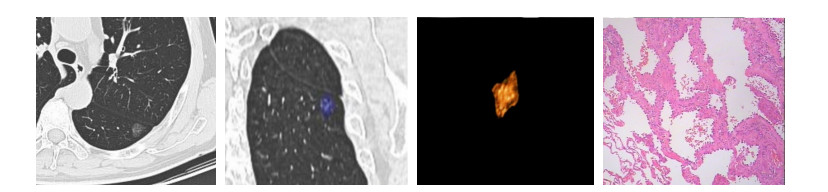
High resolution computed tomography findings, clinicopathological features and genetic characteristics of pure ground-glass opacity of the lung
-
摘要:
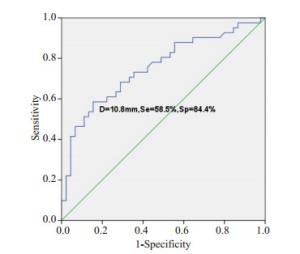
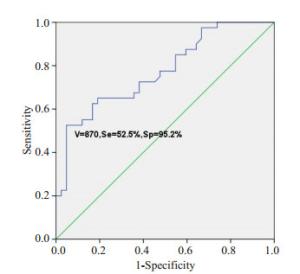
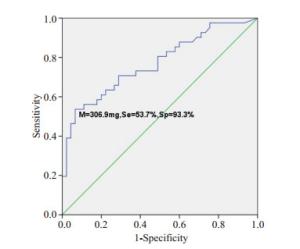
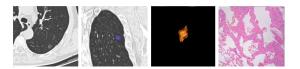
目的探讨肺纯磨玻璃结节的高分辨率CT表现与临床病理学及基因特征。 方法回顾性纳入2014年4月~2019年4月在解放军总医院海南医院胸外科行手术切除的86例肺纯磨玻璃结节(pGGOs)患者,其中男性30例,女性56例,年龄21~78岁。分析其围手术期资料,根据术后5年生存率的不同将浸润性腺癌(IA)作为IA组,将微浸润性腺癌、原位腺癌及不典型腺瘤样增生设为对照组。其中IA组患者41例,对照组45例(微浸润性腺癌患者25例,原位腺癌患者14例,不典型腺瘤样增生患者6例)。比较IA组和对照组的高分辨率CT影像特点,组织病理学特征及基因突变情况。 结果两组患者在一般资料方面差异无统计学意义(P>0.05),在手术方式上的差异有统计学意义(P=0.001),IA组患者更多接受肺叶切除术,对照组患者更多行亚肺叶切除术。两组患者在pGGOs的直径、体积及质量差异有统计学意义(P < 0.001),在平均CT值和转化密度上差异无统计学意义(P>0.05)。ROC曲线分析,以10.8 mm为临界值鉴别肺IA组和对照组,敏感度为58.5%,特异度为84.4%,曲线下面积(AUC)为0.753。体积的临界值为870 mm3,AUC为0.773,诊断敏感度52.5%,特异性95.2%。质量的临界值为306.9 mg,AUC为0.769,诊断敏感度53.7%,特异性93.3%。反映边缘特征方面,在分叶和毛刺上有统计学差异,而在pGGOs的形状、胸膜牵拉征和血管集束征等方面差异并无统计学意义(P>0.05)。基因突变状态方面,对照组8例检测患者中有2例EGFR基因突变,IA组中20例检测患者中有9例EGFR突变,两组基因突变差异无统计学意义(P>0.05)。 结论肺纯磨玻璃结节的病理表现多样,结合高分辨率CT影像特征及图像分析有助于对其定性诊断并指导后续随访策略及制定手术方案。对于直径大于10.8 mm、体积大于870 mm3及质量大于306.9 mg的纯磨玻璃结节,伴有分叶征、毛刺征等恶性影像学特征,应积极微创手术,手术方式根据影像学及患者意愿,主要以亚肺叶切除联合淋巴结活检术,可获得令人满意的预后效果。 Abstract:ObjectiveTo analyze the high resolution computed tomography findings, clinicopathological features and genetic characteristics of pure ground-glass opacities of the lung. MethodsThe retrospective study included 86 patients with pGGOs who underwent surgical resection in the thoracic surgery department of hainan hospital of PLA general hospital from April 2014 to April 2019, including 30 males and 56 females, with the age from 21 to 78 years old. The perioperative data were analyzed to distinguish HRCT imaging features, histopathological features and gene mutations in the invasive adenocarcinoma group (IA) and the control group (MIA, AIS and AAH) according to the different survival. ResultsA total of 86 patients with pGGOs were enrolled, including 41 in group IA and 45 in control group, including 25 in MIA, 14 in AIS and 6 in AAH. The difference of general data between the two groups was not significant(P>0.05). However, there was a significant difference in surgical procedure (P=0.001). Patients in the IA group were more likely to receive lobectomy, while those in the control group were more likely to receive sublobectomy. The differences of diameter, volume and mass of pGGOs between the two groups were significant (P < 0.001), but no significant differences in mean CT value and conversion density (P>0.05). According to ROC curve analysis, the lung IA group and the control group were identified with a critical value of 10.8 mm, with a sensitivity of 58.5%, specificity of 84.4% and AUC of 0.753. The critical values of volume and AUC were 870 mm3 and 0.773, respectively. The diagnostic sensitivity was 52.5% and the specificity was 95.2%. The critical value of mass was 306.9 mg, the AUC value was 0.769, the diagnostic sensitivity was 53.7% and the specificity was 93.3%. There were significant differences in lobules and burrs, but no significant differences in pGGOs shape, pleural traction and vascular cluster. In terms of gene mutation status, 2 of the 8 patients in the control group had EGFR gene mutation, and 9 of the 20 patients in the IA group had EGFR mutation. The difference of gene mutation between the two groups was not significant (P>0.05). ConclusionIn summary, the pathological manifestations of pure ground glass nodules in lungs are diverse. Combining HRCT image features and image analysis can help to make a qualitative diagnosis and guide subsequent follow-up strategies and formulate surgical plans. For pure ground glass nodules with a diameter greater than 10.8 mm, a volume greater than 870 mm3, and a mass greater than 306.9 mg, with malignant imaging features such as lobular signs and burr signs, minimally invasive surgery should be actively performed. The surgical method is based on imaging and patient wishes. Sublobar resection combined with lymph node biopsy can achieve satisfactory prognosis. -
表 1 IA组和对照组的一般临床资料
Table 1. Baseline and surgical characteristics in IA and control group
临床特征 IA组(n=41) 对照组(n=45) P 年龄(岁, Mean±SD) 53.32±11.94 48.75±10.27 0.227 性别(n) 0.445 男性 16 14 女性 25 31 吸烟史(n) 0.176 有 9 4 无 32 41 手术方式(n) 0.001 肺叶切除 23 10 肺段切除 4 5 楔形切除 14 30 淋巴结切除 未切除 2 10 清扫淋巴结站数(Mean±SD) 4.46±1.83 2.84±1.99 0.230 淋巴结转移(n) 0 0 1.000 表 2 IA组和对照组的HRCT影像特征(Mean±SD)
Table 2. HRCT findings in IA and control group
指标 IA组(n=41) 对照组(n=45) P 结节直径(mm) 12.86±5.24 8.67±3.05 0.000 CT值(HU) -543.25±133.18 -547.32±139.50 0.466 转化密度(mg/cm3) 456.75±133.18 452.86±139.86 0.460 体积(mm3) 1360.48±1463.99 401.31±396.37 0.000 质量(mg) 587.36±630.90 156.02±150.23 0.000 HRCT征象(n) 圆形/不规则形 40/1 45/0 0.295 分叶征 29 18 0.004 毛刺征 25 16 0.024 胸膜牵拉征 16 16 0.741 血管集束征 38 39 0.365 结节位置(n) 右上叶 9 12 右中叶 2 4 右下叶 9 9 左上叶 11 12 左下叶 10 8 HRCT: High Resolution CT. 表 3 IA组和对照组的基因突变结果
Table 3. Genetic mutation results in IA and control group
基因突变类型 IA组(n=20) 对照组(n=8) P EGFR突变 9 2 0.438 21外显子 6 1 - 19外显子 3 0 - 20外显子 0 1 - ALK/KRAS 0 0 - BRAF V600E/MEK 0 0 - MET/HER2 0 0 - ROS1/ERBB2 0 0 - -
[1] Godoy MCB, Naidich DP. Overview and strategic management of subsolid pulmonary nodules[J]. JThorac Imaging, 2012, 27(4): 240-8. doi: 10.1097/RTI.0b013e31825d515b [2] Katsumata S, Aokage K, Nakasone S, et al. Radiologic criteria in predicting pathologic less invasive lung cancer according to TNM 8th edition[J]. Clin Lung Cancer, 2019, 20(2): e163-70. [3] 刘士远, 范丽, 萧毅.加强肺内纯磨玻璃密度结节的影像学研究, 提升临床处理水平[J].中华放射学杂志, 2017, 51(7): 481-3. doi: 10.3760/cma.j.issn.1005-1201.2017.07.001 [4] Si MJ, Tao XF, Du GY, et al. Thin- section computed tomographyhistopathologic comparisons of pulmonary focal interstitial fibrosis, atypical adenomatous hyperplasia, adenocarcinoma in situ, and minimally invasive adenocarcinoma with pure ground-glass opacity [J]. Eur J Radiol, 2016, 85(10): 1708-15. doi: 10.1016/j.ejrad.2016.07.012 [5] Takashima S, Maruyama Y, Hasegawa M, et al. CT findings and progression of small peripheral lung neoplasms having a replacement growth pattern[J].AJRAm J Roentgenol, 2003, 180(3): 817-26. doi: 10.2214/ajr.180.3.1800817 [6] Travis WD, Brambilla E, Noguchi M, et al. International association for the study of lung cancer/American thoracic society/European respiratory society international multidisciplinary classification of lung adenocarcinoma[J]. J Thorac Oncol, 2011, 6(2): 244-85. [7] Yatabe Y, Borczuk AC, Powell CA. Do all lung adenocarcinomas follow a stepwise progression[J]? Lung Cancer, 2011, 74(1): 7-11. [8] 申磊磊, 林吉兴, 王柏霖, 等.肺原位腺癌和微浸润性腺癌的影像学表现与临床病理学、分子基因特征及预后[J].南方医科大学学报, 2019, 39(9): 1107-12. http://d.old.wanfangdata.com.cn/Periodical/dyjydxxb201909017 [9] 刘慧婷, 王颖, 冯磊, 等.亚实性肺结节CT三维质量分析:观察者内及观察者间差异[J].中国肺癌杂志, 2015, 18(5): 289-94. http://d.old.wanfangdata.com.cn/Periodical/zgfazz201505006 [10] Mull RT. Mass estimates by computed tomography: physical density from CT numbers[J]. Am J Roentgenol, 1984, 143(5): 1101-4. doi: 10.2214/ajr.143.5.1101 [11] Zhang LJ, Yankelevitz DF, Carter D, et al. Internal growth of nonsolid lung nodules: radiologic-pathologic correlation[J]. Radiology, 2012, 263(1): 279-86. doi: 10.1148/radiol.11101372 [12] Kim H, Park CM, Woo S, et al. Pure and part-solid pulmonary ground-glass nodules: measurement variability of volume and mass in nodules with a solid portion less than or equal to 5 mm[J]. Radiology, 2013, 269(2): 585-93. doi: 10.1148/radiol.13121849 [13] MacMahon H, Austin JH, Gamsu G, et al. Guidelines for management of small pulmonary nodules detected on CT scans: a statement from the fleischner society[J]. Radiology, 2005, 237(2): 395-400. doi: 10.1148/radiol.2372041887 [14] Lee SW, Leem CS, Kim TJ, et al. The long-term course of groundglass opacities detected on thin-section computed tomography[J]. Respir Med, 2013, 107(6): 904-10. doi: 10.1016/j.rmed.2013.02.014 [15] Gomez-Fernandez C, Mejias A, Walker G, et al. Immunohistochemical expression of estrogen receptor in adenocarcinomas of the lung: the antibody factor[J]. Appl Immunohistochem Mol Morphol, 2010, 18(2): 137-41. doi: 10.1097/PAI.0b013e3181bec23b [16] Lim E, Baldwin D, Beckles M, et al. Guidelines on the radical management of patients with lung cancer[J]. Thorax, 2010, 65 (Suppl 3): iii1-27. [17] Tsutani Y, Miyata Y, Nakayama H, et al. Oncologic outcomes of segmentectomy compared with lobectomy for clinical stage IA lung adenocarcinoma: Propensity score-matched analysis in a multicenter study[J]. J Thorac Cardiovasc Surg, 2013, 146(2): 358-64. doi: 10.1016/j.jtcvs.2013.02.008 [18] Sakurai H, Asamura H. Sublobar resection for early-stage lung cancer [J]. Transl Lung Cancer Res, 2014, 3(3): 164-72. [19] Jiang L, Yin WQ, Peng GL, et al. Prognosis and status of lymph node involvement in patients with adenocarcinoma in situ and minimally invasive adenocarcinoma-a systematic literature review and pooleddata analysis[J]. J Thorac Dis, 2015, 7(11): 2003-9. [20] Behera M, Owonikoko TK, Gal AA, et al. Lung adenocarcinoma staging using the 2011 IASLC/ATS/ERS classification: a pooled analysis of adenocarcinoma in situ and minimally invasive adenocarcinoma[J]. Clin Lung Cancer, 2016, 17(5): e57-64. doi: 10.1016/j.cllc.2016.03.009 [21] Singh S, Pinsky PF, Fineberg NS, et al. Evaluation of reader variability in the interpretation of follow-up CT scans at lung cancer screening[J]. Radiology, 2011, 259(1): 263-70. doi: 10.1148/radiol.10101254 [22] 姜格宁, 陈昶, 朱余明, 等.上海市肺科医院磨玻璃结节早期肺腺癌的诊疗共识(第一版[) J].中国肺癌杂志, 2018, 21(3): 147-59. [23] 范恒鑫, 樊树峰.肺纯磨玻璃结节CT定量分析研究进展[J].肿瘤学杂志, 2019, 25(3): 252-5. http://d.old.wanfangdata.com.cn/Periodical/zlxzz201903016 [24] Ko JP, Suh J, Ibidapo O, et al. Lung adenocarcinoma: correlation of quantitative CT findings with pathologic findings[J]. Radiology, 2016, 280(3): 931-9. doi: 10.1148/radiol.2016142975 [25] Son JY, Lee HY, Kim JH, et al. Quantitative CT analysis of pulmonary ground-glass opacity nodules for distinguishing invasive adenocarcinoma from non-invasive or minimally invasive adenocarcinoma: the added value of using iodine mapping[J]. Eur Radiol, 2016, 26(1): 43-54. doi: 10.1007/s00330-015-3816-y [26] Mao HX, Labh K, Han FS, et al. Diagnosis of the invasiveness of lung adenocarcinoma manifesting as ground glass opacities on highresolution computed tomography[J]. Thorac Cancer, 2016, 7(1): 129-35. doi: 10.1111/1759-7714.12269 [27] Lee HJ, Jin MG, Chang HL, et al. Nodular ground-glass opacities on thin-section CT: size change during follow-up and pathological results[J]. Korean J Radiology, 2007, 8(1): 22-31. doi: 10.3348/kjr.2007.8.1.22 [28] Heo EY, Lee KW, Jheon S, et al. Surgical resection of highly suspicious pulmonary nodules without a tissue diagnosis[J]. Jpn J Clin Oncol, 2011, 41(8): 1017-22. doi: 10.1093/jjco/hyr073 [29] Fan L, Liu SY, Li QC, et al. Multidetector CT features of pulmonary focal ground-glass opacities: differences between benign and malignant[J]. Br J Radiol, 2012, 85(1015): 897-904. doi: 10.1259/bjr/33150223 [30] Lee HJ, Goo JM, Lee CH, et al. Predictive CT findings of malignancy in ground-glass nodules on thin-section chest CT: the effects on radiologist performance[J]. Eur Radiol, 2009, 19(3): 552-60. doi: 10.1007/s00330-008-1188-2 [31] Kim SY, Lee KS, Han J, et al. Persistent pulmonary nodular groundglass opacity at thin-section CT histopathologic comparisons[J]. Radiol, 2007, 245(1): 267-75. doi: 10.1148/radiol.2451061682 [32] Aoki T, Hanamiya M, Uramoto H, et al. Adenocarcinomas with predominant ground-glass opacity: correlation of morphology and molecular biomarkers[J]. Radiology, 2012, 264(2): 590-6. doi: 10.1148/radiol.12111337 [33] Yang Y, Yang Y, Zhou X, et al. EGFR L858R mutation is associated with lung adenocarcinoma patients with dominant ground- glass opacity[J]. Lung Cancer, 2015, 87(3): 272-7. doi: 10.1016/j.lungcan.2014.12.016 [34] Wang TT, Zhang T, Han XX, et al. Impact of the international association for the study of lung cancer/american thoracic society/ european respiratory society classification of stage IA adenocarcinoma of the lung: Correlation between computed tomography images and EGFR and KRAS gene mutations[J]. Exp Ther Med, 2015, 9(6): 2095-103. doi: 10.3892/etm.2015.2422 [35] Nakamura H, Koizumi H, Kimura H, et al. Epidermal growth factor receptor mutations in adenocarcinoma in situ and minimally invasive adenocarcinoma detected using mutation-specific monoclonal antibodie[s J]. Lung Cancer, 2016, 99: 143-7. doi: 10.1016/j.lungcan.2016.07.009 -






 下载:
下载: