Prognostic value of clinic-pathological characteristics and ultrasound features for assessment of triple negative breast cancer
-
摘要:
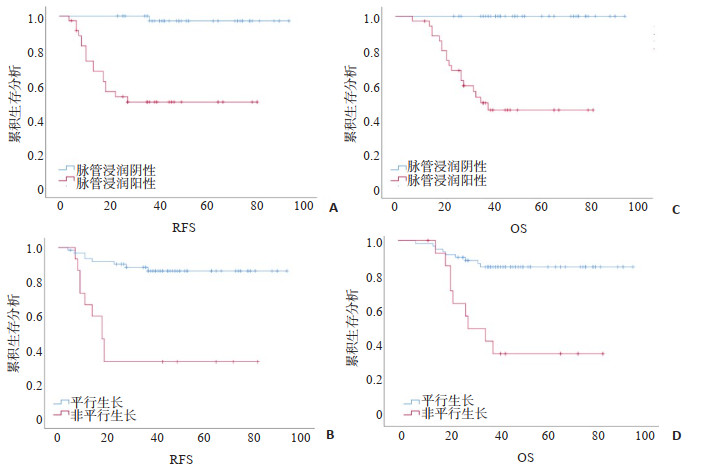
目的 探讨三阴性乳腺癌(TNBC)患者临床病理特征以及超声征象在患者预后评估中的价值。 方法 回顾性收集2012年1月~2017年12月间接受治疗的TNBC病患者的临床病理资料、超声特征、无复发生存期和总生存期;采用Cox比例风险回归及Kaplan-Meier生存分析法评价临床病理特征及超声征象对TNBC患者预后评估价值。 结果 共纳入78例TNBC患者,中位随访时间为40月,其中18例(23.1%)患者发生局部复发或转移,18例(23.1%)患者死亡。单因素分析显示绝经前期/围绝经期、病灶最大径>2 cm、腋窝淋巴结转移、Ki-67>30%、纤维腺体型乳腺、非平行生长及脉管侵犯与患者预后不良有关(P < 0.05),多因素分析显示仅非平行生长及脉管浸润为无复发生存期及总生存期独立危险因素(P < 0.05)。 结论 脉管侵犯及肿块非平行生长与TNBC患者预后不良相关。 Abstract:Objective To investigate the value of clinic-pathological characteristics and ultrasound features in predicting the prognosis of triple negative breast cancer (TNBC). Methods Patients with TNBC treated between January 2012 and December 2017 were retrospectively included. Patients' clinic-pathological, ultrasound features, recurrence-free survival, and overall survival were collected. Cox regression and Kaplan-Meier analysis were used to determine the prognostic factors of TNBC. Methods A total of 78 TNBC patients were included with a median follow-up of 40 months. Eighteen patients (23.1%) experienced recurrence and 18 patients (23.1%) occurred deaths. Univariate Cox analysis showed that pre/peri-menopausal patients, maximum tumor diameter >2 cm, positive axillary lymph node, Ki-67 expression >30%, fibroglandua breast, non-parallel oriented, and lymphatic vascular invasion are associated with poorer prognosis (P < 0.05). Multivariable Cox analysis found that only non-parallel oriented and lymphatic vascular invasion were two independent predictors of recurrence-free survival and overall survival (P < 0.05). Conclusions Peripheral lymphatic vascular invasion and tumor non-parallel growth are associated with poor prognosis in TNBC patients. -
Key words:
- triple negative breast cancer /
- prognosis /
- non-parallel oriented /
- lymphatic vascular invasion /
-
图 2 三阴性乳腺癌患者不同肿瘤生长方位的声像图表现
带箭头的线表示不同方向最大径线,“x”表示水平径线,“y”表示在前后径线。非平行生长定义为肿块水平径线与前后径线比值(x/y)< 1.0,否则平行生长。A:女性TNCB患者,80岁,肿块为非平行生长,术后5年随访无复发;B:女性TNBC患者,46岁,肿块为非平行生长,术后16月死于远处转移;C:女性TNCB患者,49岁,肿块为平行生长,术后5年随访无复发;D:女性TNCB患者,48岁,肿块为平行生长,术后5年随访无复发;E:病例A~D的病理图片(由上至下,HE×40).
Figure 2. Ultrasound imaging for different tumor growth orientations in triple negative breast cancer patients.
表 1 临床病理特征作为RFS及OS预后因素的单因素Cox回归分析
Table 1. Univariable analysis of clinic-pathological features as prognostic factors for RFS and OS events
变量 RFS OS HR(95%CI) P HR(95%CI) P 年龄(岁) >55 参考值 参考值 ≤55 1.553(0.583~4.140) 0.379 1.558(0.593~4.210) 0.361 月经情况 绝经后 参考值 参考值 绝经前/围绝经期 1.940(0.764~4.924) 0.163 2.637(1.018~6.833) 0.038 乳腺手术类型 保乳手术 参考值 参考值 乳腺根治术 0.764(0.176~3.326) 0.720 0.489(0.141~1.691) 0.247 腋窝淋巴结手术类型 前哨淋巴结活检 参考值 参考值 腋窝淋巴结清扫 0.713(0.187~0.712) 0.620 0.510(0.157~1.658) 0.309 病理类型 浸润性导管癌 参考值 参考值 其他类型 0.829(0.191~3.609) 0.803 0.388(0.052~2.914) 0.338 病理分级 Ⅰ~Ⅱ 参考值 参考值 Ⅲ 1.803(0.693~4.693) 0.227 1.87(0.712~4.918) 0.204 病灶最大直径(cm) ≤2 参考值 参考值 >2 1.355(1.072~1.712) 0.011 1.358(1.070~1.724) 0.012 腋窝淋巴结转移 阴性 参考值 参考值 阳性 2.889(1.137~7.340) 0.026 2.908(1.145~7.389) 0.018 Ki-67(%) ≤30 参考值 参考值 >30 2.760(1.033~7.372) 0.043 2.762(1.035~7.370) 0.042 脉管侵犯 阴性 参考值 参考值 阳性 5.732(2.215~9.514) 0.001 2.387(1.364~5.785) 0.019 RFS:无复发生存期;OS:总生存期;HR:危险比. 表 2 超声征象作为RFS及OS预后因素的单因素Cox回归分析
Table 2. Univariable analysis of ultrasound features as prognostic factors for RFS and OS events
变量 RFS OS HR(95%CI) P HR(95%CI) P 乳腺组织构成 脂肪型 参考值 参考值 纤维腺体型 0.889(1.116~7.475) 0.029 2.899(1.120~7.502) 0.028 边界 局限 参考值 参考值 模糊 1.758(0.475~6.502) 0.398 1.455(0.400~5.292) 0.569 浅分叶 0.749(0.203~2.766) 0.664 0.676(0.186~2.457) 0.552 成角 0.632(0.137~2.927) 0.557 0.281 (0.036~2.196) 0.226 短毛刺 1.328(0.168~9.488) 0.788 1128(0.144~8.817) 0.909 形态 规则 参考值 参考值 不规则 0.731(0.405~1.318) 0.294 0.931(0.481~1.805) 0.833 生长方向 平行牛长 参考值 参考值 非平行牛长 7.369(2.878~18.869) 0.001 5.337(2.111~13.492) <0.001 病灶后方回声 无改变 参考值 参考值 回声增强 0.431(0.131~1.420) 0.167 0.407(0.123~1348) 0.141 声影 0.305(0.097~0.966) 0.063 0.295(0.093~0.938) 0.069 腺体/导管结构 规则 参考值 参考值 扭曲 5.158(1.481~17.962) 0.060 5.425(1.549~19.002) 0.075 皮肤增厚水肿 阴性 参考值 参考值 阳性 1.061(0.378~2.976) 0.911 1.399(0.524~3.730) 0.499 血流分级 无 参考值 参考值 少量 1.376(0.305~6.213) 0.580 1.398(0.42~6.033) 0.673 中量 1.739(0.604~5.008) 0.678 1.734(0.604~5.108) 0.666 大量 1.431 (0.531~2.420) 0.305 1.586(0.551~4.564) 0.393 表 3 临床病理特征及超声征象作为RFS及OS预后因素的多因素Cox回归分析
Table 3. Multivariable analysis of clinic-pathological and ultrasound features as prognostic factors for RFS and OS events
变量 RFS OS HR(95%CI) P HR(95%CI) P 脉管侵犯 阴性 参考值 参考值 阳性 4.742(1.215~18.514) 0.025 2.658(1.257~5.785) 0.025 生长方向 平行生长 参考值 参考值 非平行生长 6.794(2.252~20.496) 0.001 3.753(1.352~10.42) 0.011 病灶最大直径(cm) ≤2 参考值 参考值 >2 1.793(0.475~6.761) 0.389 1.443(0.436~4.773) 0.548 Ki-67(%) ≤30 参考值 参考值 >30 1.720(0.605~4.889) 0.043 1.346(0.491~3.691) 0.563 乳腺组织构成 脂肪型 参考值 参考值 纤维腺体型 1.817(0.648~5.096) 0.256 1.368(0.686~4.847) 0.627 月经情况 绝经后 参考值 绝经前/围绝经期 2.632(1.617-4.223) 0.191 -
[1] Perou CM, Sørlie T, Eisen MB, et al. Molecular portraits of human breast tumours[J]. Nature, 2000, 406(6797): 747-52. doi: 10.1038/35021093 [2] Bartsch R, Ziebermayr R, Zielinski CC, et al. Triple-negative breast cancer[J]. Wiener Medizinische Wochenschrift, 2010, 160(7/8): 174-81. [3] Dent R, Trudeau M, Pritchard KI, et al. Triple-negative breast cancer: clinical features and patterns of recurrence[J]. Clin Cancer Res, 2007, 13(15 pt 1): 4429-34. http://pdfs.semanticscholar.org/623d/56600e2bac528db05270048e03c7a6107aae.pdf [4] Penault-Llorca F, Viale G. Pathological and molecular diagnosis of triple-negative breast cancer: a clinical perspective[J]. Ann Oncol, 2012, 23(Suppl 6): vi19-vi22. http://academic.oup.com/annonc/article/23/suppl_6/vi19/173229 [5] Yang Q, Liu HY, Liu D, et al. Ultrasonographic features of triplenegative breast cancer: a comparison with other breast cancer subtypes[J]. Asian Pac J Cancer Prev, 2015, 16(8): 3229-32. doi: 10.7314/APJCP.2015.16.8.3229 [6] Mema E, Schnabel F, Chun J, et al. The relationship of breast density in mammography and magnetic resonance imaging in women with triple negative breast cancer[J]. Eur J Radiol, 2020, 124: 108813. doi: 10.1016/j.ejrad.2020.108813 [7] Elsawaf Z, Sinn HP, Rom J, et al. Biological subtypes of triplenegative breast cancer are associated with distinct morphological changes and clinical behaviour[J]. Breast, 2013, 22(5): 986-92. doi: 10.1016/j.breast.2013.05.012 [8] Lawson BT, Vinnicombe S, Whelehan P, et al. Associations between the ultrasound features of invasive breast cancer and breast cancer specific survival[J]. Clin Radiol, 2020, 75(11): 879. http://www.sciencedirect.com/science/article/pii/S0009926020302816 [9] Rakha EA, El-Sayed ME, Green AR, et al. Prognostic markers in triple-negative breast cancer[J]. Cancer, 2007, 109(1): 25-32. doi: 10.1002/cncr.22381 [10] Rakha EA, Martin S, Lee AH, et al. The prognostic significance of lymphovascular invasion in invasive breast carcinoma[J]. Cancer, 2012, 118(15): 3670-80. doi: 10.1002/cncr.26711 [11] Song YJ, Shin SH, Cho JS, et al. The role of lymphovascular invasion as a prognostic factor in patients with lymph node-positive operable invasive breast cancer[J]. J Breast Cancer, 2011, 14(3): 198-203. doi: 10.4048/jbc.2011.14.3.198 [12] Fujii T, Yajima R, Hirakata T, et al. Impact of the prognostic value of vascular invasion, but not lymphatic invasion, of the primary tumor in patients with breast cancer[J]. Anticancer Res, 2014, 34 (3): 1255-9. http://www.ncbi.nlm.nih.gov/pubmed/24596369 [13] Rella R, Bufi E, Belli P, et al. Background parenchymal enhancement in breast magnetic resonance imaging: a review of current evidences and future trends[J]. Diagn Interv Imaging, 2018, 99(12): 815-26. doi: 10.1016/j.diii.2018.08.011 [14] Aleskandarany MA, Sonbul SN, Mukherjee A, et al. Molecular mechanisms underlying lymphovascular invasion in invasive breast cancer[J]. Pathobiology, 2015, 82(3/4): 113-23. http://www.onacademic.com/detail/journal_1000038571951410_ba9c.html [15] Paulinelli RR, Freitas-Junior R, de Lucena CÊ, et al. Sonobreast: predicting individualized probabilities of malignancy in solid breast masses with echographic expression[J]. Breast J, 2011, 17(2): 152-9. doi: 10.1111/j.1524-4741.2010.01046.x [16] Kim SH, Seo BK, Lee J, et al. Correlation of ultrasound findings with histology, tumor grade, and biological markers in breast cancer [J]. Acta Oncol, 2008, 47(8): 1531-8. doi: 10.1080/02841860801971413 [17] Kim SY, Kwak JY, Kim EK, et al. Association of preoperative US features and recurrence in patients with classic papillary thyroid carcinoma[J]. Radiology, 2015, 277(2): 574-83. doi: 10.1148/radiol.2015142470 [18] Keam B, Im SA, Lee KH, et al. Ki-67 can be used for further classification of triple negative breast cancer into two subtypes with different response and prognosis[J]. Breast Cancer Res, 2011, 13 (2): R22. doi: 10.1186/bcr2834 [19] Urru SAM, Gallus S, Bosetti C, et al. Clinical and pathological factors influencing survival in a large cohort of triple-negative breast cancer patients[J]. BMC Cancer, 2018, 18(1): 56. doi: 10.1186/s12885-017-3969-y [20] Pilewskie M, King TA. Age and molecular subtypes: Impact on surgical decisions[J]. J Surg Oncol, 2014, 110(1): 8-14. doi: 10.1002/jso.23604 [21] Kim MY, Choi N, Yang JH, et al. Background parenchymal enhancement on breast MRI and mammographic breast density: correlation with tumour characteristics[J]. Clin Radiol, 2015, 70(7): 706-10. doi: 10.1016/j.crad.2015.02.017 [22] Dontchos BN, Rahbar H, Partridge SC, et al. Are qualitative assessments of background parenchymal enhancement, amount of fibroglandular tissue on MR images, and mammographic density associated with breast cancer risk?[J]. Radiology, 2015, 276(2): 371-80. doi: 10.1148/radiol.2015142304 [23] Tamimi RM, Hankinson SE, Chen WY, et al. Combined estrogen and testosterone use and risk of breast cancer in postmenopausal women[J]. Arch Intern Med, 2006, 166(14): 1483-9. doi: 10.1001/archinte.166.14.1483 -







 下载:
下载: