Preliminary application of neuronal orientation dispersion and density imaging in preoperative grading of glioma
-
摘要:
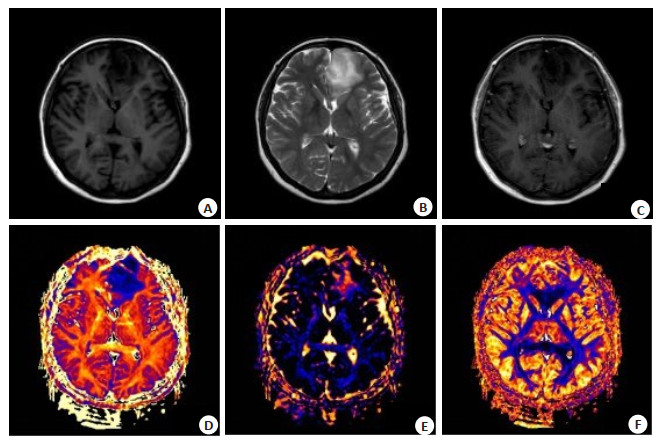
目的运用神经突方向的离散度与密度成像(NODDI)技术参数定量评估高、低级别脑胶质瘤的差异性,探讨NODDI技术在脑胶质瘤疾病术前诊断和分级上的应用价值。 方法选取术后确诊为胶质瘤的27例患者,术前行常规平扫、增强以及高级扩散扫描,经后处理软件得到神经突方向离散度(ODI)、神经突内体积分数(FICVF)及各向同性体积分数(VISO)的图像,分别测量肿瘤实性的区域、瘤周水肿的区域及对侧正常的脑白质区各参数值的平均值。并通过受试者工作特征(ROC)曲线进行参数比较分析。 结果高级别胶质瘤组肿瘤实质区域FICVF、VISO均高于低级别组,差异有统计学意义(P < 0.05)。高级别胶质瘤组瘤周水肿区域FICVF、ODI、VISO均高于低级别胶质瘤组,差异无统计学意义。肿瘤实性区域ROC曲线下面积FICVF、ODI、VISO分别为0.82(P < 0.05)、0.71(P < 0.05)、0.61(P>0.05)。 结论NODDI参数FICVF、ODI、VISO均可以用来鉴别高低级别胶质瘤,且FICVF的诊断效能最高,VISO效能最低。 -
关键词:
- 神经突起方向离散度与密度成像 /
- 脑胶质瘤 /
- 分级
Abstract:ObjectiveTo explore the application value of neuronal orientation dispersion and density imaging (NODDI) technology in preoperative diagnosis and classification of glioma disease, by using NODDI parameters to quantitatively evaluate the difference between high grade glioma and low grade glioma. MethodsTwenty-seven postoperative patients diagnosed with glioma were selected as the research subjects. Preoperative routine plain scan, enhanced scan, and advanced diffusion scan were performed. Images of neuronal orientation dispersion index (ODI), intracellular volume fraction (FICVF), volume fraction of the isotropic compartment (VISO) were obtained by the poster processing soft. The average values of various parameters of tumor parenchymal area, peritumor edema area and contralateral normal brain white matter area were measured respectively. Comparative analysis of parameters was performed using the receiver operating characteristic (ROC) curve. ResultsFICVF and VISO values in tumor parenchymal area of the high grade glioma group were higher than that of low grade glioma group, and the differences were statistically significant. FICVF, ODI and VISO values in peritumor edema area of the high grade glioma group were higher than that of low grade glioma group, and the differences were statistically significant. The FICVF, ODI and VISO values under the ROC curve of the tumor parenchymal area were 0.82 (P < 0.05), 0.71 (P < 0.05) and 0.61 (P>0.05), respectively. ConclusionNODDI parameters including FICVF, ODI and VISO can all be used to identify high and low grade glioma, and FICVF has the highest diagnostic performance, while VISO has the lowest performance. -
Key words:
- neuronal orientation dispersion and density imaging /
- glioma /
- grading
-
表 1 高、低级别胶质瘤肿瘤实质区域参数值
Table 1. Parameter values of parenchymal regions in high-grade and low-grade gliomas
患病 FICVF实质 ODI实质 VISO实质 低级別胶质瘤 0.349(0.325, 0.615) 0.327(0.267, 0.496) 0.165(0.126, 0.179) 高级別胶质瘤 0.468(0.425, 0.509) 0.342(0.301, 0.53) 0.209(0.173, 0.286) Z -1.974 -0.74 -2.418 P 0.048 0.459 0.016 FICVF: 神经突内体积分数; ODI: 神经突方向离散度; VISO: 各项同性体积分数. 表 2 高、低级别胶质瘤瘤周水肿区域参数值
Table 2. Parameter values of peritumoral edema regions in high-grade and low-grade gliomas
患病 FICVF瘤周 ODI瘤周 VISO瘤周 低级別胶质瘤 0.34(0.277, 0.674) 0.350(0.252, 0.544) 0.178(0.132, 0.189) 高级別胶质瘤 0.548(0.363, 0.853) 0.370(0.269, 0.672) 0.202(0.16, 0.218) Z -1.776 -0.493 -1.283 P 0.076 0.622 0.199 表 3 胶质瘤肿瘤实质参数值对患病的诊断ROC分析
Table 3. ROC analysis of parenchymal parameter values for diagnosis of gliomas
检验结果变量 AUC面积 标准误 P 95%CI 临界值 敏感度 特异度 下限 上限 FICVF实质 0.818 0.061 0.000 0.699 0.936 0.32405 92.60% 66.70% ODI实质 0.711 0.071 0.008 0.572 0.849 0.2805 77.80% 55.60% VISO实质 0.613 0.078 0.154 0.461 0.766 0.15885 70.40% 63.00% -
[1] Li XQ, Ouyang ZG, Zhang SH, et al. Synergistic inhibition of angiogenesis and glioma cell-induced angiogenesis by the combination of temozolomide and enediyne antibiotic lidamycin[J]. Cancer Biol Ther, 2014, 15(4): 398-408. doi: 10.4161/cbt.27626 [2] Ostrom QT, Gittleman H, Truitt G, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2011-2015[J]. Neuro Oncol, 2018, 20(suppl4): 1-86. [3] Huang J, Liu FK, Liu ZX, et al. Immune checkpoint in glioblastoma: promising and challenging[J]. Front Pharmacol, 2017, 8: 242. doi: 10.3389/fphar.2017.00242 [4] Zhang H, Schneider T, Wheeler-Kingshott CA, et al. NODDI: Practical in vivo neurite orientation dispersion and density imaging of the human brain[J]. NeuroImage, 2012, 61(4): 1000-16. doi: 10.1016/j.neuroimage.2012.03.072 [5] 付修威, 倪红艳. 神经突方向离散度和密度成像的原理及其在中枢神经系统的研究进展[J]. 国际医学放射学杂志, 2020, 43(1): 68-72. https://www.cnki.com.cn/Article/CJFDTOTAL-GWLC202001018.htm [6] Vellmer S, Tonoyan AS, Suter D, et al. Validation of DWI preprocessing procedures for reliable differentiation between human brain gliomas[J]. Z Med Phys, 2018, 28(1): 14-24. doi: 10.1016/j.zemedi.2017.04.005 [7] Zhao J, Li JB, Wang JY, et al. Quantitative analysis of neurite orientation dispersion and density imaging in grading gliomas and detecting IDH- 1 gene mutation status[J]. Neuroimage Clin, 2018, 19: 174-81. doi: 10.1016/j.nicl.2018.04.011 [8] Li SH, Jiang RF, Zhang J, et al. Application of neurite orientation dispersion and density imaging in assessing glioma grades and cellular proliferation[J]. World Neurosurg, 2019, 131(11): e247-54. http://www.sciencedirect.com/science/article/pii/S1878875019320273 [9] 张蕊, 白岩, 魏巍, 等. 体素内不相干运动成像和扩散峰度成像基本原理及其在中枢神经系统疾病中的应用[J]. 磁共振成像, 2020, 11 (9): 804-8. https://www.cnki.com.cn/Article/CJFDTOTAL-CGZC202009021.htm [10] 索学玲, 龚启勇. DWI技术在中枢神经系统中的应用及研究进展[J]. 放射学实践, 2018, 33(2): 210-4. https://www.cnki.com.cn/Article/CJFDTOTAL-FSXS201802030.htm [11] Duc NM. The role of diffusion tensor imaging metrics in the discrimination between cerebellar medulloblastoma and brainstem glioma[J]. Pediatr Blood Cancer, 2020, 67(9): e28468. doi: 10.1002/pbc.28468 [12] 王宁, 张卫东. 扩散峰度成像的基本原理及其在脑肿瘤中的应用[J]. 国际医学放射学杂志, 2018, 41(5): 558-62, 567. https://www.cnki.com.cn/Article/CJFDTOTAL-GWLC201805014.htm [13] 王婧妍, 初建平, 赵静, 等. NODDI在脑胶质瘤分级中的初步探讨[J]. 放射学实践, 2018, 33(7): 664-7. https://www.cnki.com.cn/Article/CJFDTOTAL-FSXS201807005.htm [14] Kamiya K, Hori M, Irie R, et al. Diffusion imaging of reversible and irreversible microstructural changes within the corticospinal tract in idiopathic normal pressure hydrocephalus[J]. Neuroimage Clin, 2017, 11(14): 663-71. http://europepmc.org/articles/PMC5358533/ [15] 王小峰, 王琪, 杜春亮, 等. 胶质瘤瘤周水肿的诊断与治疗[J]. 中国神经精神疾病杂志, 2019, 45(3): 190-2. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSJJ201903020.htm [16] Wu CX, Lin GS, Lin ZX, et al. Peritumoral edema on magnetic resonance imaging predicts a poor clinical outcome in malignant glioma[J]. Oncol Lett, 2015, 10(5): 2769-76. doi: 10.3892/ol.2015.3639 [17] 刘伟星, 陆鹏, 张晓斌, 等. 磁共振神经突方向离散度与密度成像对帕金森病患者壳核病变的临床应用[J]. 磁共振成像, 2020, 11(8): 610-4. https://www.cnki.com.cn/Article/CJFDTOTAL-CGZC202008005.htm -







 下载:
下载: