Assessment of quality of randomized controlled trials research papers among Chinese abstracts for Endometrial cancer
-
摘要:
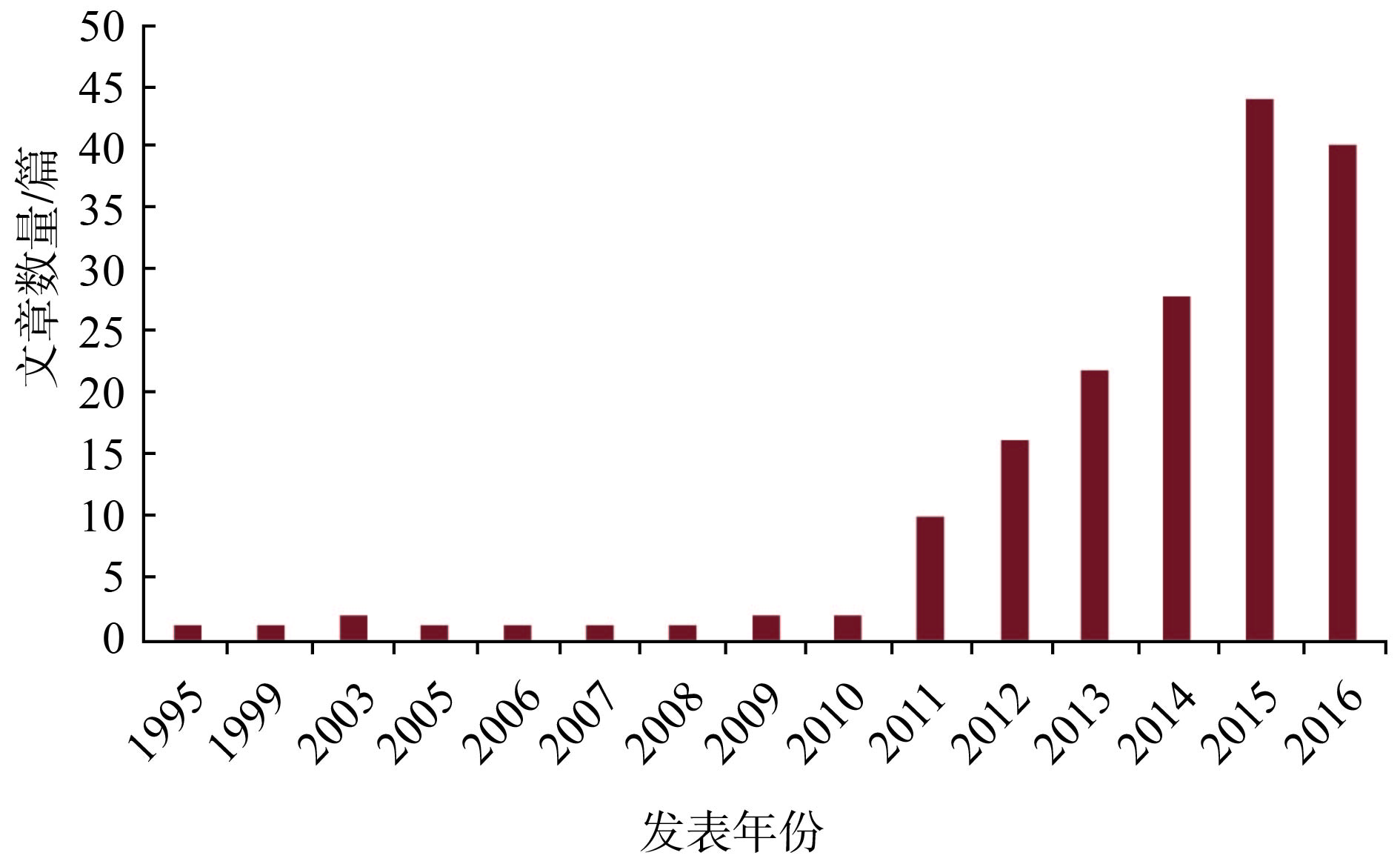
目的使用CONSORT声明评价中文发表的子宫内膜癌随机对照试验(RCT)摘要报告质量。 方法通过计算机检索中国生物医学文献数据库(1978年~2017年3月)、万方(1997年~2017年3月)、中国知网(1994年~2017年3月)和维普(1989年~2017年3月)数据库纳入的以中文发表的子宫内膜癌领域的RCT,依据摘要CONSORT 标准对所纳入RCT摘要进行质量评价,采用Meta-Analyst和RevMan5.1软件进行统计分析。 结果最终纳入符合标准的研究共171篇,全部为中文,发文量主要集中在四川、黑龙江、广东、河南、山东5省。参文作者数量以1~3位居多,共计131篇(76.6%)。自2008年“摘要CONSORT”发布之后发表文章数量明显增长,达到164篇(95.9%),且纳入的RCT中11个条目报告质量得到明显改善;被CSCD收录的5篇(2.9%)文献的完整报告率明显高于非CSCD收录的164篇文献(95.9%)。但在“标题随机[RR(95% CI) 0.3(0,4.5)]、研究对象[RR(95% CI)7.0(4.0,11.9)]、结局指标[RR(95% CI) 0.3(0,4.5)]、随机化[RR(95% CI) 12.3(8.1,18.1)]、盲法[RR(95% CI) 1.2(0.3,4.6)]、统计方法[RR(95% CI) 5.8(3.2,10.5)]、分析的例数[RR(95% CI) 4.1(2.0,8.3)]、试验注册[RR(95% CI) 0.3(0,4.5)]和资助来源[RR(95% CI) 10.5(6.7,16.1)] 9个方面仍存在严重不足。 结论目前中文发表子宫内膜癌RCT摘要质量存在严重缺陷,本研究采用CONSORT对这些RCT的摘要报告质量进行了评价,以期了解目前的现状,从而提高中文发表子宫内膜癌RCT的摘要报告质量。 Abstract:ObjectiveTo evaluate the reported quality of randomized controlled trials (RCTs) literature abstract of endometrial cancer published in Chinese using the abstract CONSORT statement. MethodsEndometrial-cancer-related RCTs were searched in CNKI, CBM, VIP and WF databases from inception to March 2017. the literature abstracts were conducted quality assessment in accordance with the abstract CONSORT standard. Meta-Analyst and RevMan5.1 software were used for statistical analysis. ResultsA total of 171 Chinese RCTs were included which the published areas were mainly Sichuan, Heilongjiang, Guangdong, Henan and Shandong. The number of reference writers ranged from 1 to 3, with a total of 131 articles (76.6%). Statistical data showed that the number of published articles has increased significantly and reached to 164 (95.9%) since the publication of the abstract CONSORT in 2008. The quality of the 11 entries in the RCTs has been significantly improved. In addition, the complete reporting rate of 5 articles (2.9%) collected by CSCD was significantly higher than that of the 164 non-CSCD articles (95.9%).The serious reporting flaws were appeared for RCT abstract of endometrial cancer regarding following items: title randomized, subject, investigated outcome indes, randomization, blinding, statistical methods, numbers analysised, trial registration, funding. The pooled RR with 95%CI were 0.3(0, 4.5), 7.0(4.0, 11.9), 0.3(0, 4.5), 12.3(8.1, 18.1), 1.2(0.3, 4.6), 5.8(3.2, 10.5), 4.1(2.0, 8.3), 0.3(0, 4.5), 10.5(6.7, 16.1), respectivly. ConclusionAt present, Chinese published endometrial cancer RCT abstract quality is poor. In this study, the quality of these RCT reports are evaluated by CONSORT in order to understand the current status and to improve the quality of Chinese abstracts published for endometrial cancer RCTs. -
Key words:
- endometrial cancer /
- randomized controlled trials /
- abstract /
- CONSORT
-
表 1 纳入研究数据提取表
项目 条目 描述 基本资料 作者 第一作者姓名 年代 文章发表年代 杂志 文章发表杂志 地区 第一作者所在省份 作者数量 文章作者数量 中(英)文摘要 是否有中(英)文摘要 摘要CONSORT 文章标题 判断是否为随机试验 文章摘要结构式摘要 判断是否为随机试验描述是否为结构式摘要 试验设计 描述实验的设计 研究对象 受试者的合格标准与数据资料收集的场所和地点 干预措施 描述每组计划如何干预 目的 具体目的或假设 结局指标 完整而确切定义该试验的主要结局 随机方法 试验对象进入不同干预组的分配方式 盲法 对受试者、医护提供者以及结局评价者是否隐瞒了分组方法 统计方法 描述试验中所使用的统计方法 随机分组例数 各组试验对象的人数 招募受试者 临床研究状态 分析的例数 各组纳入研究数据分析的受试者数目 结局 各组针对每一项主要结局指标结果,所评价效应估计值及其精确性 危害 各组出现的所有严重危害或副反应 结论 概括性的解释临床试验结果 试验注册 试验注册号和机构名称 资助 资助来源 表 2 纳入研究的一般特征(n=171)
项目 条目 篇数 95%CI 发表年代 1995~2007 7 4.1(2.0~8.3) 2008~2016 164 95.9(91.7~98.0) 发表杂志 CSCD 5 2.9(1.2~6.8) 非CSCD 164 95.9(91.7~98.0) 作者数量 1~3 131 76.6(69.7~82.4) 4~7 40 23.4(17.6~30.3) 摘要 中文 106 62.0(54.5~69.0) 中英文 65 38.0(31.0~45.5) 表 3 纳入的研究摘要报告质量评价结果(n=171)
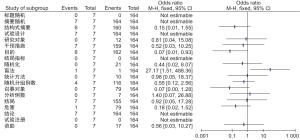
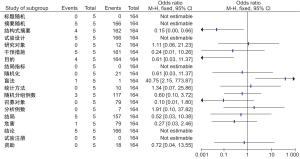
项目 条目 篇数 95%CI 标题 随机 0 0.3(0~4.5) 摘要 随机 171 99.7(95.5~1.0) 结构式 166 97.1(93.2 ~ 98.8) 方法 试验设计 171 99.7(95.5~1.0) 研究对象 12 7.0(4.0~11.9) 干预 166 97.1(93.2~98.8) 目的 168 98.2(94.7~99.4) 结局 0 0.3( 0~4.5) 随机方法 21 12.3(8.1~18.1) 盲法 2 1.2(0.3~4.6) 统计方法 10 5.8( 3.2~10.5) 结果 随机分组例数 120 70.2(62.9~76.6) 招募对象 79 46.2(38.9~53.7) 分析的例数 7 4.1(2.0~8.3) 结局 162 94.7(90.2~97.2) 危害 80 46.8( 39.4~54.3) 结论 171 99.7( 95.5~1.0) 试验注册 0 0.3(0~4.5) 资助 18 10.5(6.7~16.1) -
[1] Jemal A, Murray T, Ward E, et al. Cancer statistics[J]. CA Cancer J Clin, 2013, 310(9): 982-93. [2] Siegel RL, Miller KD, Jemal A. Cancer statistics[J]. CA Cancer J Clin, 2013, 310(9): 982-90. [3] 陈万青, 张思维, 曾红梅, 等. 中国2010年恶性肿瘤发病与死亡[J]. 中国肿瘤, 2014, 23(1): 1-10. doi: 10.3969/j.issn.1000-8179.20132205 [4] Nakagawa-Okamura C, Sato S, Tsuji I, et al. Effectiveness of mass screening for endometrial cancer[J]. Acta Cytol, 2002, 46(2): 277-83. doi: 10.1159/000326722 [5] Hopewell S, Clarke M, Moher D, et al. CONSORT for reporting randomized controlled trials in journal and conference abstracts: explanation and elaboration[J]. PLoS Med, 2008, 5(1): e20-9. doi: 10.1371/journal.pmed.0050020 [6] Kiriakou J, Pandis N, Madianos P, et al. Assessing the reporting quality in abstracts of randomized controlled trials in leading journals of oral implantology[J]. J Evid Based Dent Pract, 2014, 14(1): 9-15. doi: 10.1016/j.jebdp.2013.10.018 [7] 葛 龙, 李雅睿, 曾巧铃, 等. 发表于《中华儿科杂志》的系统评价/meta分析的报告质量和方法学质量评价[J]. 中华医学图书情报杂志, 2013, 22(12): 55-60. doi: 10.3969/j.issn.1671-3982.2013.12.016 [8] Guo JW, Iribarren SJ. Reporting quality for abstracts of randomized controlled trials in cancer nursing research[J]. Cancer Nurs, 2014, 37(6): 436-44. doi: 10.1097/NCC.0000000000000112 [9] 郑美玲, 沈 毅, 杨莉丽. 随机对照试验论文英文摘要的质量亟待提高[J]. 编辑学报, 2010, 22(2): 130-2. [10] Karbourtu AM, Knecht LS, Hunmphreys BL. Structured abstracts in MEDLINE,1989-1991[J]. Bull Med Libr Assoc, 1995, 83(2): 190-5. [11] Hopewell S, Eisinga A, Clarke M. Better reporting of randomized trials in biomedical journal and conference abstracts[J]. J Info Sci, 2008, 34(2): 162-73. doi: 10.1177/0165551507080415 [12] Dickersin K, Manheimer E, Wieland S, et al. Development of the cochrane collaborations central,register of controlled clinical trials[J]. Eval Health Prof, 2002, 25(1): 38-64. [13] Hopewell S, Clarke K, Moher D, et al. 期刊与学术会议论文摘要中报告随机对照试验的COMSORT声明:说明与详述[J]. 中西医结合学报, 2008, 6(3): 221-32. [14] Moher D, Schulz KF, Altman DG. The CONSORT statement:revised recommendations for improving the quality of reports of parallel group randomosed trials[J]. Lancet, 2001, 357(1): 2-7. [15] Gluud LL. Bias in clinical intervention research[J]. Am J Epidemiol, 2006, 163(6): 493-501. doi: 10.1093/aje/kwj069 [16] Schulz KF, Grimes DA. The lancet handbook of essential concepts in clinical research[M]. London: Elsevier, 2006. [17] Moher D, Hopewell S, Schulz KF, et al. CONSORT 2010说明与详细: 报告平行对照随机临床试验指南的更新[J]. 中西医结合学报, 2010, 8(8): 701-40. -







 下载:
下载: