Clinical application of MR Neurite orientation dispersion and density imaging technique to the changes of cerebellar dentate nucleus in patients with Parkinson's disease
-
摘要:
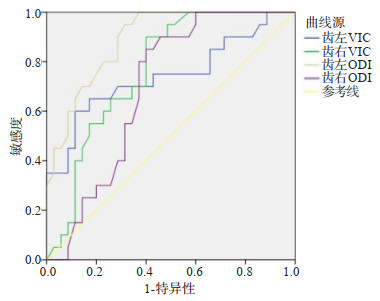
目的探讨利用轴突定向弥散和密度成像(NODDI)对帕金森病患者小脑微结构变化的临床应用价值。 方法选择我院35例帕金森病患者且进行NODDI检查的为帕金森病组,选取健康者20例为对照组。根据NODDI扫描结果获取相关参数取向弥散指数(ODI)、细胞内体积分数(VIC)和各向同性隔室的体积分数(VISO)值,分析两组感兴趣区ODI、VIC及VISO值的组间差异,以ROC曲线评估相关参数对帕金森病是否有一定的临床诊断价值。 结果帕金森病患者左、右侧齿状核、小脑白质的VIC值,及左、右侧齿状核的ODI值与对照组相比显示减低(P < 0.05),帕金森病患者左、右侧小脑白质的ODI值与对照组相比显示增加(P < 0.05)。ROC曲线显示,齿状核左、右侧VIC诊断帕金森病患者的AUC为0.743、0.767,齿状核左、右侧ODI诊断帕金森病患者的AUC为0.891、0.694。 结论NODDI可以反映帕金森病患者小脑微观结构的变化,其参数值可以作为帕金森病患者的评估指标。 -
关键词:
- 帕金森病 /
- 轴突定向弥散和密度成像 /
- 齿状核 /
- 小脑白质
Abstract:ObjectiveTo explore the clinical value of Neurite orientation dispersion and density imaging (NODDI) in cerebellar microstructural changes in patients with Parkinson's disease (PD). MethodsTwenty healthy persons and 35 patients with Parkinson's disease (PD) were selected as case group and control group respectively. According to the relevant parameters of NODDI scanning results, orientation dispersion index (ODI), intracellular volume fraction (VIC) and isotropic volume fraction (VISO) value, ROC curve was used to evaluate whether the related parameters had certain clinical diagnostic value for Parkinson's disease. ResultsThe VIC and ODI values of dentate nucleus, cerebellar white matter and dentate nucleus in PD patients were significantly lower than those in healthy controls (P < 0.05). The ODI values of cerebellar white matter in PD patients were higher than that in the controls (P < 0.05). At the same time, ROC curve showed that the AUC of patients with PD diagnosed by VIC on the left and right side of dentate nucleus were 0.743 and 0.767, and the AUC of patients with PD diagnosed by ODI on the left and right side of dentate nucleus were 0.891 and 0.694, respectively. ConclusionNODDI can reflect the changes of cerebellar microstructure in patients with PD, and its parameters can be used as an evaluation index for patients with PD. -
图 2 男,57岁,PD患者,左上肢不自主抖动2年余
A~C: 常规序列T1WI图、T2WI图、T2压水图; D~F: NODDI技术得到的参数图; D: VIC图轴位,测得患者双侧齿状核、小脑白质VIC值与健康对照组相比下降; E: ODI图轴位,测得患者双侧齿状核ODI值与健康对照组相比下降, 而双侧小脑白质ODI值与正常对照组相比较增加; F: Viso图轴位,测得患者双侧齿状核与小脑白质Viso值与正常对照组相比无明显差异.
Figure 2. A 57-year-old male PD patient, The left upper limb has been shaking involuntarily for more than 2 years.
表 1 PD患者与对照组小脑齿状核及白质扩散参数的比较
Table 1. Comparison of cerebellar dentate nucleus and white matter diffusion parameters between patients with PD and control group (Mean±SD)
指标 对照组(n=20) PD组(n=35) t P 齿状核 左侧VIC值 0.603±0.039 0.571±0.030 3.297 0.002 右侧VIC值 0.617±0.021 0.581±0.040 4.301 0.000 左侧ODI值 0.381±0.015 0.346±0.024 6.673 0.000 右侧ODI值 0.366±0.012 0.349±0.026 3.102 0.003 左侧VISO值 0.070±0.033 0.081±0.008 1.429 0.168 右侧VISO值 0.075±0.027 0.079±0.015 0.607 0.549 白质 左侧VIC值 0.615±0.017 0.587±0.038 3.963 0.001 右侧VIC值 0.627±0.027 0.591±0.030 4.372 0.000 左侧ODI值 0.416±0.015 0.433±0.029 2.791 0.007 右侧ODI值 0.421±0.011 0.431±0.024 2.057 0.045 左侧VISO值 0.054±0.017 0.049±0.012 1.059 0.294 右侧VISO值 0.053±0.017 0.050±0.013 0.811 0.421 PD: 帕金森病; VIC: 细胞内休积分数; ODI: 取向弥散指数; VISO: 各向同性隔室的休积分. 表 2 齿状核诊断PD的ROC诊断
Table 2. Diagnosis of dentate nucleus ROC diagnosis of PD
检验结果变量 AUC面积 标准误 P 95%CI 临界值 灵敏度 特异度 下限 上限 齿左VIC 0.743 0.076 0.003 0.595 0.891 0.6065 88.60% 60.00% 齿右VIC 0.767 0.063 0.001 0.644 0.89 0.595 60.00% 90.00% 齿左ODI 0.891 0.042 0.000 0.809 0.972 0.3595 68.60% 95.00% 齿右ODI 0.694 0.070 0.017 0.557 0.831 0.355 60.00% 85.00% -
[1] Zeng LL, Xie L, Shen H, et al. Differentiating patients with Parkinson's disease from normal controls using gray matter in the cerebellum[J]. Cerebellum, 2017, 16(1): 151-7. doi: 10.1007/s12311-016-0781-1 [2] Braak H, Del Tredici K. Neuropathological staging of brain pathology in sporadic Parkinson's disease: separating the wheat from the chaff[J]. J Parkinsons Dis, 2017, 7(s1): S71-85. doi: 10.3233/JPD-179001 [3] Mirdamadi JL. Cerebellar role in Parkinson's disease[J]. J Neurophysiol, 2016, 116(3): 917-9. doi: 10.1152/jn.01132.2015 [4] Ma XX, Su W, Li SH, et al. Cerebellar atrophy in different subtypes of Parkinson's disease[J]. J Neurol Sci, 2018, 392: 105-12. doi: 10.1016/j.jns.2018.06.027 [5] Seidel K, Bouzrou M, Heidemann N, et al. Involvement of the cerebellum in Parkinson disease and dementia with Lewy bodies[J]. Ann Neurol, 2017, 81(6): 898-903. doi: 10.1002/ana.24937 [6] Lope-Piedrafita S. Diffusion tensor imaging (DTI)[J]. Methods Mol Biol, 2018, 1718: 103-116. [7] 余健敏, 李蒙燕, 周进, 等. 磁共振弥散张量成像在帕金森病诊断中的临床价值[J]. 现代医院, 2019, 19(4): 600-4. https://www.cnki.com.cn/Article/CJFDTOTAL-XDYU201904041.htm [8] Kamiya K, Hori M, Aoki S. NODDI in clinical research[J]. J Neurosci Methods, 2020, 346: 108908. doi: 10.1016/j.jneumeth.2020.108908 [9] Hansen B, Khan AR, Shemesh N, et al. White matter biomarkers from fast protocols using axially symmetric diffusion kurtosis imaging[J]. NMR Biomed, 2017, 30(9): 1-33. http://www.ncbi.nlm.nih.gov/pubmed/28543843 [10] Kamagata K, Hatano T, Aoki S. What is NODDI and what is its role in Parkinson's assessment?[J]. Expert Rev Neurother, 2016, 16(3): 241-3. doi: 10.1586/14737175.2016.1142876 [11] Palacios EM, Owen JP, Yuh EL, et al. The evolution of white matter microstructural changes after mild traumatic brain injury: a longitudinal DTI and NODDI study[J]. Sci Adv, 2020, 6(32): eaaz6892. doi: 10.1126/sciadv.aaz6892 [12] 杨改清, 祁萌萌, 张晓艺. 功能磁共振在帕金森病诊断中的价值研究[J]. 航空航天医学杂志, 2018, 29(12): 1530-1. doi: 10.3969/j.issn.2095-1434.2018.12.049 [13] Mitchell T, Archer DB, Chu WT, et al. Neurite orientation dispersion and density imaging (NODDI) and free-water imaging in Parkinsonism[J]. Hum Brain Mapp, 2019, 40(17): 5094-107. doi: 10.1002/hbm.24760 [14] Deligianni F, Carmichael DW, Zhang GH, et al. NODDI and tensorbased microstructural indices as predictors of functional connectivity [J]. PLoS One, 2016, 11(4): e0153404. doi: 10.1371/journal.pone.0153404 [15] Zhang H, Schneider T, Wheeler- Kingshott CA, et al. NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain[J]. NeuroImage, 2012, 61(4): 1000-16. doi: 10.1016/j.neuroimage.2012.03.072 [16] Juttukonda MR, Franco G, Englot DJ, et al. White matter differences between essential tremor and Parkinson disease[J]. Neurology, 2019, 92(1): e30-9. doi: 10.1212/WNL.0000000000006694 [17] Gao LL, Zhang JR, Hou YN, et al. The cerebellum in dual- task performance in Parkinson's disease[J]. Sci Rep, 2017, 7(1): 45662. doi: 10.1038/srep45662 [18] Becerra-Laparra I, Cortez-Conradis D, Garcia-Lazaro HG, et al. Radial diffusivity is the best global biomarker able to discriminate healthy Elders, mild cognitive impairment, and Alzheimer's disease: a diagnostic study of DTI- derived data[J]. Neurol India, 2020, 68 (2): 427-34. doi: 10.4103/0028-3886.284376 [19] Gazdzinski LM, Mellerup M, Wang T, et al. White matter changes caused by mild traumatic brain injury in mice evaluated using neurite orientation dispersion and density imaging[J]. J Neurotrauma, 2020, 37(16): 1818-28. doi: 10.1089/neu.2020.6992 [20] Gong T, Tong QQ, He HJ, et al. MTE-NODDI: Multi-TE NODDI for disentangling non-T2-weighted signal fractions from compartmentspecific T2 relaxation times[J]. Neuroimage, 2020, 217: 116906. doi: 10.1016/j.neuroimage.2020.116906 [21] Wu T, Hallett M. The cerebellum in Parkinson's disease[J]. Brain, 2013, 136(3) http://brain.oxfordjournals.org/content/136/3/696 -







 下载:
下载: