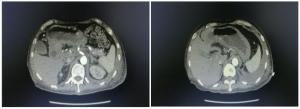
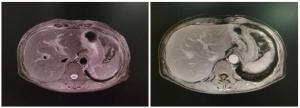
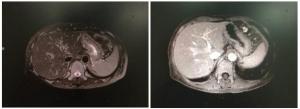
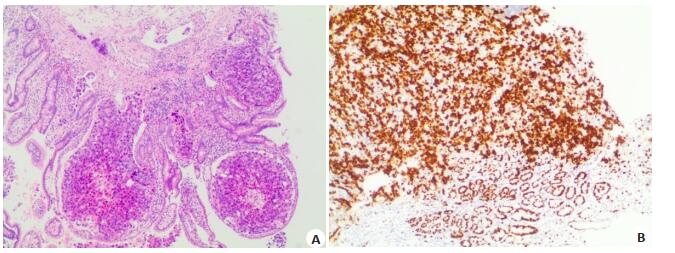
| [1] |
Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394-424. doi: 10.3322/caac.21492
|
| [2] |
孙可欣, 郑荣寿, 张思维, 等. 2015年中国分地区恶性肿瘤发病和死亡分析[J].中国肿瘤, 2019, 28(1): 1-11. http://d.old.wanfangdata.com.cn/Periodical/zgzl201901001
|
| [3] |
Koizumi W, Narahara H, Hara T, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase Ⅲ trial [J]. Lancet Oncol, 2008, 9(3): 215-21. https://www.ncbi.nlm.nih.gov/pubmed/18282805?format=text
|
| [4] |
Hironaka S. Anti-angiogenic therapies for gastric cancer[J]. Asia Pac J Clin Oncol, 2019, 15(4): 208-17. doi: 10.1111/ajco.13174
|
| [5] |
Wagner AD, Unverzagt S, Grothe W, et al. Chemotherapy for advanced gastric cancer[J]. Cochrane Database Syst Rev, 2010, 32 (3): CD004064-72. http://d.old.wanfangdata.com.cn/Periodical/zgazyj201804003
|
| [6] |
Wu WK, Cho CH, Lee CW, et al. Dysregulation of cellular signaling in gastric cancer [J]. Cancer Lett, 2010, 295(2): 144-53. https://pubmed.ncbi.nlm.nih.gov/20488613/
|
| [7] |
Abdel-Rahman O. Targeting vascular endothelial growth factor (VEGF) pathway in gastric cancer: preclinical and clinical aspects [J]. Crit Rev Oncol Hematol, 2015, 93(1):18-27. https://www.sciencedirect.com/science/article/abs/pii/S1040842814001000
|
| [8] |
van Cutsem E, Rivera F, Berry S, et al. Safety and efficacy of first-line bevacizumab with FOLFOX, XELOX, FOLFIRI and fluoropyrimidines in metastatic colorectal cancer: the BEAT study [J]. Ann Oncol, 2009, 20(11):1842-7. https://www.sciencedirect.com/science/article/pii/S092375341940001X
|
| [9] |
秦叔逵, 邓艳红, 毕锋, 等.贝伐珠单抗联合含氟尿嘧啶类化疗方案治疗晚期转移性结直肠癌的前瞻性、非干预性、全国多中心临床研究(REACT) [J].临床肿瘤学杂志, 2016, 21(10): 865-73. http://d.old.wanfangdata.com.cn/Periodical/lczlxzz201610001
|
| [10] |
von Minckwitz G, Puglisi F, Cortes J, et al. Bevacizumab plus chemotherapy versus chemotherapy alone as second-line treatment for patients with HER2-negative locally recurrent or metastatic breast cancer after first-line treatment with bevacizumab plus chemotherapy (TANIA): an open-label, randomised phase 3 trial [J]. Lancet Oncol, 2014, 15(11): 1269-78. https://www.sciencedirect.com/science/article/abs/pii/S1470204514704395
|
| [11] |
Ohtsu A, Shah MA, van Cutsem E, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase Ⅲ study[J]. J Clin Oncol, 2011, 29(30): 3968-76. https://www.ncbi.nlm.nih.gov/pubmed/?term=Bevacizumab+in+combination+with+chemotherapy+as+first-line+therapy+in+advanced+gastric+cancer%3A+a+randomized%2C+double-blind%2C+placebo-controlled+phase+III+study
|
| [12] |
Wilke H, Muro K, van Cutsem E, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial[J]. Lancet Oncol, 2014, 15(11): 1224-35. https://www.thelancet.com/journals/lanonc/article/PIIS1470-2045(14)70420-6/fulltext
|
| [13] |
Fuchs CS, Tomasek J, Yong CJ, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial[J]. Lancet, 2014, 383(9911): 31-9. https://www.thelancet.com/pdfs/journals/lancet/PIIS0140-6736(13)61719-5.pdf
|
| [14] |
Roviello G, Ravelli A, Fiaschi AI, et al. Apatinib for the treatment of gastric cancer[J]. Expert Rev Gastroenterol Hepatol, 2016, 10 (8): 887-92. http://d.old.wanfangdata.com.cn/Periodical/lcywzlzz201908015
|
| [15] |
秦叔逵, 李进.阿帕替尼治疗胃癌的临床应用专家共识[J].临床肿瘤学杂志, 2015, (9): 841-7. http://d.old.wanfangdata.com.cn/Periodical/lczlxzz201509014
|
| [16] |
Qin SK. phase Ⅲ study of apatinib in advanced gastric cancer: a randomized, double-blind, placebo-controlled trial [J]. J Clin Oncol, 2014, 32: 513-9. https://www.researchgate.net/publication/311729990_O-0029_PHASE_III_STUDY_OF_APATINIB_IN_ADVANCED_GASTRIC_CANCER_A_RANDOMIZED_DOUBLE-BLIND_PLACEBO-CONTROLLED_TRIAL
|
| [17] |
Li S, Zhong YJ, Teng Y, Zhu LJ. Clinical efficacy and prognosis of apatinib combined with chemotherapy in the treatment of advanced gastric cancer [J]. Chin J Clin Oncol Rehab, 2018, 25(1): 44-6. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zgzllcykf201801013
|
| [18] |
钱祥云, 戈伟.阿帕替尼联合化疗治疗晚期胃癌临床分析[J].中华实用诊断与治疗杂志, 2017, 31(12): 1222-4. http://d.old.wanfangdata.com.cn/Periodical/syzdyzlzz201712023
|
| [19] |
朱华云, 孙小峰, 周青, 等.阿帕替尼联合化疗治疗晚期胃癌的临床研究[J].中国肿瘤外科杂志, 2016, 8(6): 394-6. http://d.old.wanfangdata.com.cn/Periodical/zgyxwz-wkxfc201606013
|
| [20] |
Xu ZY, Hu C, Chen SQ, et al. Apatinib enhances chemosensitivity of gastric cancer to paclitaxel and 5-fluorouracil[J]. Cancer Manag Res, 2019, 11: 4905-15. https://www.researchgate.net/publication/333456784_Apatinib_enhances_chemosensitivity_of_gastric_cancer_to_paclitaxel_and_5-fluorouracil
|
| [21] |
韩忠诚, 马丽丽, 崔智文, 等. SOX方案联合阿帕替尼一线治疗晚期胃癌的临床研究[J].中国癌症防治杂志, 2019, 11(1): 62-7. http://d.old.wanfangdata.com.cn/Periodical/zgyxwz-zlxfc201901011
|
| [22] |
Galdy S, Cella CA, Spada F, et al. Systemic therapy beyond firstline in advanced gastric cancer: an overview of the main randomized clinical trials[J]. Crit Rev Oncol Hematol, 2016, 99: 1- 12. https://www.sciencedirect.com/science/article/abs/pii/S1040842815300445
|
| [23] |
Chan MM, Sjoquist KM, Zalcberg JR. Clinical utility of ramucirumab in advanced gastric cancer [J]. Biologics, 2015, 9(2): 93-105. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=Doaj000004008776
|
| [24] |
Parry RV, Chemnitz JM, Frauwirth KA, et al. CTLA-4 and PD-1 receptors inhibit T-cell activation by distinct mechanisms[J]. Mol Cell Biol, 2005, 25(21): 9543-53. https://core.ac.uk/display/3786575
|
| [25] |
Kang YK, Boku N, Satoh T, et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial[J]. Lancet, 2017, 390(10111): 2461-71. https://www.sciencedirect.com/science/article/pii/S0140673617318275
|
| [26] |
Fuchs CS, DoiT, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE- 059 trial [J]. J Am Med Assco Oncol, 2018, 4(5): e180013-8. https://reference.medscape.com/medline/abstract/29543932
|
| [27] |
Kim ST, Cristescu R, Bass AJ, et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer [J]. Nat Med, 2018, 24(9): 1449-58. https://pubmed.ncbi.nlm.nih.gov/30013197/
|
| [28] |
Tabernero J, Bang YJ, Fuchs CS, et al. KEYNOTE-062: phase Ⅲ study of pembrolizumab (MK-3475) alone or in combination with chemotherapy versus chemotherapy alone as first-line therapy for advanced gastric or gastroesophageal junction (GEJ) adenocarcinoma [J]. J Clin Oncol, 2016, 34(4): TPS185-92. http://www.socolar.com/Article/Index?aid=100023665876&jid=100000000205
|
| [29] |
Kang YK, Kato K, Chung HC, et al. Interim safety and clinical activity of nivolumab (Nivo) in combination with S-1/capecitabine plus oxaliplatin in patients (pts) with previously untreated unresectable advanced or recurrent gastric/gastroesophageal junction (G/GEJ) cancer: part 1 study of ATTRACTION-04 (ONO- 4538-37)[ J]. Ann Oncol, 2017, 28: v228-9.
|






 下载:
下载: