Preparation and preliminary biological evaluation of DNA bipyramid fluorescent molecular probes
-
摘要:
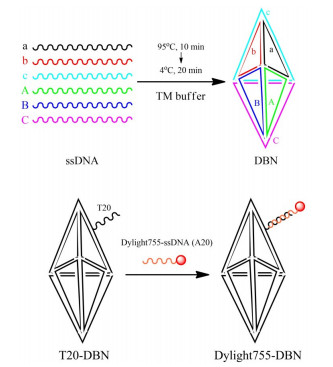
目的 构建基于DNA三角双锥纳米结构(DBN)的荧光分子探针,于活体动物水平探索探针的生物学性质。 方法 采用一步退火法,制备携带侧臂链的DBN;与DyLight 755偶联的寡聚核苷酸单链A20(DyLight 755-A20)混匀,制备荧光分子探针DyLight 755-DBN;将DyLight 755-DBN通过尾静脉注射实验鼠体内,于不同时间点处死,取感兴趣脏器测定荧光计数,探索分子探针的体内分布情况;将DyLight 755-DBN通过尾静脉注射体内,于不同时间点行小动物活体成像研究。 结果 成功制备携带侧臂链的DBN,利用聚丙烯酰胺凝胶电泳表征;通过与DyLight 755-A20等摩尔量杂交,成功制备荧光分子探针DyLight 755-DBN。体内分布实验显示DyLight 755-DBN进入体内后,荧光信号主要集中在肾脏、肝脏、脾脏、胃;注射分子探针5~15 min,在实验鼠的肺部有一定的荧光信号,但15 min后,荧光信号几乎没有。活体成像结果显示DyLight 755-DBN进入实验鼠体内主要集中在肝脏和胃,膀胱中维持较强的荧光信号。 结论 荧光分子探针DyLight 755-DBN是一种性质优良的分子影像探针。 -
关键词:
- DNA三角双锥纳米结构 /
- 荧光分子探针 /
- 体内分布 /
- 活体成像
Abstract:Objective To construct a kind of fluorescent molecular probes based on DNA Bipyramid Nanostructure (DBN), and to explore the biological properties of the probes at the level of living animals. Methods DBN with side arm chains, which was prepared through the one-step annealing method, was mixed with single-stranded oligonucleotide A20 coupled with DyLight 755 (DyLight 755-A20) to prepare fluorescent molecular probe DyLight 755-DBN. DyLight 755-DBN was injected into the mice through the tail vein, then the mice were executed at different time points, and fluorescence counts were taken from organs of interest to explore the in-vivo distribution of the molecular probes. DyLight 755-DBN was injected into the mouse through the tail vein, and in-vivo imaging studies of small animals were performed at different time points. Results The DBN with side arm chains was successfully prepared and characterized by polyacrylamide gel electrophoresis. The fluorescent molecular probe DyLight 755-DBN was successfully prepared by the equimolar hybridization of DBN with side arm chains and DyLight 755-A20. In vivo distribution experiments showed that after DyLight 755-DBN was entering the body, the fluorescent signal was mainly concentrated in the kidney, liver, spleen and stomach. There was a certain fluorescent signal in the lungs of experimental mouse during 5-15 min, but the fluorescence signal was almost absent after 15 min. In vivo imaging results showed that the fluorescent signal of DyLight 755-DBN was mainly concentrated in the liver and stomach, and a strong fluorescent signal in the bladder. Conclusion The fluorescent molecular probe DyLight 755-DBN is a kind of molecular imaging probe with excellent properties. -
表 1 DNA寡聚核苷酸单链的序列信息
Table 1. Sequence information of DNA oligonucleotide single strand
ssDNA 序列信息 a AGGCAGTTGAGACGAACATTCCTAAGTCTGAAATTTATCACCCGCCATAGTAGACGTATCACC b CTTGCTACACGATTCAGACTTAGGAATGTTCGACATGCGAGGGTCCAATACCGACGATTACAG c GGTGATAAAACGTGTAGCAAGCTGTAATCGACGGGAAGAGCATGCCCATCCACTACTATGGCG A CTGCGCGGATGACTCAACTGCCTGGTGATACGATCTAGTCTCTACGTCAAGTAAGAACCTTAG B CCTCGCATGACATCCGCGCAGCTAAGGTTCAAAGTTCCTGCCGCTTCACGGACGGTATTGGAC C CTCTTCCCGACCGTGAAGCGGCAGGAACTTATACTTGACGTAGAGACTAGAAGGATGGGCATG T20-a* TTTTTTTTTTTTTTTTTTTTCTAAGTCTGAAATTTATCACCCGCCATAGTA GACGTATCACCAGGCAGTTGAGACGAACATTC Dylight755-A20 Dylight755-AAAAAAAAAAAAAAAAAAAA -
[1] Ni JS, Li YX, Yue WT, et al. Nanoparticle- based cell trackers for biomedical applications[J]. Theranostics, 2020, 10(4): 1923-47. doi: 10.7150/thno.39915 [2] Wagner AM, Knipe JM, Orive G, et al. Quantum dots in biomedical applications[J]. Acta Biomater, 2019, 94: 44-63. doi: 10.1016/j.actbio.2019.05.022 [3] Lee SH, Jun BH. Silver nanoparticles: synthesis and application for nanomedicine[J]. Int J Mol Sci, 2019, 20(4): 865. doi: 10.3390/ijms20040865 [4] Wang ZH, Tang M. The cytotoxicity of core-shell or non-shell structure quantum dots and reflection on environmental friendly: a review[J]. Environ Res, 2021, 194: 110593. doi: 10.1016/j.envres.2020.110593 [5] Dong XM, Wu ZH, Li XP, et al. The size- dependent cytotoxicity of amorphous silica nanoparticles: a systematic review of in vitro studies[J]. Int J Nanomedicine, 2020, 15: 9089-113. doi: 10.2147/IJN.S276105 [6] Pérez-Garnes M, Gutiérrez-Salmerón M, Morales V, et al. Engineering hollow mesoporous silica nanoparticles to increase cytotoxicity[J]. Mater Sci Eng C Mater Biol Appl, 2020, 112: 110935. doi: 10.1016/j.msec.2020.110935 [7] Kahn JS, Minevich B, Gang O. Three-dimensional DNA-programmable nanoparticle superlattices[J]. Curr Opin Biotechnol, 2020, 63: 142-50. doi: 10.1016/j.copbio.2019.12.025 [8] Ma NN, Minevich B, Liu JL, et al. Directional assembly of nanoparticles by DNA shapes: towards designed architectures and functionality[J]. Top Curr Chem (Cham), 2020, 378(2): 36. doi: 10.1007/s41061-020-0301-0 [9] Kim KR, Jegal H, Kim J, et al. A self-assembled DNA tetrahedron as a carrier for in vivo liver-specific delivery of siRNA[J]. Biomater Sci, 2020, 8(2): 586-90. doi: 10.1039/C9BM01769K [10] Hong CY, Zhang XX, Dai CY, et al. Highly sensitive detection of multiple antibiotics based on DNA tetrahedron nanostructure-functionalized magnetic beads[J]. Anal Chim Acta, 2020, 1120: 50-8. doi: 10.1016/j.aca.2020.04.024 [11] Deng T. Construction and analysis of double helix for triangular bipyramid and pentangular bipyramid[J]. Comput Math Methods Med, 2020, 2020: 5609593. [12] Lin HY, Li J, Liu SY. The calculation of topological structures of strands-based DNA trigonal bipyramids[J]. J Mol Graph Model, 2020, 95: 107492. doi: 10.1016/j.jmgm.2019.107492 [13] Märcher A, Nijenhuis MAD, Gothelf KV. A wireframe DNA cube: antibody conjugate for targeted delivery of multiple copies of monomethyl auristatin E[J]. Angew Chem Int Ed Engl, 2021, 60(40): 21691-6. doi: 10.1002/anie.202107221 [14] Duan XY, Du YR, Wang CM, et al. Radiolabeling and preliminary evaluation of 99mTc- labeled DNA cube nanoparticles as potential tracers for SPECT imaging[J]. Int J Nanomed, 2021, 16: 5665-73. doi: 10.2147/IJN.S325791 [15] Hu QQ, Li H, Wang LH, et al. DNA nanotechnology- enabled drug delivery systems[J]. Chem Rev, 2019, 119(10): 6459-506. doi: 10.1021/acs.chemrev.7b00663 [16] Li JB, Jiang DW, Bao BL, et al. Radiolabeling of DNA bipyramid and preliminary biological evaluation in mice[J]. Bioconjug Chem, 2016, 27(4): 905-10. doi: 10.1021/acs.bioconjchem.5b00680 -







 下载:
下载: