Short-term effect of Sacubitril/Valsartan in the heart failure patients with reduced of ejection fraction on echocardiography, biochemical and clinical
-
摘要:
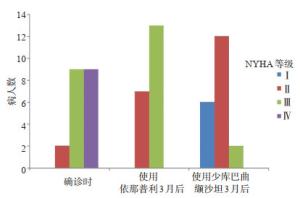
目的从超声、生化、临床方面探讨沙库巴曲缬沙坦对射血分数降低心力衰竭(HFrEF)患者心功能改善的短期疗效。 方法选择2018年6月~2019年9月在我院首诊为HFrEF的患者为研究对象,共20例,年龄51.4±9.923岁,规律服用血管紧张素转化酶抑制剂依那普利3月后换用沙库巴曲缬沙坦继续治疗3月。自身前后对比,分别比较患者确诊时、依那普利治疗3月时和换用沙库巴曲缬沙坦治疗3月后N末端脑钠肽前体、经胸超声心动图测定左室射血分数、6 min步行实验和纽约心功能分级。 结果沙库巴曲缬沙坦治疗3月后患者左室射血分数的增幅大于依那普利治疗3月后对比于确诊时的增幅(P < 0.05),沙库巴曲缬沙坦治疗3月后患者N末端脑钠肽前体的降幅大于依那普利治疗3月后对比于确诊时的降幅(P < 0.05);同时患者使用2类药物后6 min步行实验、纽约心功能分级均有改善,差异有统计学意义(P < 0.05),且沙库巴曲缬沙坦改善程度大于依那普利。结论沙库巴曲缬沙坦对射血分数降低心力衰竭患者心功能改善方面的短期疗效(3月)优于依那普利。 -
关键词:
- 沙库巴曲缬沙坦 /
- 射血分数降低心力衰竭 /
- 短期疗效 /
- 超声心动图
Abstract:ObjectiveTo investigate the short- term functionality efficacy of Sacubitril/Valsartan in heart failure patients with reduced ejection fraction (HFrEF) on echocardiography, biochemical and clinical. MethodsTwenty HFrEF patients who were first diagnosed in our hospital from June 2018 to September 2019 were selected, with age of 51.4±9.923 years old. Patients were regularly treated with ACEI (angiotensin-converting enzyme inhibitor) Enalapril for 3 months and then switched to Sacubitril/ Valsartan for 3 months. The effect of the two medication between the baseline and 3 months after Enalapril treatment, and 3 months after switching to Sacubitril/Valsartan for N-terminal brain natriuretic peptide, left ventricular ejection fraction measured by transthoracic echocardiography, and 6 min walking test and New York Heart Association(NYHA)scale assessment were compared. ResultsAfter 3 months of treatment with Sacubitril/Valsartan, the increasing amplitude of left ventricular ejection fraction in patients was greater than the before (P < 0.05). The decreasing amplitude of N-terminal brain natriuretic peptide after 3 months of treatment with Sacubitril/Valsartan was greater than that after 3 months of treatment with Enalapril (P < 0.05). The difference in the improvement of distance walked on the 6 min walk test and NYHA scale significant (P < 0.05). ConclusionThe short-term (3 months) functionality efficacy of Sacubitril/Valsartan in HFrEF patients is better than that of Enalapril. -
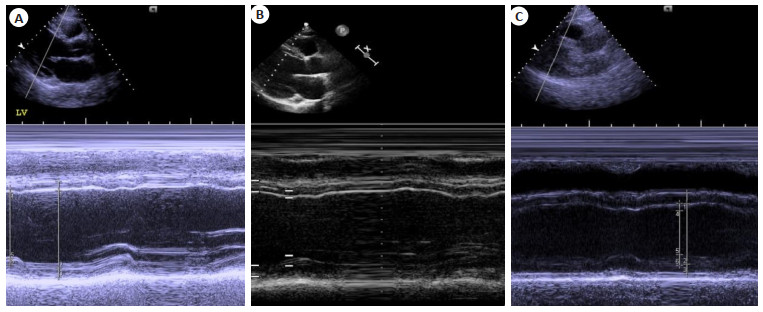
图 1 两种药物治疗3月后经胸超声心动图监测患者LVEF变化
A:患者确诊HFrEF时测量的LVEF; B:使用依那普利3月后测量的LVEF; C:使用沙库巴曲缬沙坦3月后测量的LVEF; LVEF: Left ventricular ejection fraction
Figure 1. Comparison of LVEF of the patients monitored by transthoracic echocardiography in baseline, 3 months after Enalapril treatment and 3 months after switching to Sacubitril/Valsartan
表 1 两种药物治疗3月后LVEF增幅比较(n=20, Mean±SD, %)
Table 1. Comparison of the increasing amplitude of the LVEF between the two drugs 3 months later
药物 LVEF水平 用药前 用药3月后 增幅 依那普利 34.35±6.36 37.25±3.84 2.90±4.89 沙库巴曲缬沙坦 37.25±3.84 43.30±2.81 6.05±5.26 t - - -2.103 P - - 0.049 LVEF: Left ventricular ejective fraction. 表 2 两种药物治疗3月后NT-proBNP降幅比较(n=20, Mean±SD, pg/mL)
Table 2. Comparison of the decreasing amplitude of the NT-proBNP between the two drugs 3 months later
药物 NT-proBNP水平 用药前 用药3月后 降幅 依那普利 12 727.15±10 984.40 8893.15±6651.51 3834.00±5123.31 沙库巴曲缬沙坦 8893.15±6651.51 3291.55±2638.93 5601.60±6054.00 t - - 2.147 P - - 0.045 NT-proBNP: N terminal pro brain natriuretic peptide. 表 3 两种药物治疗3月后6MWT距离比较(n=20, Mean±SD, m)
Table 3. Comparison of the increasing amplitude of the improvement of distance between the two drugs 3 months later
药物 6MWT距离(m) 用药前 用药3月后 增幅 依那普利 217.25±99.49 301.60±48.48 84.35±78.68 沙库巴曲缬沙坦 301.60±48.48 477.75±104.84 176.15±101.50 t - - -4.295 P - - 0.000 6MWT: 6-minute walk test. 表 4 两种药物治疗3月后NYHA分级改善有效率比较[n=20, n(%)]
Table 4. Comparison of the effective of NYHA scale improvement between the two drugs 3 months later
药物 NYHA分级改善 有效 无效 有效率 依那普利 13 (65) 7 (35) 13 (65) 沙库巴曲缬沙坦 14 (70) 6 (30) 14 (70) χ2 - - 4.615 P - - 0.032 -
[1] Daniela T, Marianna A, Carlo M, et al. Highlights in heart failure[J]. Esc Heart Fail, 2019, 6(6): 1105-27. doi: 10.1002/ehf2.12555 [2] Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure[J]. Eur J Heart Fail, 2016, 18(8): 891-975. doi: 10.1002/ejhf.592 [3] McMurray JJV, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure[J]. N Engl J Med, 2014, 371: 993-1004. doi: 10.1056/NEJMoa1409077 [4] Beltrán P, Palau P, Domínguez E, et al. Sacubitril/valsartan and short-term changes in the 6-minute walk test: a pilot study[J]. Int J Cardiol, 2018, 252: 136-9. doi: 10.1016/j.ijcard.2017.10.074 [5] Chandra A, Lewis EF, Claggett BL, et al. Effects of sacubitril/ valsartan on physical and social activity limitations in patients with heart failure[J]. J Am Med Assco Cardiol, 2018, 3(6): 498-505. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0555b1fa6b0ccedb8ae772fa65037939 [6] Januzzi JL Jr, Prescott MF, Butler J, et al. Association of change in N-terminal pro-B-type natriuretic peptide following initiation of sacubitril-valsartan treatment with cardiac structure and function in patients with heart failure with reduced ejection fraction[J]. J Am Med Assco, 2019, 322(11): 1085-9. doi: 10.1001/jama.2019.12821 [7] Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: The task force for the diagnosis and treatment of acute and chronic heart failure of the european society of cardiology (ESC) developed with the special contribution of the heart failure association (HFA) of the ESC[J]. Eur Heart J, 2016, 27(27): 128-32. https://www.ncbi.nlm.nih.gov/pubmed/27207191 [8] Sgorbini L, Rossetti A, Galati A. Sacubitril/valsartan: effect on walking test and physical capability[J]. Cardiol, 2017, 138(1): 17-20. doi: 10.1159/000484879 [9] Fraile RR, Malafarina V, Lopez GT. Sacubitril-valsartan in heart failure and multimorbidity patients[J]. Esc Heart Fail, 2018, 5(5): 956-9. doi: 10.1002/ehf2.12338 [10] Mazzetti S, Scifo C, Abete R, et al. Short-term echocardiographic evaluation by global longitudinal strain in patients with heart failure treated with sacubitril/valsartan[J]. ESC Heart Fail, 2020. https://doi.org/10.1002/ehf2.12656. [11] Nandal S, Chow CL, Hannah V, et al. Tolerability and efficacy of Sacubitril/ Valsartan in clinical practice[J/OL]. Intern Med J, 2020. https://doi.org/10.1111/imj.14749 [12] Kemp CD, Conte JV. The pathophysiology of heart failure[J]. Cardiovasc Pathol, 2012, 21(5): 365-71. doi: 10.1016/j.carpath.2011.11.007 [13] Langenickel TH, Dole WP. Angiotensin receptor-neprilysin inhi-bition with LCZ696: a novel approach for the treatment of heart failure[J]. Drug Discov Today: Ther Strateg, 2012, 9(4): e131-9. doi: 10.1016/j.ddstr.2013.11.002 [14] Feng LL, Karpinski PH, Sutton P, et al. LCZ696: a dual-acting sodium supramolecular complex[J]. Tetrahedron Lett, 2012, 53(3): 275-6. doi: 10.1016/j.tetlet.2011.11.029 [15] 中华医学会心血管病学分会, 中华心血管病杂志编辑委员会.慢性心力衰竭诊断治疗指南[J].中华心血管病杂志, 2007, 35(12): 1076-95. doi: 10.3760/j.issn:0253-3758.2007.12.002 [16] Martens P, Beliën H, Dupont M, et al. The reverse remodeling response to sacubitril/valsartan therapy in heart failure with reduced ejection fraction[J]. Cardiovasc Ther, 2018, 36(4): e12435-42. doi: 10.1111/1755-5922.12435 [17] Potter LR, Abbey-Hosch S, Dickey DM. Natriuretic peptides, their receptors, and cyclic guanosine monophosphate-dependent signaling functions[J]. Endocr Rev, 2006, 27(1): 47-72. doi: 10.1210/er.2005-0014 [18] Gardner DG, Chen SC, Glenn DJ, et al. Molecular biology of the natriuretic peptide system: implications for physiology and hypertension[J]. Hypertension, 2007, 49(3): 419-26. http://cn.bing.com/academic/profile?id=6ce6769e328cb3bb8d7ea874d7ffa8eb&encoded=0&v=paper_preview&mkt=zh-cn [19] Iborra-Egea O, Gálvez-Montón C, Roura S, et al. Mechanisms of action of sacubitril/valsartan on cardiac remodeling: a systems biology approach[J]. Npj Syst Biol Appl, 2017, 3: 12-8. doi: 10.1038/s41540-017-0013-4 [20] Romano G, Vitale G, Ajello L, et al. The effects of sacubitril/valsartan on clinical, biochemical and echocardiographic parameters in patients with heart failure with reduced ejection fraction: the "hemodynamic recovery"[J]. J Clin Med, 2019, 8(12): 2165-9. doi: 10.3390/jcm8122165 [21] Bayes-Genis A, Barallat J, Richards AM. A test in context: Neprilysin: Function, inhibition, and biomarker[J]. J Am Coll Cardiol, 2016, 68(6): 639-53. doi: 10.1016/j.jacc.2016.04.060 [22] Campbell DJ. Long-term neprilysin inhibition [mdash] implications for ARNIs[J]. Nat Rev Cardiol, 2017, 14(3): 171-86. doi: 10.1038/nrcardio.2016.200 [23] Fraile RR, Malafarina V, Lopez GT. Sacubitril-valsartan in heart failure and multimorbidity patients[J]. Esc Heart Fail, 2018, 5(5): 956-9. doi: 10.1002/ehf2.12338 [24] Hickey MS, Franke WD, Herbert WG, et al. Opioid antagonism, perceived exertion and tolerance to exercise-thermal stress[J]. Int J Sports Med, 1992, 13(4): 326-31. doi: 10.1055/s-2007-1021275 [25] Merenlender-Wagner A, Dikshtein Y, Yadid G. The β-endorphin role in stress-related psychiatric disorders[J]. Curr Drug Targets, 2009, 10 (11): 1096-108. doi: 10.2174/138945009789735147 [26] Sha JC, Meng CD, Li L, et al. Correlation of serumβ- endorphin and the quality of life in allergic rhinitis[J]. Dis Markers, 2016, 2016: 1-5. https://www.hindawi.com/journals/dm/2016/2025418/ [27] Maslov MY, Foianini S, Orlov MV, et al. A novel paradigm for sacubitril/valsartan: beta- endorphin elevation as a contributor to exercise tolerance improvement in rats with preexisting heart failure induced by pressure overload[J]. J Cardiac Fail, 2018, 24(11): 773-82. doi: 10.1016/j.cardfail.2018.10.006 -






 下载:
下载: