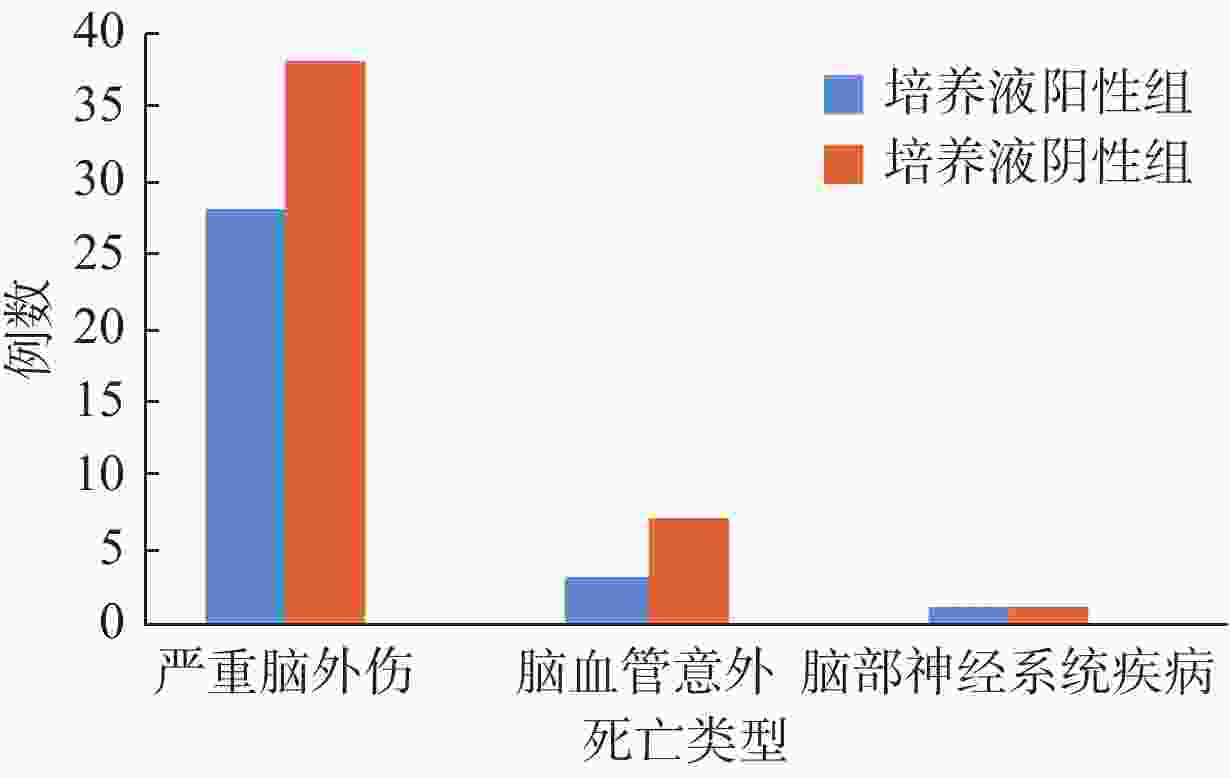
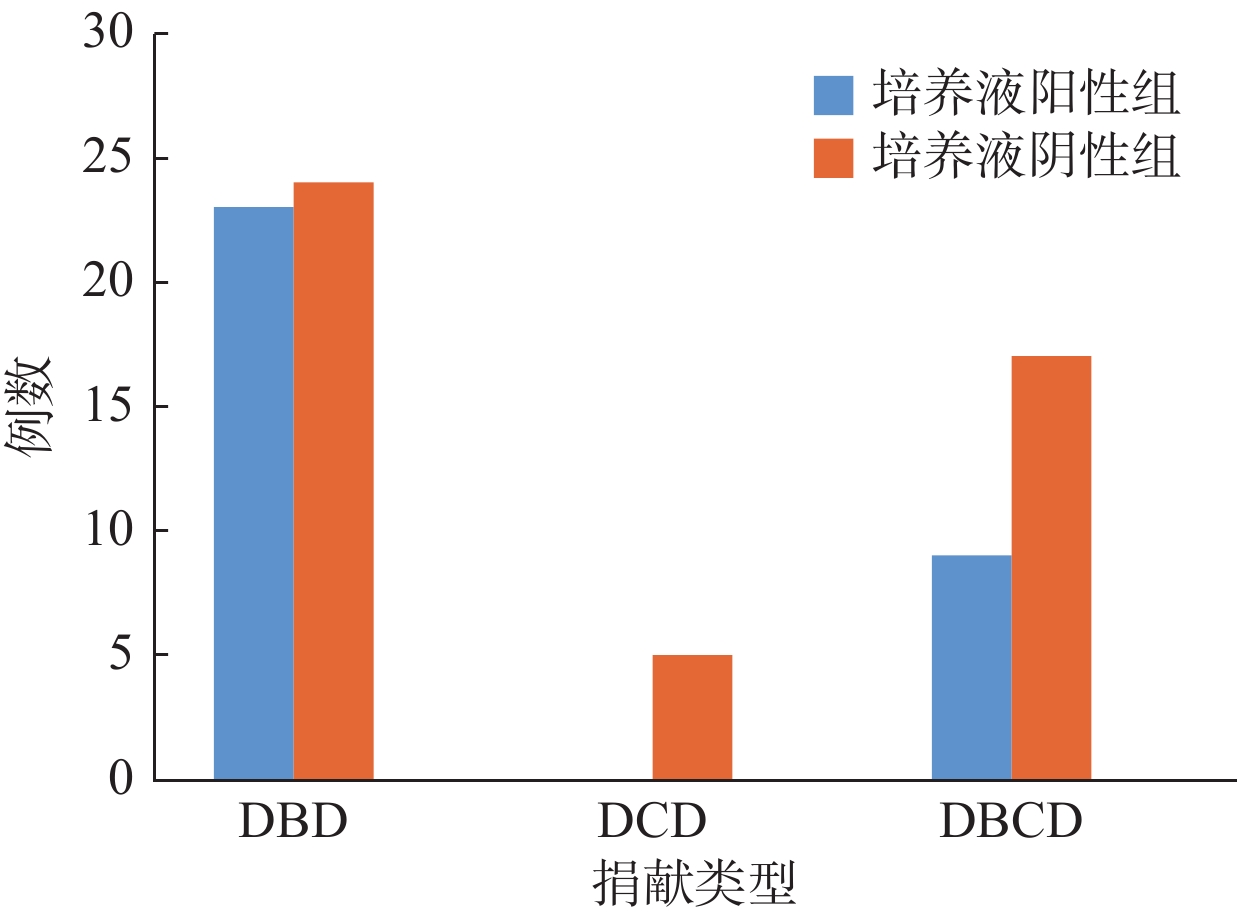
Single-center analysis on preservation solution contamination and initial experience of liver transplantation from organ donation by citizens after death
-
摘要:
目的分析公民逝世后器官捐献肝移植保存液(PS)污染情况。 方法根据捐献肝脏PS培养结果将2016年3月~2017年10月广州某三甲医院78例公民逝世后器官捐献肝移植受者分为阳性组和阴性组。阳性组围手术期序贯应用厄他培南和亚胺培南,疗程为1周;阴性组停用亚胺培南。对保存液污染病原菌分布、术后3月感染和预后情况进行统计分析。 结果78例肝移植患者中,32例PS培养阳性,阳性率41.03%,共分离出33株病原菌。分离出最常见的病原菌是革兰氏阴性杆菌(9株,27.27%)和凝固酶阴性葡萄糖球菌(9株,27.27%)。培养液阳性组和阴性组肝移植术后感染率分别为31.25%和13.04%(χ2=3.837,P=0.048)。感染例次最多的部位是下呼吸道和腹腔(5例,31.25%),其次是手术切口(4例,25.00%)。不同肝功能CTP、MELD评分以及手术方式患者术后感染率差异无统计学意义(P>0.05)。1例(1.28%)术后3周发生与PS污染相同的病原菌感染,并因多脏器功能衰竭而死亡。在术后随访3月内,阳性组和阴性组患者急性排斥反应率和死亡率分别为3.13%(1例)、4.35%(2例)和6.25%(2例)、10.87%(5例),差异均无统计学意义(P>0.05)。 结论公民逝世后捐献肝脏PS经常受到污染,PS污染是受者术后感染的危险因素。早期针对性抗菌素治疗对降低公民逝世后捐献肝脏PS污染相关的感染有着积极作用。 Abstract:ObjectiveTo discuss the preservation solution (PS) contamination and initial experience of liver transplantation from organ donation by citizens after death and initial experience. MethodsThe 78 liver transplant recipients in a hospital in Guangzhou from March 2016 to October 2017 were divided into two groups based on the finding of the culture of PS. The positive group received the sequential therapy of antibiotics with ertapenem and imipenem for one week. The negative group stopped using imipenem. The situation of PS contamination and infection after liver transplantation and prognosis during the follow-up 3 months of the recipients were analyzed. ResultsAmong the 78 recipients, 32 PSs culture were positive (41.03%), and 33 strains of pathogens were isolated. The most common pathogenic bacteria were gram-negative bacilli (9 strains, 27.27%) and coagulase–negative staphylococci (9 strains, 27.27%). The infection rates after liver transplantation in culture positive group and culture negative group were 31.25% and 13.04%, respectively (χ2=3.837, P=0.048). The most frequent infection sites were lower respiratory tract (5 cases, 31.25%), abdominal cavity (5 cases, 31.25%) and surgical incision (4 cases, 25.00%). There was no significant difference of postoperative infection rate among patients with different CTP, MELD and surgical methods (P>0.05). One case (1.28%) was infected with the same pathogenic bacteria as PS contamination 3 weeks after liver transplantation, and died of multiple organ failure. There was no significant difference of the acute rejection rate (1, 3.13% and 2, 4.35%) and mortality (2, 6.25% and 5, 10.87%) between the two groups (P>0.05). ConclusionContamination of the PS is frequent in liver transplantation, which is the risk factor for postoperative infection of recipients. Early targeted antimicrobial treatment against pathogens cultured from PS is positive in reducing the contamination-associated infection rate after liver transplantation. -
Key words:
- bacterial contamination /
- preservation solution /
- liver /
- transplantation /
- recipient infection /
- ertapenem
-
表 1 纳入研究的肝移植受者基础肝脏疾病构成比[n(%)]
分组 肝癌 肝炎后
肝硬化慢性重型
肝炎胆管
细胞癌其他 培养阳性组(n=32) 20(40.82) 6(37.50) 4(57.14) 1(50.00) 1(25.00) 培养阴性组(n=46) 29(59.18) 10(62.50) 3(42.86) 1(50.00) 3(75.00) 合计 49(100.00) 16(100.00) 7(100.00) 2(100.00) 4(100.00) 表 2 保存液培养病原菌构成比
病原菌 株数(n=33) 构成比(%) 革兰阳性球菌 16 48.48 金黄色葡萄球菌 1 3.03 凝固酶阴性葡萄糖球菌 9 27.27 肠球菌 4 12.12 其他 2 6.06 革兰阴性杆菌 9 27.27 肠杆菌属 6 18.18 恶臭假单胞菌 2 6.06 鲍曼不动杆菌 1 3.03 革兰阳性杆菌 3 9.09 蜡状芽孢杆菌 2 6.06 棒状杆菌 1 3.03 酵母菌 5 15.15 白假丝酵母 2 6.06 热带假丝酵母 2 6.06 光滑假丝酵母 1 3.03 表 3 肝移植受者术后3月感染率、急性排斥发生率、术后发热率和死亡率分布[n(%)]
分组 感染 急性排斥反应 术后发热 死亡 培养液阳性组 10(31.25) 1(3.13) 13(40.63) 2(6.25) 培养液阴性组 6(13.04) 2(4.35) 9(19.57) 5(10.87) P 0.048 0.635 0.042 0.391 表 4 术后不同部位感染例次数构成比[n(%)]
分组 感染部位 下呼吸道 腹腔 手术切口 血液 皮肤和软组织 培养液阳性组 3(30.00) 2(20.00) 3(30.00) 1(10.00) 1(10.00) 培养液阴性组 2(33.33) 3(50.00) 1(16.67) 0(0.00) 0(0.00) P 0.331 0.669 0.185 0.571 0.571 表 5 纳入研究的供者一般资料和临床指标比较
分组 男性
[n(%)]平均年龄(岁) 平均ICU
存留(d)器官获取
时间(min)培养液阳性组(n=32) 29(90.63) 39(16~61) 4(2~7) 37(19~60) 培养液阴性组(n=46) 41(89.13) 38(14~61) 3(2~5) 37(18~60) P 0.572 0.745 0.000 0.917 表 6 PS培养阳性组与阴性组受者手术指标比较(Mean±SD)
受者因素 培养液阳性组(n=32) 培养液阴性组(n=46) P CTP 8.19±2.61 8.41±3.09 0.737 MELD 19.01±12.20 18.72±10.5 0.618 手术方式(n) 0.294 经典式 21 34 背驮式 11 12 无肝期(min) 62.31±14.53 63.42±17.51 0.765 冷缺血时间(min) 301.62±82.83 308.21±88.62 0.741 术中输红细胞量(u) 10.10±7.22 8.73±7.32 0.435 肝移植手术持续时间(h) 7.43±1.81 7.82±1.93 0.287 CTP: Child-Turcotte-Pugh; MELD: Model for End-Stage Liver Disease. 表 7 不同CTP或MELD肝功能分级患者的术后感染率
项目 调查例数(n) 感染例数(n) 感染率(%) CTP A级 29 5 17.24 B级 22 8 26.67 C级 27 3 11.11 MELD(分) <20 51 8 15.69 20~30 10 3 30.00 30~40 15 4 26.67 >40 2 1 50.00 -
[1] Dorschner P, Mcelroy LM, Ison MG. Nosocomial infections within the first month of solid organ transplantation[J]. Transpl Infect Dis, 2014, 16(2): 171-87. doi: 10.1111/tid.12203 [2] Avkan-Oguz V, Ozkardesler S, Unek T, et al. Risk factors for early bacterial infections in liver transplantation[J]. Transplant Proc, 2013, 45(8): 993-7. [3] Fishman JA. From the classic concepts to modern practice[J]. Clin Microbiol Infect, 2014, 20(1): 4-9. [4] Oriol I, Sabé N, Tebé C, et al. Clinical impact of culture-positive preservation fluid on solid organ transplantation: a systematic review and meta-analysis[J]. Transplant Rev, 2018, 32(3): 85-91. [5] Janny S, Bert F, Dondero F, et al. Microbiological findings of culture-positive preservation fluid in liver transplantation[J]. Transpl Infect Dis, 2011, 13(1): 9-14. doi: 10.1111/j.1399-3062.2010.00558.x [6] Audet M, Piardi T, Panaro F, et al. Incidence and clinical significance of bacterial and fungal contamination of the preservation solution in liver transplantation[J]. Transpl Infect Dis, 2011, 13(2): 84-8. [7] 中华医学会器官移植学分会. 中国心脏死亡器官捐献工作指南[J/CD]. 中华移植杂志: 电子版, 2012, 6(3): 221-4. [8] 中华人民共和国卫生部. 医院感染诊断标准[J]. 中华医药杂志, 2001, 81(5): 314-20. [9] Oriol I, Lladó L, Vila M, et al. The etiology, incidence, and impact of preservation fluid contamination during liver transplantation[J]. PLoS One, 2016, 11(8): e0160701-12. doi: 10.1371/journal.pone.0160701 [10] 臧晓青, 钟 林, 彭志海. 肝移植后感染的遗传易感性研究[J]. 中华临床医师杂志: 电子版, 2013, 7(1): 238-40. [11] Garcia-Zamora C, Segura J, Lopez-Lopez V, et al. Clinical significance of contamination of the preservation solution in liver transplantation[J]. Transplant Proc, 2015, 47(14): 2322-3. [12] Ruiz P, Gastaca M, Gonzalez J, et al. Incidence and clinical relevance of bacterial contamination in preservation solution for liver transplantation[J]. Transplant Proc, 2009, 41(12): 2169-71. [13] Sauget M, Verdy S, Slekovec C, et al. Bacterial contamination of organ graft preservation solution and infection after transplantation[J]. Transpl Infect Dis, 2011, 13(9): 331-4. [14] Levesque E, Suet G, Merle JC, et al. Candida vascular complication in a liver transplant recipient due to yeast contamination of preservation solution[J]. Transpl Infect Dis, 2014, 16(5): 827-9. doi: 10.1111/tid.12260 [15] Bud O, Golling M, Von Frankenberg M, et al. Intramucosal pH and serum endotoxin concentrations as early predictive parameters for primary nonfunction after experimental liver transplantation[J]. Transplant Proc, 2000, 32(12): 2537-38. [16] 赵纪强, 霍 枫, 李 鹏, 等. 中国心脏死亡器官捐献工作发展及影响因素: 单中心经验[J/CD]. 中华移植杂志: 电子版, 2017, 11(1): 32-6. [17] Shah PM, Ertapenem IR. The first of a new group of carbapenems[J]. J Antimicrob Chemother, 2003, 52(11): 538-42. [18] Goegele H, Berger N, Kafka R, et al. Course of transplant recipients treated with Ertapenem in the prophylaxis and treatment of infections: a first experience[J]. Eur Surg, 2007, 39(3): 196-202. doi: 10.1007/s10353-007-0326-4 [19] Oliva A, Scorzolini L, Cipolla A, et al. In vitro evaluation of different antimicrobial combinations against carbapenemase-producing Klebsiella pneumoniae: the activity of the double-carbapenem regimen is related to meropenem MIC value[J]. J Antimicrob Chemother, 2017, 72(20): 1981-4. [20] Piedra-Carrasco N, Miguel L, Fàbrega A, et al. Effectiveness of a double-carbapenem regimen in a KPC-Producing Klebsiella pneumoniae infection in an immunocompromised patient[J]. Microbial Drug Resistance, 2018, 24(2): 199-202. doi: 10.1089/mdr.2017.0129 [21] Botterel F, Foulet F, LegrandP, et al. Yeast contamination of kidney, liver and cardiac preservation solutions before graft: need for standardisation of microbial evaluation[J]. J Hosp Infect, 2010, 76(1): 52-5. doi: 10.1016/j.jhin.2010.02.008 -






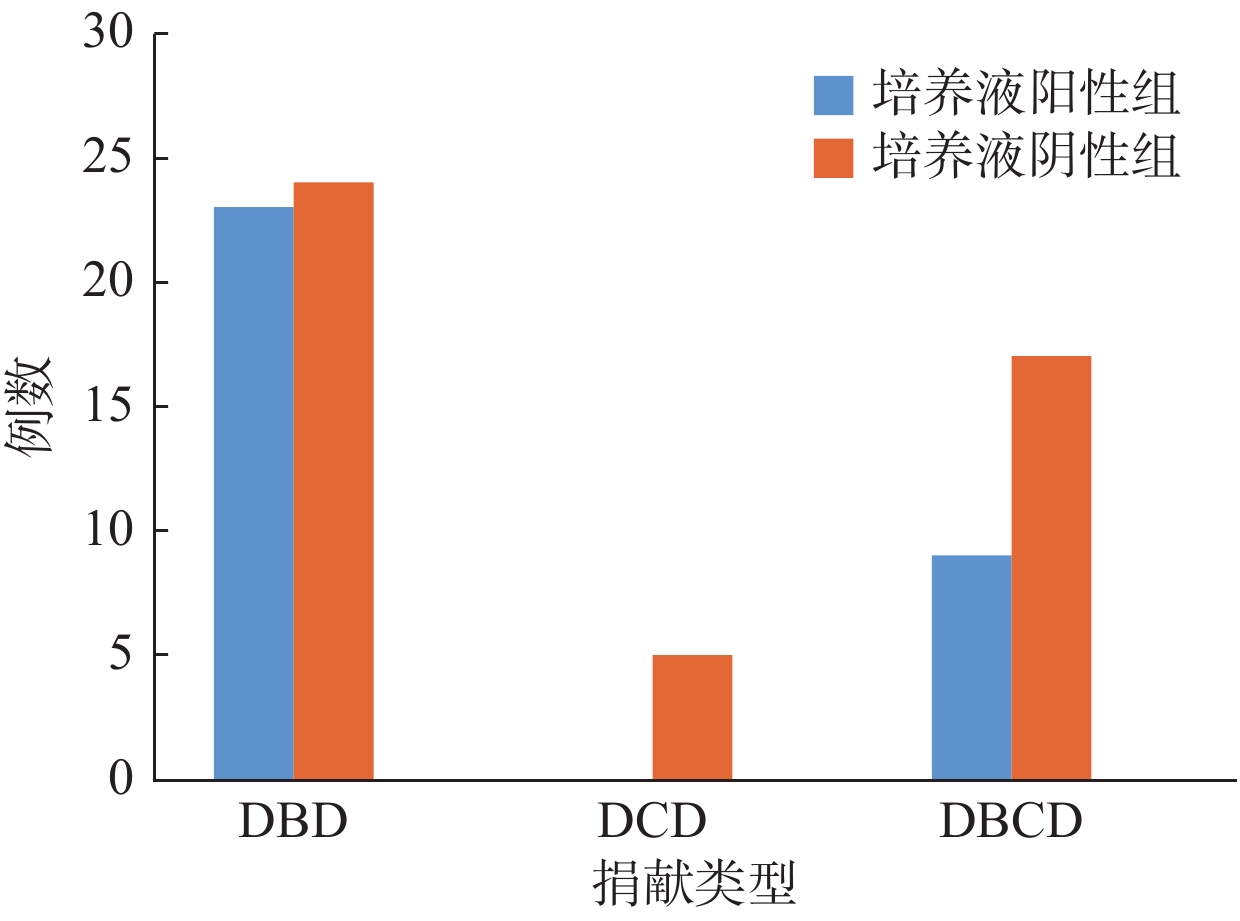
 下载:
下载: