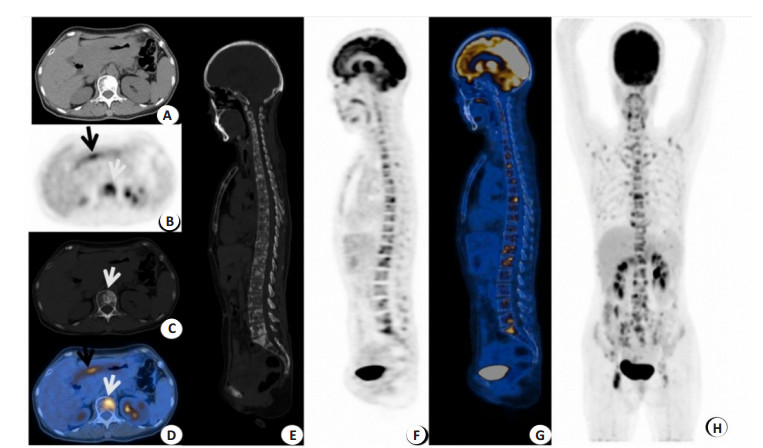
Clinical application of 18F-FDG PET/CT in bone metastases from gastric cancer
-
摘要:
目的 探讨胃癌骨转移患者的临床特征以及18F-FDG PET/CT在胃癌骨转移中的临床应用。 方法 选择2010年1月~2020年12月在中山大学肿瘤防治中心治疗前行18F-FDG PET/CT检查且原发灶经病理确诊的21例胃癌骨转移患者,其中男性9例、女性12例,中位年龄57.0(28.0,81.0)岁,回顾性分析其临床资料及18F-FDG PET/CT定性、半定量[最大标准化摄取值(SUVmax)] 指标特点。根据原发灶组织分化情况,将患者分为:低分化组13例(65.0%)和中-低分化组7例(35.0%)。根据Lauren分型,将患者分为:肠型组4例(20.0%),混合型组9例(45.0%),弥漫型组7例(35.0%)。根据Soloway分级将骨转移灶数量分为3组(Ⅰ级≤5,Ⅱ级6~20,Ⅲ级 > 20):Ⅰ级(组)6例,Ⅱ级(组)4例,Ⅲ级(组)11例。根据PET/CT上溶骨性、混合性及成骨性表现将骨转移类型分为3组:纯溶骨性转移6例,混合性(溶骨性/成骨性)11例,成骨性4例。 结果 本组胃癌患者发生骨转移部位依次为脊柱(19/21),肩胛骨、肋骨、锁骨、胸骨(18/21)及骨盆(17/21),其中3例伴有骨髓浸润。患者同期血清碱性磷酸酶升高11例(57.9%),乳酸脱氢酶升高7例(36.8%)。不同骨转移类型组间乳酸脱氢酶水平存在差异(χ2=6.823,P=0.047),混合性转移乳酸脱氢酶升高更常见。骨转移灶SUVmax大小在性别、Lauren分型及Soloway分级的差异有统计学意义(Z=-1.990,H=6.326,H= 6.070,P < 0.05);女性组中位SUVmax为11.6(7.3, 32.1),高于男性组的7.2(3.7, 17.1);弥漫型[12.2(5.3, 32.1)]及混合型组[10.8 (7.2, 17.2)]高于肠型组[6.7(3.7, 7.3)];Soloway分级越高,即骨转移数目越多,骨转移灶的SUVmax越高。Soloway分级在性别上存在差异(χ2=6.832,P=0.033),女性患者Soloway分级普遍高于男性。原发灶及骨转移灶SUVmax大小与年龄有关,低龄组(< 中位年龄57.0岁)骨转移灶SUVmax多较高而原发灶SUVmax多较低(χ2=5.838、10.831,P < 0.05)。 结论 18F-FDG PET/CT能够全面评估胃癌骨转移情况。胃癌骨转移具有一定的特点,常发生于中轴骨,以溶骨性及多灶转移多见,常伴有乳酸脱氢酶、碱性磷酸酶的升高。乳酸脱氢酶升高更常见于混合性骨转移患者。 Abstract:Objective To explore the clinical characteristics of patients with bone metastases from gastric cancer and the clinical application of 18F-FDG PET/CT in bone metastasis from gastric cancer. Methods From January 2010 to December 2020, 21 gastric cancer patients with bone metastases from gastric cancer who had undergone 18F-FDG PET/CT before treatment at the Cancer Prevention and Treatment Centre of Sun Yat-sen University and whose primary foci were pathologically confirmed, including 9 males and 12 females with a median age of 57.0 (28.0, 81.0) years old. The clinical data and the characteristics of the 18F-FDG PET/CT qualitative and semi-quantitative (maximum standardized uptake value SUVmax) index were retrospectively analyzed. According to the tissue differentiation of the primary foci, the patients were divided into: 13 cases (65.0%) in the hypofractionated group and 7 cases (35.0%) in the intermediate-hypofractionated group. According to the Lauren typing, the patients were divided into: 4 cases (20.0%) in the intestinal group, 9 cases (45.0%) in the mixed group and 7 cases (35.0%) in the diffuse group. According to Soloway classification, the number of bone metastases was divided into 3 groups (class Ⅰ≤5, class Ⅱ 6-20, class Ⅲ > 20), including 6 cases of class (group) Ⅰ, 4 cases of class (group) Ⅱ, and 11 cases of class (group) Ⅲ. Bone metastasis types were divided into 3 groups according to the manifestations on PET/CT (osteolytic, mixed, and osteogenic). There were 6 cases of pure osteolytic metastasis, 11 cases of mixed(osteolytic/osteogenic), and 4 cases of osteogenic metastasis. Results In this group of patients with gastric cancer, bone metastases occurred in the spine (19/21), scapula, ribs, collarbone, sternum (18/21), and pelvis (17/21), of which 3 cases were accompanied by bone marrow infiltration. The serum alkaline phosphatase and lactate dehydrogenase were elevated in 11 cases (57.9%) and 7 cases (36.8%) during the same period. There were differences in lactate dehydrogenase levels between different types of bone metastasis (χ2=6.823, P=0.047), with elevated lactate dehydrogenase being more common in mixed metastases. There were statistically significant differences in the SUVmax of metastatic bone lesions in gender, Lauren's classification, and Soloway classification (Z=-1.990, H=6.326, H=6.070, all P < 0.05). The median SUVmax 11.6 (7.3, 32.1) in the female group was higher than that in the male group [7.2 (3.7, 17.1)]; The diffuse-type group [12.2 (5.3, 32.1)] and the mixed-type group [10.8 (7.2, 17.2)] were higher than the intestinal-type group [6.7 (3.7, 7.3)]. The higher the Soloway classification, i.e. the higher the number of bone metastases, the higher the SUVmax of metastatic bone lesions. There was a difference in the Soloway grading by gender (χ2=6.832, P=0.033), with female patients generally having a higher Soloway grade than males. Primary and bone metastases SUVmax size was age dependent, with the lower age group (< median age 57.0 years) having higher bone metastases SUVmax and lower primary SUVmax. (χ2=5.838, 10.831, P < 0.05). Conclusion 18F-FDG PET/CT can provide a comprehensive assessment of bone metastases from gastric cancer. Bone metastasis from gastric cancer have specific characteristics, such as often occurring in the mid-axis bone, with osteolytic and multifocal metastases commonly and are often associated with elevated lactate dehydrogenase and alkaline phosphatase. Elevated lactate dehydrogenase is more common in patients with mixed bone metastases. -
Key words:
- gastric cancer /
- bone metastasis /
- PET/CT /
- 18F-FDG
-
表 1 21例胃癌骨转移患者临床及肿瘤特征
Table 1. Clinical and tumor characteristics of 21 cases gastric cancer patients with bone metastasis
一般情况 统计值 肿瘤情况 统计值[n (%)] 年龄(岁) 57.0 (28.0, 81.0) 骨痛 性别 有 7 (33.3) 男 9 (42.9) 无 11 (52.4) 女 12(57.1) 不详 3 (14.3) 红细胞(×109/L) 4.2 (1.3, 5.4) 骨折 下降 7(36.8) 有 0 (0) 正常或升高 12(63.2) 无 21 (100) 血小板(×109/L) 212.7±115.3 原发灶部位 下降 4(21.1) 上1/3 4 (19.0) 正常或升高 15(78.9) 中1/3 6 (28.6) 白细胞(×109/L) 7.9±2.2 下1/3 11 (52.4) 升高 4(21.1) 淋巴结转移 正常 15(78.9) 区域 2 (9.5) 血钙(mmol/L) 2.2±0.1 远处 14 (66.7) 下降 4(21.1) 无 5 (23.8) 正常 15(78.9) 远处转移(n) ALP (U/L) 173.6 (44.9, 926.4) 肝 5 升高 11 (57.9) 肾上腺 4 正常 8 (42.1) 卵巢 4 LDH (U/L) 238.6 (123.5, 1176.9) 肺 1 升高 7(36.8) 腹膜 6 正常 12(63.2) ALP: 碱性磷酸酶; LDH: 乳酸脱氢酶; 服从近似正态分布的计量资料以均数±标准差表示, 不服从近似正态分布的资料以中位数(四分位数间距)表示. 表 2 21例胃癌骨转移患者临床、病理特征在定性、半定量参数间差异
Table 2. Differences in clinical and pathological characteristics between qualitative and semiquantitative parameters in 21 cases gastric cancer patients with bone metastasis.
因素 骨转移灶 原发灶SUVmax 中位SUVmax 骨转移类型 SUVmax 骨转移灶 原发灶 H/χ2 P t/Z/H P t/Z/H P χ2 P χ2 P 年龄 4.781 0.112 -1.830 0.072 -3.310 0.001 5.838 0.030 10.831 0.002 性别 2.099 0.504 -1.990 0.049 -1.421 0.155 5.743 0.030 0.398 0.670 组织分化 0.189 0.189 0.803 0.433 -1.545 0.122 0.220 1.000 5.495 0.057 Lauren分型 5.120 0.217 6.326 0.034 2.204 0.332 5.143 0.076 2.397 0.302 Soloway分级 7.793 0.117 6.070 0.041 4.121 0.217 4.093 0.086 1.440 0.487 淋巴结转移 0.245 1.000 1.487 0.153 0.226 0.823 0.153 1.000 2.759 0.149 远处淋巴结转移 0.170 1.000 1.111 0.280 -0.054 0.958 0.095 1.000 2.386 0.183 远处转移 1.357 0.598 -0.048 0.962 -1.350 0.177 0.064 1.000 0.398 0.670 红细胞 2.645 0.391 0.960 0.350 -0.845 0.432 2.100 0.350 0.095 0.953 血小板 2.454 0.368 -0.550 0.305 -1.576 0.133 3.958 0.138 4.293 0.064 白细胞 1.619 0.777 -0.800 0.469 0.000 1.000 3.026 0.220 0.019 0.990 血钙 2.454 0.368 -0.105 0.307 -1.400 0.185 1.017 0.582 1.017 0.582 ALP 6.807 0.056 -0.304 0.765 -1.651 1.109 2.048 0.359 1.273 0.529 LDH 6.823 0.047 -0.772 0.451 -2.028 0.045 4.250 0.650 4.866 0.057 原发灶部位 2.048 0.689 3.299 0.196 1.850 0.186 1.775 0.412 10.066 0.007 骨转移类型 - - 3.316 0.195 1.636 0.201 1.775 0.412 3.091 0.133 原发灶SUVmax 4.187 0.122 - - - - - - - - -
[1] Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018[J]. CA Cancer J Clin, 2018, 68(1): 7-30. doi: 10.3322/caac.21442 [2] Thomassen I, van Gestel YR, van Ramshorst B, et al. Peritoneal carcinomatosis of gastric origin: a population-based study on incidence, survival and risk factors[J]. Int J Cancer, 2014, 134(3): 622-8. doi: 10.1002/ijc.28373 [3] Ma DW, Kim JH, Jeon TJ, et al. 18F-fluorodeoxyglucose positron emission tomography-computed tomography for the evaluation of bone metastasis in patients with gastric cancer[J]. Dig Liver Dis, 2013, 45(9): 769-75. doi: 10.1016/j.dld.2013.02.009 [4] Yoshioka T, Yamaguchi K, Kubota K, et al. Evaluation of 18F-FDG PET in patients with advanced, metastatic, or recurrent gastric cancer[J]. J Nucl Med, 2003, 44(5): 690-9. http://www.researchgate.net/profile/Takashi_Yoshioka/publication/10772175_Evaluation_of_18F-FDG_PET_in_Patients_with_Advanced_Metastatic_or_Recurrent_Gastric_Cancer/links/00b7d524a0a3fd3d05000000.pdf [5] Takenaka D, Ohno Y, Matsumoto K, et al. Detection of bone metastases in non-small cell lung cancer patients: comparison of whole-body diffusion-weighted imaging (DWI), whole-body MR imaging without and with DWI, whole-body FDG-PET/CT, and bone scintigraphy[J]. J Magn Reson Imaging, 2009, 30(2): 298-308. doi: 10.1002/jmri.21858 [6] Yoo SW, Chowdhury MSA, Jeon S, et al. Clinical impact of F-18 FDG PET-CT on biopsy site selection in patients with suspected bone metastasis of unknown primary site[J]. Nucl Med Mol Imaging, 2020, 54(4): 192-8. doi: 10.1007/s13139-020-00649-4 [7] Başdelioğlu K. Bone metastasis: evaluation of 1100 patients with breast cancer[J]. Int J Clin Exp Pathol, 2021, 14(3): 338-47. http://www.ncbi.nlm.nih.gov/pubmed/33786150 [8] Hong LD, Cristiano L, Peters E, et al. Detection of bone metastases in uterine cancer: how common are they and should PET/CT be the standard for diagnosis?[J]. Gynecol Oncol Rep, 2021, 35: 100698. doi: 10.1016/j.gore.2021.100698 [9] Otis-Chapados S, Goulet CR, Dubois G, et al. 18F-fluorodeoxyglucose positron emission tomography/computed tomography (PET/CT) is accurate for high-grade prostate cancer bone staging when compared to bone scintigraphy[J]. Can Urol Assoc J, 2021, 15(10): 301-7. http://www.researchgate.net/publication/350179467_18F-Fluorodeoxyglucose_positron_emission_tomographycomputed_tomography_PETCT_is_accurate_for_high-grade_prostate_cancer_bone_staging_when_compared_to_bone_scintigraphy [10] 中国临床肿瘤学会指南工作委员会组织. 中国临床肿瘤学会(CSCO) 胃癌诊疗指南-2018. V1[M]. 北京: 人民卫生出版社, 2018. [11] Namikawa T, Okabayshi T, Nogami M, et al. Assessment of (18)F-fluorodeoxyglucose positron emission tomography combined with computed tomography in the preoperative management of patients with gastric cancer[J]. Int J Clin Oncol, 2014, 19(4): 649-55. doi: 10.1007/s10147-013-0598-6 [12] Arslan E, Aksoy T, Gündoğan C, et al. Metabolic characteristics and diagnostic contribution of 18F-FDG PET/CT in gastric carcinomas[J]. Mol Imaging Radionucl Ther, 2020, 29(1): 25-32. doi: 10.4274/mirt.galenos.2020.75537 [13] Maman A, Sahin A, Ayan AK. The relationship of SUV value in PET-CT with tumor differentiation and tumor markers in gastric cancer[J]. Eurasian J Med, 2020, 52(1): 67-72. doi: 10.5152/eurasianjmed.2019.19016 [14] Bosch KD, Chicklore S, Cook GJ, et al. Staging FDG PET-CT changes management in patients with gastric adenocarcinoma who are eligible for radical treatment[J]. Eur J Nucl Med Mol Imaging, 2020, 47(4): 759-67. doi: 10.1007/s00259-019-04429-x [15] Ma DW, Kim JH, Jeon TJ, et al. 18F-fluorodeoxyglucose positron emission tomography-computed tomography for the evaluation of bone metastasis in patients with gastric cancer[J]. Dig Liver Dis, 2013, 45(9): 769-75. doi: 10.1016/j.dld.2013.02.009 [16] Soloway MS, Hardeman SW, Hickey D, et al. Stratification of patients with metastatic prostate cancer based on extent of disease on initial bone scan[J]. Cancer, 1988, 61(1): 195-202. doi: 10.1002/1097-0142(19880101)61:1<195::AID-CNCR2820610133>3.0.CO;2-Y [17] Kusumoto H, Haraguchi M, Nozuka Y, et al. Characteristic features of disseminated carcinomatosis of the bone marrow due to gastric cancer: the pathogenesis of bone destruction[J]. Oncol Rep, 2006, 16(4): 735-40. http://www.spandidos-publications.com/or/16/4/735/download [18] Ahn JB, Ha TK, Kwon SJ. Bone metastasis in gastric cancer patients[J]. J Gastric Cancer, 2011, 11(1): 38-45. doi: 10.5230/jgc.2011.11.1.38 [19] Turkoz FP, Solak M, Kilickap S, et al. Bone metastasis from gastric cancer: the incidence, clinicopathological features, and influence on survival[J]. J Gastric Cancer, 2014, 14(3): 164-72. doi: 10.5230/jgc.2014.14.3.164 [20] Zhou Z, Sehn LH, Rademaker AW, et al. An enhanced International Prognostic Index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era[J]. Blood, 2014, 123(6): 837-42. doi: 10.1182/blood-2013-09-524108 [21] Evangelista L, Panunzio A, Polverosi R, et al. Early bone marrow metastasis detection: the additional value of FDG-PET/CT vs. CT imaging[J]. Biomed Pharmacother, 2012, 66(6): 448-53. doi: 10.1016/j.biopha.2012.06.004 [22] Budak E, Yanarateş A. Papel de la PET/TC con 18F-FDG en la detección del tumor primario en pacientes con metástasis óseas de Origen desconocido[J]. Revista Española De Med Nucl E Imagen Mol, 2020, 39(1): 14-9. doi: 10.1016/j.remn.2019.06.002 [23] Yoshikawa K, Kitaoka H. Bone metastasis of gastric cancer[J]. Jpn J Surg, 1983, 13(3): 173-6. doi: 10.1007/BF02469472 [24] Koemans WJ, Luijten JCHBM, van der Kaaij RT, et al. The metastatic pattern of intestinal and diffuse type gastric carcinoma - A Dutch national cohort study[J]. Cancer Epidemiol, 2020, 69: 101846. doi: 10.1016/j.canep.2020.101846 [25] Findlay JM, Antonowicz S, Segaran A, et al. Routinely staging gastric cancer with 18F-FDG PET-CT detects additional metastases and predicts early recurrence and death after surgery[J]. Eur Radiol, 2019, 29(5): 2490-8. doi: 10.1007/s00330-018-5904-2 [26] Bergers G, Fendt SM. The metabolism of cancer cells during metastasis[J]. Nat Rev Cancer, 2021, 21(3): 162-80. doi: 10.1038/s41568-020-00320-2 [27] Sun ZQ, Jin LF, Zhang S, et al. Preoperative prediction for lauren type of gastric cancer: a radiomics nomogram analysis based on CT images and clinical features[J]. J Xray Sci Technol, 2021, 29(4): 675-86. http://www.ncbi.nlm.nih.gov/pubmed/34024809 [28] Pang YZ, Zhao L, Luo ZM, et al. Comparison of 68 Ga-FAPI and 18F-FDG uptake in gastric, duodenal, and colorectal cancers[J]. Radiology, 2021, 298(2): 393-402. doi: 10.1148/radiol.2020203275 [29] Qin CX, Shao FQ, Gai YK, et al. 68 Ga-DOTA-FAPI-04 PET/MR in the evaluation of gastric carcinomas: comparison with 18F-FDG PET/CT[J]. J Nucl Med, 2022, 63(1): 81-8. doi: 10.2967/jnumed.120.258467 -







 下载:
下载: