ, Pan CJ, et al. Intravoxel incoherent motion and dynamic contrast-enhanced magnetic resonance imaging to early detect tissue injury and microcirculation alteration in hepatic injury induced by intestinal ischemia-reperfusion in a rat model[J]. J Magn Reson Imaging, 2021, 54(3): 751-60. doi: 10.1002/jmri.27604 [25] DeLeve LD, McCuskey RS, Wang X, et al. Characterization of a reproducible rat model of hepatic veno-occlusive disease[J]. Hepatology, 1999, 29(6): 1779-91. doi: 10.1002/hep.510290615 [26] Hong EK, Joo I, Park J, et al. Assessment of hepatic sinusoidal obstruction syndrome with intravoxel incoherent motion diffusion-weighted imaging: an experimental study in a rat model[J]. J Magn Reson Imaging, 2020, 51(1): 81-9. doi: 10.1002/jmri.26790 [27] Lyu J, Yang GX, Mei YJ, et al. Non-Gaussian diffusion models and T1 rho quantification in the assessment of hepatic sinusoidal obstruction syndrome in rats[J]. J Magn Reson Imaging, 2020, 52(4): 1110-21. doi: 10.1002/jmri.27156 [28] Narita M, Hatano E, Tamaki N, et al. Dai-Kenchu-to attenuates rat sinusoidal obstruction syndrome by inhibiting the accumulation of neutrophils in the liver[J]. J Gastroenterol Hepatol, 2009, 24(6): 1051-7. doi: 10.1111/j.1440-1746.2009.05795.x [29] Deleve LD, Wang XD, Tsai J, et al. Sinusoidal obstruction syndrome (veno-occlusive disease) in the rat is prevented by matrix metalloproteinase inhibition[J]. Gastroenterology, 2003, 125(3): 882-90. doi: 10.1016/S0016-5085(03)01056-4 [30] Prié S, Stewart DJ, Dupuis J. EndothelinA receptor blockade improves nitric oxide-mediated vasodilation in monocrotaline-induced pulmonary hypertension[J]. Circulation, 1998, 97(21): 2169-74. doi: 10.1161/01.CIR.97.21.2169 [31] Panagiotaki E, Walker-Samuel S, Siow B, et al. Noninvasive quantification of solid tumor microstructure using VERDICT MRI[J]. Cancer Res, 2014, 74(7): 1902-12. doi: 10.1158/0008-5472.CAN-13-2511 [32] Guo CW, Shen SD, Zhang Y, et al. Determination of apparent diffusion coefficient to quantitatively study partial hepatic ischemia reperfusion injury in a rabbit model[J]. Transplant Proc, 2011, 43(5): 1474-9. doi: 10.1016/j.transproceed.2011.01.183 -







 下载:
下载: