Value of diffusion tensor imaging in the diagnosis and prognosis of neonatal hypoxic-ischemic encephalopathy
-
摘要:
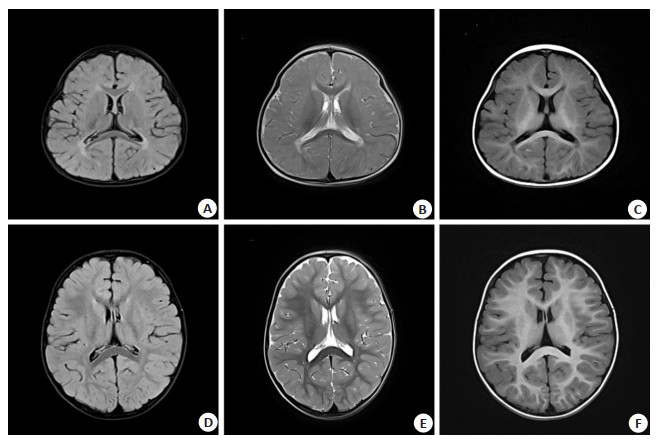
目的 分析扩散张量成像(DTI)在新生儿缺氧缺血性脑病诊断中对脑白质的损伤和预后的价值。 方法 将本院于2018年8月~2021年6月收治的60例行MRI检查的新生儿作为研究对象,年龄均 < 2周岁,且均有缺氧缺血性脑病史。将患者分为脑瘫组和非脑瘫组(30例/组),记录其常规颅脑平扫征象和得分,于DTI图像上找出感兴趣区域并测定各向异性分数(FA)值和表观弥散系数(ADC)值。测定的白质区包括:内囊前肢、丘脑后辐射、额叶白质、中脑大脑脚、胼胝体压部、下纵束、胼胝体体部、内囊后肢、视辐射、内囊膝部、皮质脊髓束、半卵圆中心、听辐射、胼胝体膝部和顶叶白质等。生成胼胝体纤维束和皮质脊髓束的三维图像。对比分析两组患儿平扫的征象和FA值与ADC值;采用Logistic回归分析多因素筛选脑白质损伤的独立影响因素,并评估其拟合度和效能。 结果 经MRI平扫,两组患儿颅脑左侧的丘脑、两侧的半卵圆中心区和胼胝体体部的征象差异具有统计学意义(P < 0.05);其中脑瘫组患儿各个部位的FA值均比非脑瘫组小,且两组患儿的FA值在内囊后肢、听辐射、双侧中脑大脑脚、视辐射、顶叶白质、皮质脊髓束、胼胝体压部、下纵束、半卵圆中心、胼胝体膝部、丘脑后辐射、胼胝体体部和左侧豆状核的差异具有统计学意义(P < 0.05);两组患儿的ADC值在双侧半卵圆中心、胼胝体体部、左侧内囊后肢、左侧丘脑后辐射、双侧皮质脊髓束、左侧顶叶及压部和左侧丘脑差异具有统计学意义(P < 0.05);左侧内囊后肢、胼胝体压部和左侧皮质脊髓束的FA值可作为脑白质损伤的独立预测因素,三者联合用于缺氧缺血性脑病患儿脑白质损伤的ROC AUC是0.887,特异性和敏感度分别是90.38% 和85.12%,准确度为0.761。 结论 缺氧缺血性脑病会使其白质纤维束发生损伤,使其FA值降低,ADC值升高,DTI可明确反映出患儿神经纤维束发育和受损的程度,可作为缺氧缺血性脑病的诊断和预后依据。 Abstract:Objective To explore the value of diffusion tensor imaging (DTI) in the diagnosis of cerebral white matter injury and prognosis of neonates with hypoxic-ischemic encephalopathy. Methods Sixty neonates undergoing MRI examination admitted to our hospital from August 2018 to June 2021 were selected as the research subjects. All patients were younger than 2 years old and had a history of hypoxic-ischemic encephalopathy. The patients were divided into two groups, the signs and scores of conventional craniocerebral scans were recorded. The regions of interest were identified on DTI images, the values of FA and ADC were measured. The measured areas of white matter including forelimbs of the internal capsule, craniocerebrum of the midbrain, hind limbs of the internal capsule, knee of the internal capsule, corticospinal tract, center of half ovale, retrothalamic radiation, inferior longitudinal tract, auditory radiation, visual radiation, corpus callosum body, knee of the corpus callosum, frontal white matter, pressure of the corpus callosum, and white matter of the parietal lobe. Three-dimensional images of the fibrous tracts of the corpus callosum and the corticospinal tracts were produceded. The plain scan signs, FA values and ADC values of the two groups were compared and analyzed. The independent influencing factors of white matter injury was screened by multivariate logistic regression analysis, the fitting degree and effectiveness were evaluated. Results Brain MRI plain scan showed significant differences in the signs of left thalamus, bilateral center of semi- ovale and corpus callosum between the two groups (P < 0.05). The FA value of each part of the group of children with cerebral palsy were smaller than the cerebral palsy group, sac and FA values of two groups of children, hind legs, bilateral middle cerebral cerebral peduncle, corticospinal tract, half egg circle center, thalamus after radiation, the longitudinal fasciculus, listen to the radiation, radiation, corpus callosum knee, parietal white matter, corpus callosum, the body and the left side of the corpus callosum lentiform nucleus have significant difference (P < 0.05). There were significant differences in ADC values between the two groups in bilateral semi-ovale center, left posterior limb of internal capsule, left posterior thalamic radiation, bilateral corticospinal tract, left parietal lobe, corpus callosum body part and compression part, and left thalamus (P < 0.05). Multivariate regression showed that the FA values of the left posterior limb of the internal capsule, the left cortical cord and the corpus callosum were independent predictors of white matter injury. The ROC AUC of the three factors combined in the treatment of white matter injury in children with hypoxic- ischemic encephalopathy was 0.887, the specificity and sensitivity were 90.38% and 85.12% respectively, and the accuracy was 0.761. Conclusion Hypoxic-ischemic encephalopathy can damage the white matter fiber bundles, decrease the FA value and increase the ADC value. DTI can clearly reflect the development and damage degrees of the nerve fiber bundles in children, which can be used as the diagnosis and prognosis basis of hypoxic-ischemic encephalopathy. -
表 1 两组患儿一般资料比较
Table 1. Comparison of general data between the two groups (n= 30, Mean±SD)
组别 脑瘫组 非脑瘫组 t/χ2 P 性别(男/女, n) 18/12 20/10 0.000 1.000 胎龄(周) 34.8±3.4 36.5±3.1 -1.614 0.127 体质量(kg) 2.66±0.75 3.15±1.13 -0.875 0.393 检测周龄(月) 22.3±16.6 16.9±13.6 -1.057 0.284 表 2 两组患儿颅脑MRI平扫结果比较
Table 2. Comparison of brain MRI plain scan results between the two groups [n=30, n(%)]
组别 脑瘫组 非脑瘫组 χ2 P 右侧中脑大脑脚 21(70.0) 24(80.0) 0.524 0.456 左侧中脑大脑脚 20(66.7) 16(53.3) 0.912 0.328 右侧内囊后肢 25(83.3) 26(86.7) 0.164 0.668 左侧内囊后肢 23(76.7) 22(73.3) 0.134 0.702 右侧半卵圆中心 15(50.0) 0(0.0) 13.324 < 0.001 左侧半卵圆中心 17(56.7) 7(23.3) 6.658 0.011 右侧豆状核 13(43.3) 13(43.3) 0 1.012 左侧豆状核 14(46.7) 11(36.7) 0.408 0.521 右侧尾状核 4(13.3) 0(0.0) 1.432 0.229 左侧尾状核 4(13.3) 0(0.0) 1.432 0.229 右侧丘脑 7(23.3) 0(0.0) 3.648 0.047 左侧丘脑 13(43.3) 0(0.0) 7.647 0.007 胼胝体膝部 6(20.0) 6(20.0) 0 1.012 胼胝体体部 28(93.3) 19(63.3) 4.812 0.019 胼胝体压部 14(46.7) 7(23.3) 1.017 0.302 表 3 两组患儿各部位的FA值比较
Table 3. Comparison of FA values of different parts between the two groups (n=30, Mean±SD)
部位 脑瘫组 非脑瘫组 t P 右侧中脑大脑脚 0.45±0.08 0.53±0.04 -2.618 0.013 左侧中脑大脑脚 0.45±0.08 0.53±0.03 -2.918 0.007 右侧内囊前肢 0.30±0.07 0.33±0.05 -0.552 0.579 左侧内囊前肢 0.31±0.08 0.35±0.04 -0.988 0.325 右侧内囊膝部 0.37±0.07 0.40±0.04 -0.715 0.486 左侧内囊膝部 0.36±0.07 0.38±0.03 -0.075 0.934 右侧内囊后肢 0.43±0.09 0.53±0.03 -3.966 < 0.001 左侧内囊后肢 0.42±0.08 0.54±0.03 -5.344 < 0.001 右侧半卵圆中心 0.37±0.12 0.53±0.03 -4.834 < 0.001 左侧半卵圆中心 0.34±0.12 0.51±0.05 -5.271 < 0.001 右侧皮质脊髓束 0.43±0.08 0.52±0.04 -3.488 0.002 左侧皮质脊髓束 0.43±0.07 0.53±0.04 -4.487 < 0.001 右侧下纵束 0.36±0.07 0.44±0.04 -3.558 0.002 左侧下纵束 0.36±0.06 0.43±0.05 -2.674 0.012 右侧丘脑后辐射 0.37±0.07 0.48±0.04 -5.166 < 0.001 左侧丘脑后辐射 0.36±0.07 0.50±0.05 -6.138 < 0.001 右侧视辐射 0.39±0.07 0.45±0.05 -2.542 0.016 左侧视辐射 0.38±0.07 0.48±0.05 -3.569 0.002 右侧听辐射 0.32±0.07 0.40±0.04 -3.865 < 0.001 左侧听辐射 0.33±0.05 0.42±0.05 -4.477 < 0.001 右侧额叶白质 0.33±0.08 0.37±0.03 -1.187 0.240 左侧额叶白质 0.32±0.10 0.38±0.04 -1.838 0.074 右侧顶叶白质 0.35±0.07 0.40±0.03 -2.173 0.036 左侧顶叶白质 0.35±0.09 0.41±0.02 -2.405 0.022 胼胝体膝部 0.47±0.06 0.57±0.03 -5.208 < 0.001 胼胝体体部 0.37±0.09 0.55±0.08 -5.775 < 0.001 胼胝体压部 0.49±0.08 0.65±0.05 -6.788 < 0.001 右侧豆状核 0.29±0.07 0.30±0.03 0.705 0.481 左侧豆状核 0.27±0.07 0.33±0.03 -2.409 0.021 右侧尾状核 0.27±0.09 0.32±0.03 -1.682 0.100 左侧尾状核 0.26±0.07 0.30±0.04 -1.375 0.175 右侧丘脑 0.32±0.07 0.36±0.03 -1.146 0.256 左侧丘脑 0.29±0.07 0.33±0.05 -1.236 0.222 右前扣带回 0.34±0.08 0.37±0.03 -0.703 0.482 左前扣带回 0.38±0.17 0.38±0.03 0.287 0.770 右后扣带回 0.30±0.08 0.36±0.04 -1.755 0.087 左后扣带回 0.30±0.10 0.35±0.05 -1.263 0.212 表 4 两组患儿各部位的ADC值比较
Table 4. Comparison of ADC values of different parts between the two groups (n=30, Mean±SD)
部位 脑瘫组 非脑瘫组 t P 右侧中脑大脑脚 1.05±0.15 0.95±0.19 1.596 0.120 左侧中脑大脑脚 1.02±0.13 0.93±0.14 1.647 0.109 右侧内囊前肢 0.91±0.07 0.91±0.08 -0.794 0.437 左侧内囊前肢 0.95±0.13 0.90±0.10 0.896 0.381 右侧内囊膝部 0.90±0.07 0.90±0.07 -0.846 0.408 左侧内囊膝部 0.94±0.09 0.9±0.11 0.742 0.468 右侧内囊后肢 0.91±0.12 0.84±0.08 1.436 0.161 左侧内囊后肢 0.93±0.12 0.81±0.08 2.688 0.009 右侧半卵圆中心 1.07±0.29 0.84±0.07 0.062 0.003 左侧半卵圆中心 1.12±0.48 0.86±0.08 2.164 0.037 右侧皮质脊髓束 0.97±0.11 0.89±0.07 2.253 0.030 左侧皮质脊髓束 0.99±0.11 0.88±0.09 2.764 0.008 右侧下纵束 1.03±0.13 0.98±0.11 0.694 0.498 左侧下纵束 1.07±0.12 1.01±0.13 1.013 0.322 右侧丘脑后辐射 1.07±0.19 0.95±0.16 1.872 0.069 左侧丘脑后辐射 1.20±0.28 1.01±0.09 2.296 0.026 右侧视辐射 0.95±0.06 0.93±0.14 0.072 0.941 左侧视辐射 1.02±0.10 1.01±0.09 -0.419 0.684 右侧听辐射 0.97±0.08 0.97±0.08 -0.591 0.564 左侧听辐射 0.99±0.09 0.97±0.09 0.191 0.857 右侧额叶白质 1.02±0.21 0.94±0.11 1.059 0.300 左侧额叶白质 1.09±0.47 0.95±0.07 1.079 0.291 右侧顶叶白质 1.01±0.10 0.94±0.14 1.759 0.087 左侧顶叶白质 1.02±0.12 0.91±0.12 2.055 0.047 胼胝体膝部 0.99±0.12 0.98±0.19 -0.194 0.857 胼胝体体部 1.39±0.33 1.15±0.14 2.512 0.016 胼胝体压部 1.10±0.14 0.96±0.11 2.661 0.011 右侧豆状核 0.93±0.15 0.89±0.11 0.579 0.573 左侧豆状核 0.98±0.15 0.93±0.11 0.674 0.509 右侧尾状核 0.93±0.12 0.87±0.09 0.929 0.363 左侧尾状核 0.94±0.20 0.94±0.27 -0.183 0.863 右侧丘脑 0.95±0.10 0.89±0.08 1.711 0.096 左侧丘脑 0.97±0.10 0.85±0.13 2.646 0.011 右前扣带回 0.99±0.10 0.94±0.09 0.839 0.412 左前扣带回 0.98±0.08 0.96±0.09 -0.198 0.851 右后扣带回 0.99±0.11 0.94±0.10 0.819 0.423 左后扣带回 0.98±0.12 0.93±0.10 0.676 0.509 表 5 Logistic模型的拟合度结果
Table 5. Fitting results of logistic model
组别 步骤1 步骤2 步骤3 步骤4 步骤5 步骤6 步骤7 步骤8 步骤9 步骤10 脑瘫组 实际值 2 4 5 6 6 8 11 10 13 15 期望值 1.014 4.022 3.943 4.120 4.656 7.450 11.880 9.532 11.467 13.285 非脑瘫组 实际值 9 11 12 14 17 14 14 18 12 11 期望值 8.734 9.616 11.047 12.905 15.880 14.043 12.476 16.422 12.536 11.921 总计 11 15 17 20 23 22 25 28 25 26 P 0.318 表 6 患儿引发脑瘫的多因素Logistic分析
Table 6. Multivariate logistic analysis of cerebral palsy in children
参数 标准误 回归系数 OR Waldχ2 95%CI P 上限 下限 常量 0.952 -1.224 - 12.743 - - < 0.001 胼胝体压部 0.282 0.364 0.401 21.317 0.105 0.602 < 0.001 左侧皮质脊髓束 1.232 0.260 0.536 14.324 0.158 0.788 < 0.001 左侧内囊后肢 0.768 0.301 0.523 16.683 0.123 0.648 < 0.001 表 7 三因素对缺氧缺血性脑病患儿脑白质损伤预后的价值分析
Table 7. Value analysis of the above three factors on the prognosis of white matter injury in children with HIE
参数 AUC(95% CI) 截点 标准误 准确度 特异性(%) 敏感度(%) P 胼胝体压部 0.667 0.48 0.078 0.547 85.72 69.88 < 0.001 左侧皮质脊髓束 0.812 0.54 0.058 0.588 79.87 79.89 < 0.001 左侧内囊后肢 0.683 0.49 0.073 0.572 85.72 72.48 < 0.001 三项联合 0.887 - 0.036 0.761 90.38 85.12 < 0.001 -
[1] 王葆辉, 耿丽娟, 刘俊. 亚低温治疗的缺氧缺血性脑病新生儿MRI弥散值与长期神经预后的相关性[J]. 中国急救医学, 2021, 41(6): 484-9. doi: 10.3969/j.issn.1002-1949.2021.06.006 [2] Back SA. White matter injury in the preterm infant: pathology and mechanisms[J]. Acta Neuropathol, 2017, 134(3): 331-49. doi: 10.1007/s00401-017-1718-6 [3] 马青, 聂芳. 缺氧缺血性脑病脑损伤早期诊断及预后评估影像学研究进展[J]. 医学影像学杂志, 2021, 31(3): 517-20. https://www.cnki.com.cn/Article/CJFDTOTAL-XYXZ202103044.htm [4] 马瑞, 马瑜徽, 张新月, 等. 不同褪黑素治疗方案对缺氧缺血性脑损伤新生大鼠脑白质损伤的影响[J]. 中国当代儿科杂志, 2021, 23(3): 300-5. https://www.cnki.com.cn/Article/CJFDTOTAL-DDKZ202103019.htm [5] 李刚, 郑继爱, 于鹏. MRI检查在新生儿缺氧缺血性脑病中的应用[J]. 中国妇幼保健, 2021, 36(1): 220-3. https://www.cnki.com.cn/Article/CJFDTOTAL-ZFYB202101074.htm [6] 王芳. 新生儿缺氧缺血性脑病的多种影像学表现[J]. 影像研究与医学应用, 2020, 4(21): 3-5. doi: 10.3969/j.issn.2096-3807.2020.21.002 [7] 潘鸿, 於志华, 侯春晓. 磁共振扩散张量成像技术在新生儿缺氧缺血性脑病诊断中的价值[J]. 中国妇幼保健, 2020, 35(21): 4136-9. https://www.cnki.com.cn/Article/CJFDTOTAL-ZFYB202021076.htm [8] Burns CM, Rutherford MA, Boardman JP, et al. Patterns of cerebral injury and neurodevelopmental outcomes after symptomatic neonatal hypoglycemia[J]. Pediatrics, 2008, 122(1): 65-74. doi: 10.1542/peds.2007-2822 [9] Trivedi SB, Vesoulis ZA, Rao R, et al. A validated clinical MRI injury scoring system in neonatal hypoxic-ischemic encephalopathy[J]. Pediatr Radiol, 2017, 47(11): 1491-9. doi: 10.1007/s00247-017-3893-y [10] 尉胜男, 张志红, 郑建民, 等. 新生儿HIE患儿MRI图像表现及与预后相关性研究[J]. 中国CT和MRI杂志, 2020, 18(8): 1-3. doi: 10.3969/j.issn.1672-5131.2020.08.001 [11] Huang BY, Castillo M. Hypoxic-ischemic brain injury: imaging findings from birth to adulthood[J]. RadioGraphics, 2008, 28(2): 417-39. doi: 10.1148/rg.282075066 [12] Salas J, Reddy N, Carson KA, et al. Ultrasound predicts white matter integrity after hypothermia therapy in neonatal hypoxic-ischemic injury[J]. J Neuroimaging, 2019, 29(6): 743-9. doi: 10.1111/jon.12644 [13] Shao R, Sun D, Hu Y, et al. White matter injury in the neonatal hypoxic-ischemic brain and potential therapies targeting microglia[J]. J Neurosci Res, 2021, 99(4): 991-1008. doi: 10.1002/jnr.24761 [14] Muttikkal TJ, Wintermark M. MRI patterns of global hypoxic-ischemic injury in adults[J]. J Neuroradiol, 2013, 40(3): 164-71. doi: 10.1016/j.neurad.2012.08.002 [15] 田静, 张靖, 任亚方, 等. NICU缺血缺氧性脑病新生儿MRI与CT诊断结果分析[J]. 中国CT和MRI杂志, 2020, 18(7): 7-10. doi: 10.3969/j.issn.1672-5131.2020.07.003 [16] Liauw L, van der Grond J, Slooff V, et al. Differentiation between peritrigonal terminal zones and hypoxic-ischemic white matter injury on MRI[J]. Eur J Radiol, 2008, 65(3): 395-401. doi: 10.1016/j.ejrad.2007.04.016 [17] Dorner RA, Burton VJ, Allen MC, et al. Preterm neuroimaging and neurodevelopmental outcome: a focus on intraventricular hemorrhage, post-hemorrhagic hydrocephalus, and associated brain injury[J]. J Perinatol, 2018, 38(11): 1431-43. doi: 10.1038/s41372-018-0209-5 [18] Xu MY, Wang YF, Wei PJ, et al. Hypoxic preconditioning improves long-term functional outcomes after neonatal hypoxia- ischemic injury by restoring white matter integrity and brain development[J]. CNS Neurosci Ther, 2019, 25(6): 734-47. doi: 10.1111/cns.13102 [19] 刘波. CT及MRI检查早期新生儿缺氧缺血性脑病的差异[J]. 中国继续医学教育, 2020, 12(9): 91-3. doi: 10.3969/j.issn.1674-9308.2020.09.038 [20] Feng X, Li HX, Wang Q, et al. Diffusion tensor imaging assesses white matter injury in neonates with hypoxic-ischemic encephalopathy[J]. Neural Regen Res, 2017, 12(4): 603. doi: 10.4103/1673-5374.205102 [21] Chavali M, Ulloa-Navas MJ, Pérez-Borredá P, et al. Wnt-dependent oligodendroglial- endothelial interactions regulate white matter vascularization and attenuate injury[J]. Neuron, 2020, 108(6): 1130-45. e5. doi: 10.1016/j.neuron.2020.09.033 [22] Lee JK, Santos PT, Chen MW, et al. Combining hypothermia and oleuropein subacutely protects subcortical white matter in a swine model of neonatal hypoxic-ischemic encephalopathy[J]. J Neuropathol Exp Neurol, 2021, 80(2): 182-98. doi: 10.1093/jnen/nlaa132 [23] Gerner GJ, Newman EI, Burton VJ, et al. Correlation between white matter injury identified by neonatal diffusion tensor imaging and neurodevelopmental outcomes following term neonatal asphyxia and therapeutic hypothermia: an exploratory pilot study[J]. J Child Neurol, 2019, 34(10): 556-66. doi: 10.1177/0883073819841717 [24] Montaldo P, Lally PJ, Oliveira V, et al. Therapeutic hypothermia initiated within 6 hours of birth is associated with reduced brain injury on MR biomarkers in mild hypoxic-ischaemic encephalopathy: a non-randomised cohort study[J]. Arch Dis Child Fetal Neonatal Ed, 2019, 104(5): F515-20. doi: 10.1136/archdischild-2018-316040 [25] Zeng Y, Wang H, Zhang L, et al. The optimal choices of animal models of white matter injury[J]. Rev Neurosci, 2019, 30(3): 245-59. doi: 10.1515/revneuro-2018-0044 [26] 王妍. 神经节苷脂与胞二磷胆碱联合还原型谷胱甘肽对轻和中度新生儿缺氧缺血性脑病患儿NBNA评分的影响分析[J]. 当代医学, 2020, 26(5): 32-4. doi: 10.3969/j.issn.1009-4393.2020.05.014 -







 下载:
下载: