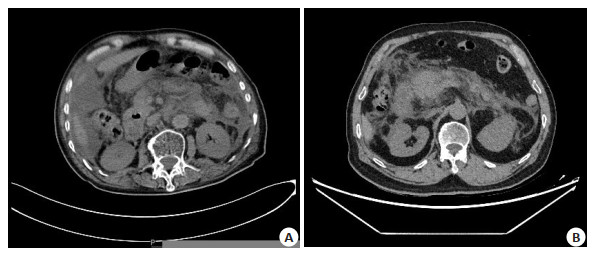
Prognostic evaluation of pancreatic CT density and maximum cross-sectional area combined with NLR detection in severe pancreatitis
-
摘要:
目的 探讨胰腺CT密度和最大截面积联合中性粒细胞淋巴细胞比值(NLR)检测在重症胰腺炎预后的评估价值。 方法 选取2020年6月~2022年1月我院收治的46例重症胰腺炎患者作为观察组,另选同期40例健康体测者作为对照组,对所有研究对象进行CT检查,结合Image J计算胰腺CT密度和最大截面积,检测NLR,利用ROC曲线分析三者联合对重症胰腺炎预后评估的价值。 结果 观察组患者胰腺CT密度小于对照组(P < 0.05),最大截面积大于对照组(P < 0.05);观察组患者NLR高于对照组(P < 0.05),BISAP评分较高;CT密度和最大截面积评估重症胰腺炎预后的曲线下面积为0.786,敏感度为80.31%,特异性为80.84%;NLR评估重症胰腺炎预后的曲线下面积为0.775,敏感度为62.51%,特异性为56.27%;CT密度及最大截面积联合NLR评估重症胰腺炎预后的曲线下面积为0.884,敏感度为87.51%,特异性为82.15%。 结论 利用胰腺CT密度和最大截面积联合NLR检测对重症胰腺炎预后进行评估具有一定的临床应用价值。 -
关键词:
- CT密度 /
- 最大截面积 /
- 中性粒细胞淋巴细胞比值检测 /
- 重症胰腺炎 /
- 预后
Abstract:Objective To investigate the prognostic value of pancreatic CT density and maximum cross-sectional area combined with neutrophil lymphocyte ratio (NLR) detection in severe pancreatitis. Methods Forty-six patients with severe pancreatitis who admitted to our hospital from November 2019 to November 2021 were selected, and 40 healthy subjects were selected as the control group during the same period. CT examination was performed on all subjects, combined with Image J to calculate the CT density and maximum cross-sectional area of the pancreas, and NLR was detected. Results The CT density of the pancreas in the observation group was significantly lower than that in the control group (P < 0.05). The maximum cross-sectional area was significantly larger than that in the control group (P < 0.05). The NLR in the observation group was significantly higher than that in the control group (P < 0.05), and the score was higher. The area under the curve of CT density and maximum cross-sectional area for evaluating the prognosis of severe pancreatitis was 0.786, the sensitivity was 80.31%, and the specificity was 80.84%. The area under the curve of NLR for evaluating the prognosis of severe pancreatitis was 0.775, and the sensitivity was 62.51%, specificity was 56.27%. The area under the curve of CT density and maximum cross-sectional area combined with NLR to evaluate the prognosis of severe pancreatitis was 0.884, the sensitivity was 87.51%, and the specificity was 82.15%. Conclusion The use of pancreatic CT density and maximum cross-sectional area combined with NLR detection has a certain clinical value in evaluating the prognosis of severe pancreatitis. -
表 1 两组患者胰腺CT密度及最大截面积比较
Table 1. Comparison of CT density and maximum cross- sectional area of pancreas between the two groups (Mean±SD)
组別 CT密度(unit) 最大截面积(unit) 观察组(n=46) 105.27±10.63 4627.25±532.84 对照组(n=40) 122.05±15.34 1352.33±367.19 t 5.957 32.692 P < 0.001 < 0.001 表 2 两组患者NLR及BISAP评分比较
Table 2. Comparison of NLR and bisap scores between the two groups (Mean±SD)
组別 NLR BISAP评分(分) 观察组(n=46) 5.36±1.50 3.65±0.64 对照组(n=40) 1.68±0.57 - t 14.617 P < 0.001 NLR: 中性粒细胞淋巴细胞比值. 表 3 胰腺CT密度和最大截面积联合NLR评估重症胰腺炎预后的价值
Table 3. Value of pancreatic CT density and maximum cross-sectional area combined with NLR in evaluating the prognosis of severe pancreatitis
变量 SE AUC(95%CI) P 敏感度(%) 特异度(%) CT密度+最大截面积 0.0316 0.786 0.007 80.31 80.84 NLR 0.0405 0.775 0.015 62.51 56.27 CT密度+最大截面积+NLR 0.0187 0.884 < 0.001 87.51 82.15 -
[1] 蒋鑫, 徐欢, 严永峰, 等. 酒精性急性胰腺炎的临床及预后特征分析[J]. 临床肝胆病杂志, 2019, 35(11): 2528-32. doi: 10.3969/j.issn.1001-5256.2019.11.027 [2] 丁伟超, 许铁, 燕宪亮, 等. 老年重症急性胰腺炎病人的临床特点及预后分析[J]. 实用老年医学, 2019, 33(5): 505-7. doi: 10.3969/j.issn.1003-9198.2019.05.025 [3] 赵彦, 刘俐, 吕辰艳, 等. 重症胰腺炎患者预后生存情况及与其临床特征的相关性研究[J]. 解放军医药杂志, 2020, 32(5): 78-81, 98. doi: 10.3969/j.issn.2095-140X.2020.05.019 [4] 李成, 王丽, 李建红, 等. 血清甘油三酯与IL-6及APACHEⅡ评分对重症急性胰腺炎患者感染及预后的预测价值[J]. 中华医院感染学杂志, 2020, 30(20): 3125-9. https://www.cnki.com.cn/Article/CJFDTOTAL-ZHYY202020021.htm [5] 姚伟, 孔雷, 吴庆华, 等. 早期腹部CT检查在制定急性胆源性胰腺炎诊疗方案中的作用[J]. 外科理论与实践, 2019, 24(3): 236-41. https://www.cnki.com.cn/Article/CJFDTOTAL-WKLL201903018.htm [6] Favre PA, de Molliens L, Petit L, et al. May the neutrophil-to-lymphocyte ratio at admission predict the occurrence and the severity of ARDS after blunt chest trauma patients? A retrospective study[J]. Am J Emerg Med, 2021, 39: 137-42. doi: 10.1016/j.ajem.2020.09.050 [7] 黄景玉, 刘红书. 多层螺旋CT检查联合生化指标检测在急性胰腺炎诊断中的应用价值[J]. 影像科学与光化学, 2020, 38(4): 637-41. https://www.cnki.com.cn/Article/CJFDTOTAL-GKGH202004008.htm [8] 裴建军, 董齐, 董明, 等. CT分类标准在评估重症急性胰腺炎预后中的价值[J]. 中华外科杂志, 2010, 48(15): 1133-6. doi: 10.3760/cma.j.issn.0529-5815.2010.15.004 [9] 孟红兵, 曹兴健, 范玉萍, 等. Balthazar CT评分联合血浆D-二聚体对重症急性胰腺炎患者预后的预测价值[J]. 检验医学与临床, 2019, 16 (8): 1082-5. doi: 10.3969/j.issn.1672-9455.2019.08.021 [10] Ünal Y, Barlas AM. Role of increased immature granulocyte percentage in the early prediction of acute necrotizing pancreatitis[J]. Ulus Travma Acil Cerrahi Derg, 2019, 25(2): 177-82. [11] 张治琴, 魏茂碧, 王白莹, 等. 中性粒细胞/淋巴细胞比值、红细胞分布宽度联合C反应蛋白对急性胰腺炎并发严重急性肾损伤的预测价值[J]. 临床肾脏病杂志, 2020, 20(11): 891-6. doi: 10.3969/j.issn.1671-2390.2020.11.008 [12] 刘媛媛, 祝壮, 王秋月, 等. 中性粒细胞/淋巴细胞比值与急性胰腺炎严重程度及全身并发症的关系[J]. 临床消化病杂志, 2020, 32(2): 108-12. doi: 10.3870/lcxh.j.issn.1005-541X.2020.02.09 [13] O'Connell RM, Boland MR, O'Driscoll J, et al. Red cell distribution width and neutrophil to lymphocyte ratio as predictors of outcomes in acute pancreatitis: a retrospective cohort study[J]. Int J Surg, 2018, 55: 124-7. doi: 10.1016/j.ijsu.2018.05.028 [14] 尚鑫, 王静. 重症急性胰腺炎预后危险因素分析[J]. 临床医学研究与实践, 2021, 6(17): 28-30. https://www.cnki.com.cn/Article/CJFDTOTAL-YLYS202117010.htm [15] 曲娟, 杨继志. NLR联合RDW宽度对急性胰腺炎预后的预测价值探讨[J]. 世界华人消化杂志, 2018, 26(18): 1119-24. https://www.cnki.com.cn/Article/CJFDTOTAL-XXHB201818008.htm [16] 桂恩杨, 孙学虎, 王兴宇. 中性粒细胞与淋巴细胞比值作为重症急性胰腺炎早期预测指标的临床研究[J]. 肝胆外科杂志, 2021, 29(3): 187-90. doi: 10.3969/j.issn.1006-4761.2021.03.009 [17] 宁莹, 沙粒. RTR、NLR和BISAP评分判断急性胰腺炎患者病情和预后的临床意义[J]. 检验医学与临床, 2021, 18(17): 2528-32. doi: 10.3969/j.issn.1672-9455.2021.17.019 [18] 何海航, 张亚欣, 陈琳, 等. Balthazar CT分级联合中性粒细胞淋巴细胞比值在重症急性胰腺炎患者预后的预测作用[J]. 现代消化及介入诊疗, 2021, 26(10): 1303-7. doi: 10.3969/j.issn.1672-2159.2021.10.023 [19] 王东旭, 杨俊, 关雅迪, 等. CT在自身免疫性胰腺炎与胰腺癌鉴别诊断中的价值[J]. 中华实用诊断与治疗杂志, 2020, 34(11): 1161-4. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZD202011023.htm [20] 陈莹, 李越. BISAP评分联合血清TG、MAP1-LC3检测对急性重症胰腺炎患者病情及预后的评估价值[J]. 山东医药, 2020, 60(1): 21-4. doi: 10.3969/j.issn.1002-266X.2020.01.006 -







 下载:
下载: