Levels of Bax and Bcl-2 mRNA and their relationship with cardiac function in patients with stable CHF
-
摘要:
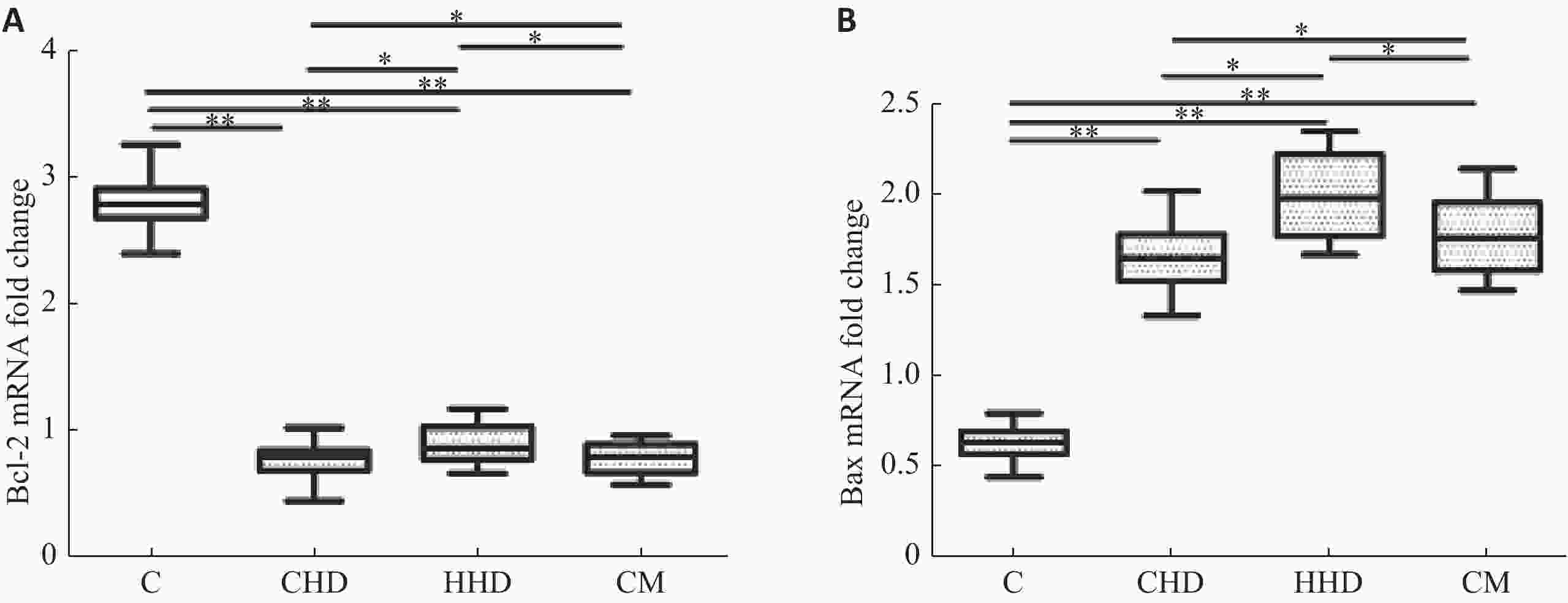
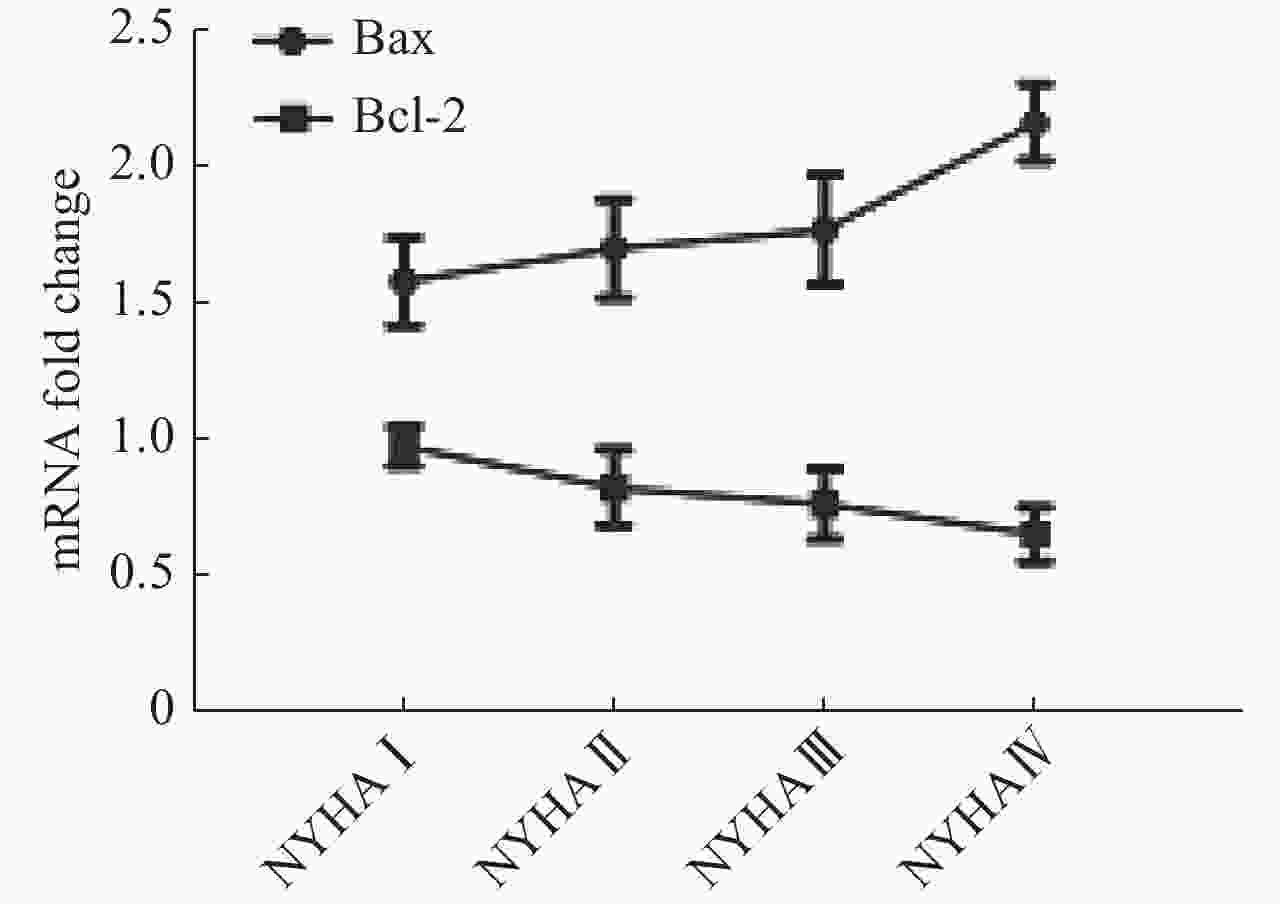
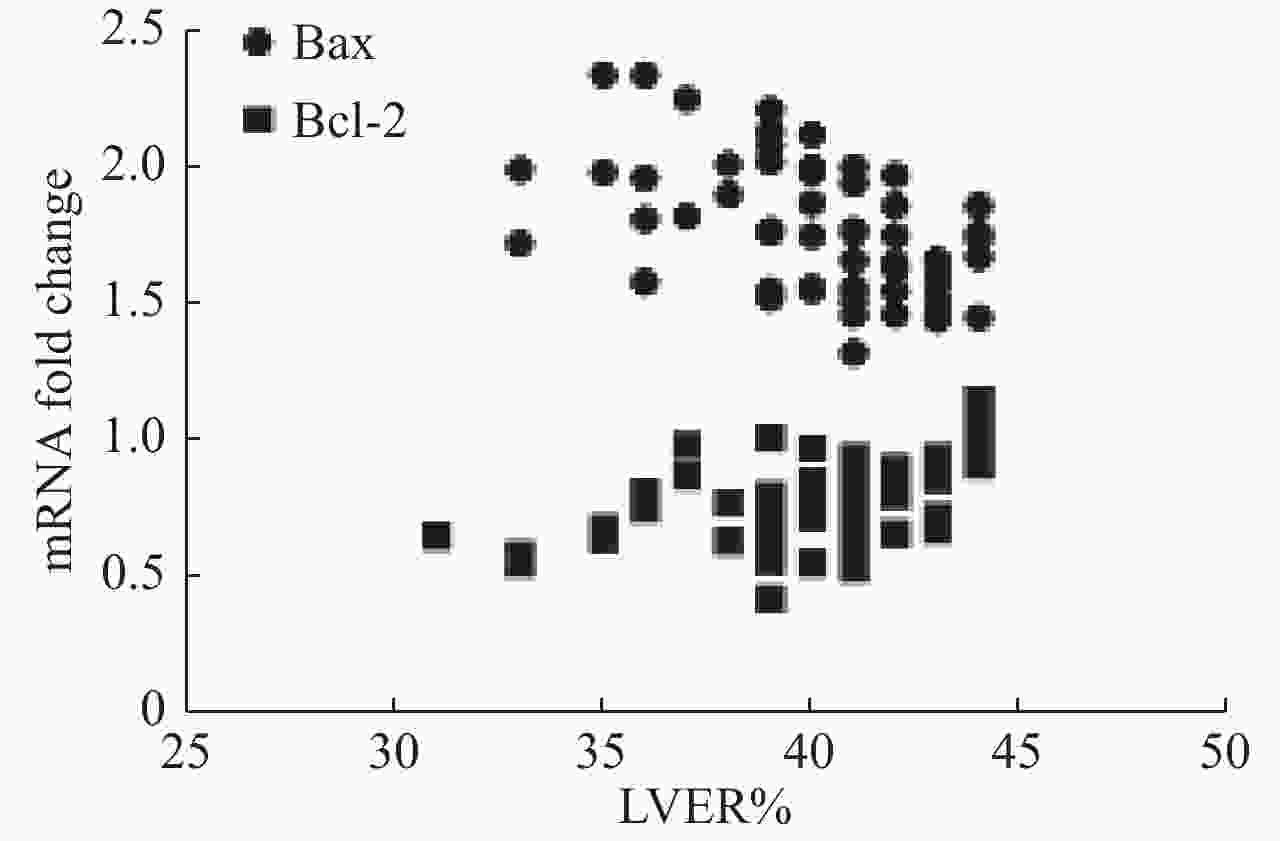
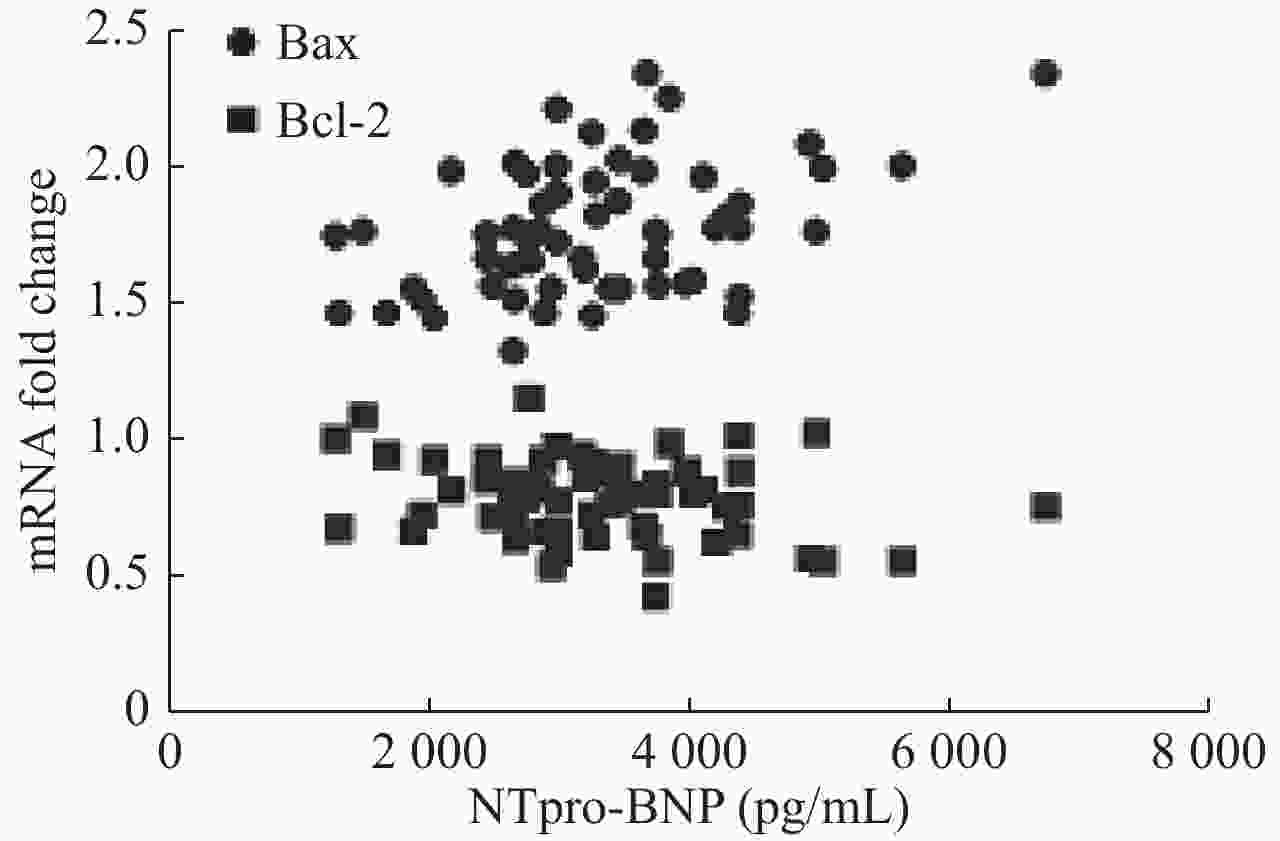
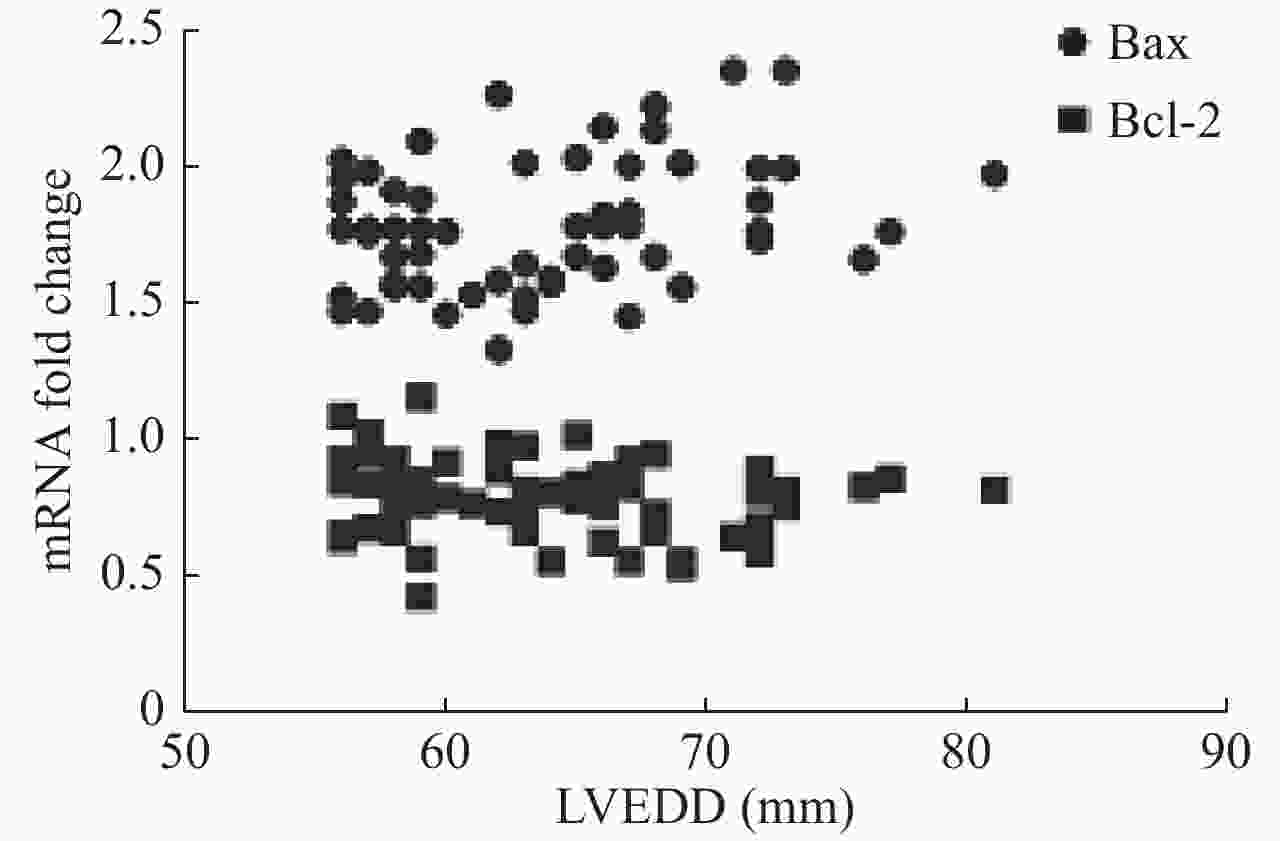
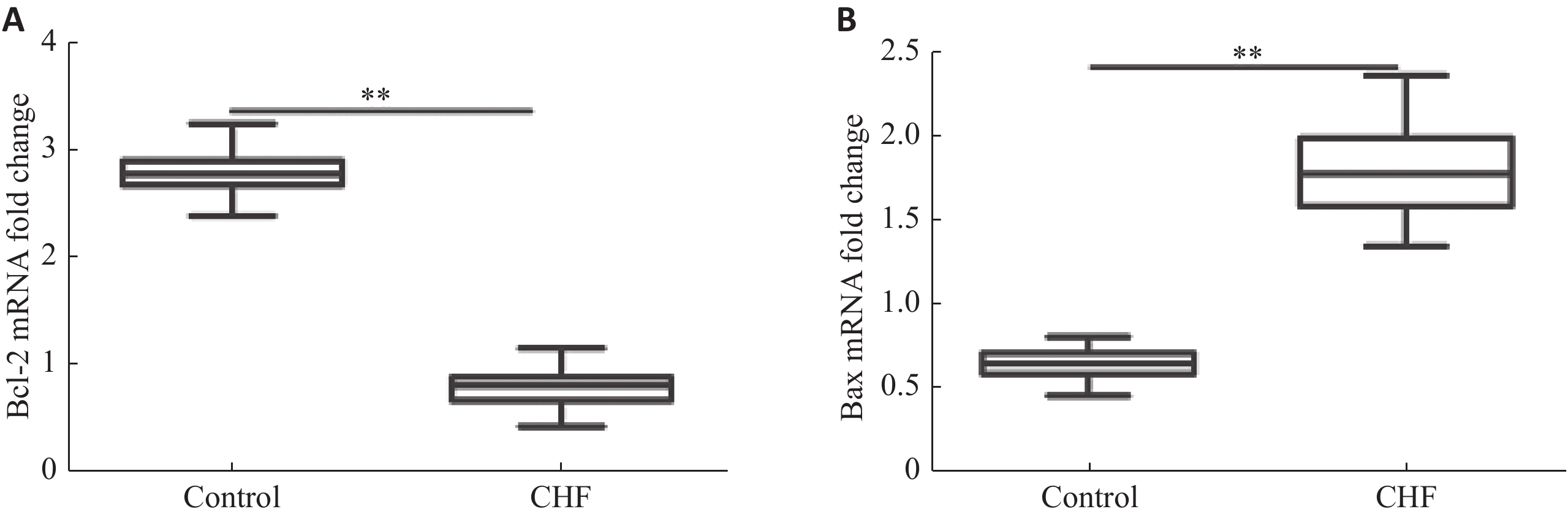
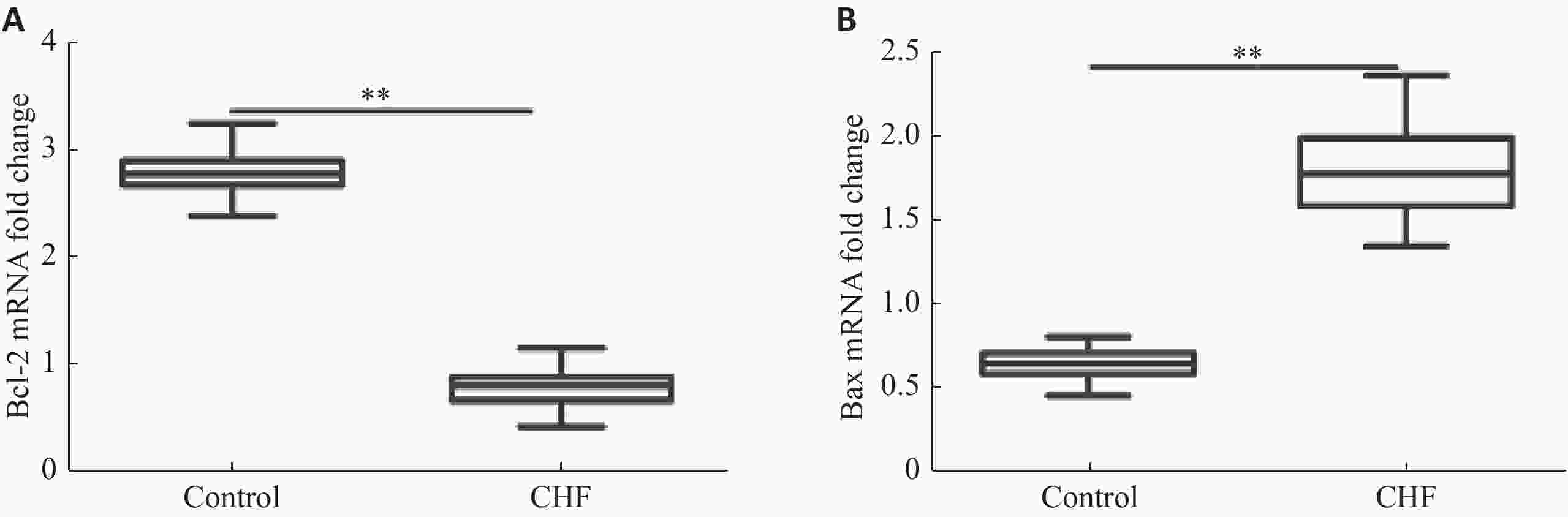
目的探讨外周血单核细胞Bcl-2和Bax水平与心功能和左室射血分数的关系。 方法纳入60例稳定期慢性心衰患者和30例健康对照,采用qRT-PCR法测量外周血单核细胞Bcl-2和Bax mRNA相对含量;NYHA心功能分级评价心功能,并采用彩色超声多普勒测量LVEF。 结果CHF组外周血单核细胞Bcl-2 mRNA水平低于对照组(P<0.01),Bax mRNA水平高于对照组(P<0.01)。CHF组外周血单核细胞Bcl-2 mRNA水平与NHYA心功能呈负相关(P<0.05),与LVEF呈正相关(P<0.01);Bax mRNA水平与NHYA心功能呈负相关(P<0.01),与LVEF呈负相关(P<0.01)。CHF组外周血单核细胞Bax mRNA水平与N-末端B型钠尿肽前体浓度呈正相关(P<0.01)。Bcl-2 mRNA水平与N-末端B型钠尿肽前体浓度不相关(P>0.05)。CHF组外周血单核细胞Bax mRNA水平与左室舒张末期内径呈正相关(P<0.05),Bcl-2 mRNA水平与左室舒张末期内径不相关(P>0.05)。 结论Bcl-2和Bax这一对凋亡相关基因在慢性心衰稳定期表达水平发生明显变化,各种病因所致的CHF可能导致这一对凋亡相关基因共同的改变,且mRNA水平与CHF病情严重程度相关,可能成为CHF预后因子之一。 Abstract:ObjectiveTo explore the relationship between mRNA level of Bax and Bcl-2 in peripheral blood mononuclear cells (PBMCs) and cardiac function and left ventricular ejection fraction. MethodsSixty patients with stable chronic heart failure and 30 healthy controls were enrolled. Monocytes were isolated and cultured from peripheral blood and mRNA were extracted. Relative content of Bax and Bcl-2 mRNA were measured by quantitative reverse transcription- polymerase chain reaction (qRT-PCR). Cardiac functions were evaluated by New York Heart Association (NYHA) functional classification system. Left ventricular ejection fraction was measured by transthoracic color doppler echocardiography. ResultsCompared with the control group, the level of Bcl-2 mRNA in PBMCs in CHF group decreased (P<0.01), and the level of Bax mRNA increased (P<0.01). The level of Bcl-2 mRNA was negatively correlated with NYHA class in CHF group (P<0.05). The level of Bax mRNA was positively correlated (P<0.05). The level of Bcl-2 mRNA in PBMCs were positively correlated with LVEF in CHF group (P<0.01. The level of Bax mRNA was negatively correlated (P<0.01). The level of Bax mRNA in PBMCs was positively correlated with the levels of NT-proBNP in CHF group (P<0.01), and the level of Bcl-2 mRNA was not correlated (P>0.05). The level of Bax mRNA in PBMCs was positively correlated with LVEDD in CHF group (P<0.05), and the level of Bcl-2 mRNA was not correlated (P>0.05). ConclusionBax and Bcl-2, a pair of apoptosis-related genes, change significantly in the expression of stable chronic heart failure. Various etiologies may lead to the common changes of these apoptosis-related genes. The mRNA of these genes is correlated with the severity of CHF. It may be one of the prognostic factors of CHF. -
Key words:
- chronic heart failure /
- genes, cell apoptosis /
- cardiac function /
-
表 1 研究对象一般临床资料
因素 CHD组 HHD组 CM组 Control组 年龄(Mean±SD,岁) 68.2±5.5 57.4±4.2* 60.7±4.4* 60.7±6.1 男/女(n) 16/12 9/6 7/10 16/14 吸烟[n(%)] 15(53.6) 5(33.3)** 6(35.3)** 12(40.0) 高血压[n(%)] 8(28.6) 15(100)** 2(11.76)**## - 糖尿病[n(%)] 9(32.1) 5(33.33)* 2(11.76)**## - 纽约心功能分级(Mean±SD) 2.5±0.8 2.3±0.7 3.1±0.8 - 6 min步行试验(Mean±SD,m) 276.4±42.2 248.0±63.9 246.5±51.7 - NT-proBNP(Mean±SD,pg/mL) 3121.3±982.8 3492.3±1231.7 3390.0±924.7 73.6±18.9&& LVEF(Mean±SD,%) 40.7±2.3 40.3±2.8 38.6±3.6 57.7±4.3&& LVEDD(Mean±SD,mm) 63.5±6.4 64.3±5.8 63.4±6.4 49.5±3.2&& 左房内径(Mean±SD,mm) 40.4±4.4 41.0±4.0 38.6±6.1 33.0±2.9&& 右房横径(Mean±SD,mm) 37.0±2.0 36.1±2.3 37.6±2.4 37.0±2.0 右室舒张期横径(Mean±SD,mm) 27.8±3.4 29.2±2.4 26.1±4.6 18.5±3.5&& 药物[n(%)] 血管紧张素转化酶抑制剂/血管紧张素受体拮抗剂 16(57.1) 7(46.7) 9(52.9) - β受体阻滞剂 13(72.2) 10(66.7) 8(47.1) - 利尿剂 24(85.8) 13(86.7) 15(88.2) - 阿司匹林 27(96.4) 3(20.2)** 2(11.8)** - 他汀类 26(92.9) 5(33.3)** 1(5.9)**## - *P<0.05,**P<0.01 vs CHD;## P<0.01 vs HHD;&&P<0.01 vs CHF. CHD:冠心病组;HHD:高血压心脏病;CM:心肌病;Control:对照组. -
[1] Savarese G, Lund LH. Global public health burden of heart failure[J]. Card Fail Rev, 2017, 3(1): 7-11. doi: 10.15420/cfr.2016:25:2 [2] Abbate A, Biondi-Zoccai GG, Bussani R, et al. Increased myocardial apoptosis in patients with unfavorable left ventricular remodeling and early symptomatic post-infarction heart failure[J]. J Am Coll Cardiol, 2003, 41(5): 753-60. doi: 10.1016/S0735-1097(02)02959-5 [3] Johnson FL. Pathophysiology and etiology of heart failure[J]. Cardiol Clin, 2014, 32(1): 9-19. doi: 10.1016/j.ccl.2013.09.015 [4] Sabbah HN, Sharov VG, Goldstein S. Cell death, tissue hypoxia and the progression of heart failure[J]. Heart Fail Rev, 2000, 5(2): 131-8. doi: 10.1023/A:1009880720032 [5] Fiedler LR, Maifoshie E, Schneider MD. Mouse models of heart failure: cell signaling and cell survival[J]. Curr Top Dev Biol, 2014, 109(3): 171-247. [6] Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death[J]. Nat Rev Mol Cell Biol, 2008, 9(1): 47-59. doi: 10.1038/nrm2308 [7] Walensky LD, Gavathiotis E. BAX unleashed: the biochemical transformation of an inactive cytosolic monomer into a toxic mitochondrial pore[J]. Trends Biochem Sci, 2011, 36(12): 642-52. doi: 10.1016/j.tibs.2011.08.009 [8] Diwan A, Krenz M, Syed FM, et al. Inhibition of ischemic cardiomyocyte apoptosis through targeted abla¬tion of Bnip3 restrains postinfarction remodeling in mice[J]. J Clin Invest, 2007, 117(21): 2825-33. [9] Condorelli G, Morisco C, Stassi G, et al. Increased cardiomyocyte apoptosis and changes in proapoptotic and antiapoptotic genes bax and bcl-2 during left ventricular adaptations to chronic pressure overload in the rat[J]. Circulation, 1999, 99(23): 3071-8. doi: 10.1161/01.CIR.99.23.3071 [10] Shimizu S, Narita M, Tsujimoto Y. Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial Channel VDAC[J]. Nature, 1999, 399(6735): 483-7. doi: 10.1038/20959 [11] 郭 丽, 邓 萌, 王露露, 等. 心力衰竭大鼠氧化应激水平与炎症因子, 细胞凋亡因子间的相关性[J]. 心血管康复杂志, 2018, 5(27): 495-8. [12] Chatterjee S, Stewart AS, Bish LT, et al. Viral gene transfer of the antiapoptotic factor Bcl-2 protects against chronic postischemic heart failure[J]. Circulation, 2002, 106(12 Suppl 1): I212-7. [13] Akyurek O, Akyurek N, Sayin T, et al. Association between the severity of heart failure and the susceptibility of myocytes to apoptosis in patients with idiopathic dilated cardiomyopathy[J]. Int J Cardiol, 2001, 80(1): 29-36. doi: 10.1016/S0167-5273(01)00451-X [14] Ngo J, Matsuyama M, Kim C, et al. Bax deficiency extends the survival of Ku70 knockout mice that develop lung and heart diseases[J]. Cell Death Dis, 2015, 6(12): e1706-17. [15] Chao Z, Liuyang T, Nan L, et al. Mitochondrial tRNA mutation with high-salt stimulation on cardiac damage: underlying mechanism associated with change of Bax and VDAC[J]. Am J Physiol Heart Circ Physiol, 2016, 311(5): H1248-57. doi: 10.1152/ajpheart.00874.2015 [16] Zhang L, Ji H, Huang Y, et al. Association of BAX hypermethylation with coronary heart disease is specific to individuals aged over 70[J]. Medicine, 2019, 98(4): e14130-42. doi: 10.1097/MD.0000000000014130 [17] Zhang M, Huang B. The multi-differentiation potential of peripheral blood mononuclear cells[J]. Stem Cell Res Ther, 2012, 3(6): 48-59. doi: 10.1186/scrt139 [18] Zhang Z, Yan Y, Zhu Y, et al. In vivo study on the potential differention capabilities of human peripheral blood-derived mononuclear cells transplanted into the nude mice with myocardial infarction[J]. J Biomed Eng, 2008, 25(2): 424-8. [19] Fuerstenau-Sharp M, Zimmermann ME, Stark K, et al. Generation of highly purified human cardiomyocytes from peripheral blood mononuclear cell-derived induced pluripotent stem cells[J]. PLoS One, 2015, 10(5): e0126596-607. doi: 10.1371/journal.pone.0126596 [20] Diao SL, Xu HP, Zhang B, et al. Associations of MMP-2, BAX, and Bcl-2 mRNA and protein expressions with development of atrial fibrillation[J]. Med Sci Monit, 2016, 22(22): 1497-507. -






 下载:
下载: